Solvent free selective oxidation of alcohols catalyzed by a trinuclear complex with a dicopper(ii)–monozinc(ii) centre using hydrogen peroxide as an oxidant†
Abstract
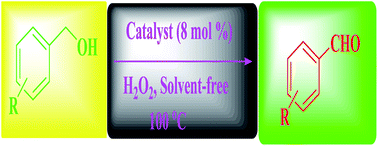
The oxidation of alcohols to carbonyl compounds was investigated by an efficient catalytic system comprising of a new heterotrinuclear complex containing a dicopper(II)–monozinc(II) centre, [ZnCu2(slsch)(NO3)2(H2O)8]·2H2O with hydrogen peroxide under solvent free conditions. Primary and secondary alcohols were oxidized to the corresponding aldehydes and ketones in good to excellent yield. The newly developed catalyst system can be reused in four consecutive runs without any significant loss of activity.

 Please wait while we load your content...
Please wait while we load your content...