A copper-catalysed amidation of aldehydes via N-hydroxysuccinimide ester formation†
Abstract
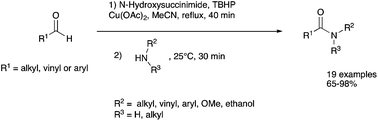
A copper-catalysed oxidative amidation of aldehydes via N-hydroxysuccinimide ester formation is reported. The methodology employed to prepare amides directly from aldehydes has a very wide scope, is high yielding, and does not need dry conditions. This cross-coupling reaction appears to be simple and makes use of cheap, abundant and easily available reagents.

 Please wait while we load your content...
Please wait while we load your content...