A comprehensive investigation into sample extraction and method validation for the identification of antifungal compounds produced by lactic acid bacteria using HPLC-UV/DAD†
Abstract
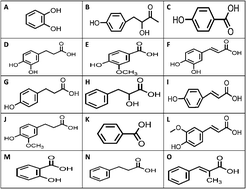
High Performance Liquid Chromatography with Ultra Violet/Diode Array Detection (HPLC-UV/DAD) is an analytical tool that has the potential to develop and advance analytical research and routine analyses in industrially-important Lactic Acid Bacteria (LAB) with the aim of detecting metabolites with antifungal capabilities. Diode array detection is a multi-wavelength detection system that enables the simultaneous collection of absorption data from a range of UV and visible (UV/vis) wavelengths. The majority of antifungal organic compounds have characteristic UV/vis absorption spectra due to the presence of chromophoric groups or structures, this feature can be utilised to assist in identifying and quantifying the components (known and unknown) as they pass through the detector. HPLC-UV/DAD is robust, cheap to purchase and run, easy to maintain, the software is user friendly and automated for quantitation and the methodology can be easily switched/up-scaled to semi-preparative applications for target analyte isolation and purification. However inefficient sample preparation has become the limiting factor for the routine use of HPLC-UV/DAD in the LAB area. This manuscript will outline the development and validation of an HPLC-UV/DAD method that addresses all of the factors that has limited the routine use of this technology for LAB samples. The method has been developed to quantitatively profile fifteen antifungal compounds in LAB strains in a run time of 40 minutes, allowing the assessment of crude samples (direct injection), eliminating sample matrix chromatographic baseline rise effects through the optimisation of liquid–liquid extraction (LLE with average recoveries = 87%) and assessing a widely applied SPE methodology that is shown to yield poor recoveries for the suite of target analytes. The method reported is validated (linear calibration curve: 1–50 μg mL−1; R2 ≥ 0.999; intra-day repeatability gave RSD values (n = 3) ranging from 0–8.8%; inter-day repeatability gave RSD values (n = 9) ranging from 0.3–7.9%; LODs of 0.2–0.6 μg mL−1 and LOQs of 0.5–0.9 μg mL−1 following LLE applied to a range of crude culture broths and extracted LAB cultures, and all results were confirmed against a liquid chromatography (LC) linear ion trap quadrupole (LTQ) Orbitrap hybrid Fourier transform mass spectrometer (FTMS). In summary, through this methodology, HPLC-UV/DAD can be routinely used as a quantitative screening technique for both culture broth (e.g. de Man, Rosaga and Sharpe (MRS)) and LLE-extracted LAB cultures.

 Please wait while we load your content...
Please wait while we load your content...