Surfactant micelles as microreactors for the synthesis of quinoxalines in water: scope and limitations of surfactant catalysis†‡
Abstract
The scope and limitations of surfactants as catalysts for the synthesis of quinoxalines using microreactors made of the surfactants in water has been assessed. The catalytic potential followed the order: non-ionic surfactants > anionic surfactants > Brønsted acid surfactants > cationic surfactants. The non-ionic surfactant, Tween 40, is the most effective catalyst affording excellent yields within a short reaction time at room temperature and is compatible with different variations of the 1,2-diketones and 1,2-diamines. The reaction medium (spent water) containing the catalyst, as well as the catalyst itself (recovered Tween 40) can be reused for five consecutive reactions. The better catalytic efficiency of the surfactant (Tween 40) compared to the various Lewis/Brønsted acids, as well as the surfactant combined Lewis acid, suggests that surfactants, which generate microreactor assemblies at the interface, are better suited as catalytic aids to promote organic reactions in water. The inferior results obtained in organic solvents, which provide a homogeneous reaction mixture compared to those obtained in water, indicate the specific role of water. This has been depicted as a synergistic dual activation through the hydrogen bond mediated formation of supramolecular assemblies involving a water dimer and the reactants. The catalytic assistance of the surfactant could be ascribed to the ability of the surfactant molecule to undergo hydrophobic and hydrogen bond forming interactions with water and the reactants in orienting the reactants at the water interface and encapsulating inside the microreactors to facilitate the cyclocondensation.
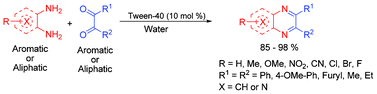

 Please wait while we load your content...
Please wait while we load your content...