The effect of MFI zeolite lamellar and related mesostructures on toluene disproportionation and alkylation†
Abstract
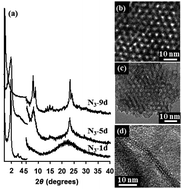
The effect of MFI zeolite nanosheet morphology and related mesostructures on the accessibility of acid sites evaluated from FTIR spectra of different adsorbed probe molecules and conversions and selectivities in toluene disproportionation and alkylation with isopropyl alcohol was investigated. Two-dimensional MFI lamellar zeolite (1.5 nm thickness of layers) and related mesoporous sponge morphologies, most probably with 10-ring channel system not yet structurally fully determined, were synthesized and characterized by XRD, SEM, TEM, and adsorption measurements. FTIR spectroscopy using pyridine, deuterated acetonitrile and 2,6-di-tert-butyl pyridine evidenced high concentrations of accessible Lewis acid sites at the expense of Brønsted acid sites. Ordered mesoporous zeolites exhibited slightly higher toluene conversion in its alkylation with isopropyl alcohol than lamellar MFI or conventional MFI due to easy access to active sites. The lamellar zeolite consisting of less than a unit cell layer was slightly more active than MFI but still exhibited high selectivity to p-cymene. It evidences that alkylation of toluene with isopropyl alcohol proceeds mainly in the 10-ring channel system of MFI zeolite, although the effect of large external surface cannot be neglected. Increased para-selectivity compared with thermodynamic value is achieved even in the case of extremely thin zeolite nanosheets.

 Please wait while we load your content...
Please wait while we load your content...