Solid state crystallization of amorphous calcium carbonate nanoparticles leads to polymorph selectivity†
Abstract
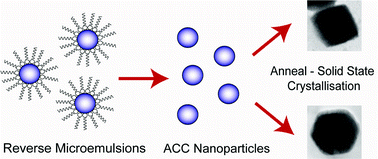
While amorphous calcium carbonate (ACC) has been widely exploited as a precursor to crystalline calcium carbonate in solution, the synthetic potential of crystallising ACC in the solid state has received little attention. Building on our recent results which demonstrate that additives occluded within ACC can facilitate its

 Please wait while we load your content...
Please wait while we load your content...