Simultaneous determination of oxysterols, phytosterols and cholesterol precursors by high performance liquid chromatography tandem mass spectrometry in human serum
Abstract
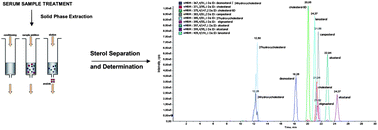
A fast and sensitive high performance liquid chromatography with atmospheric pressure chemical ionization tandem mass spectrometry (HPLC-APCI-MS/MS) method to identify and quantify oxysterols, phytosterols and non-cholesterol sterols at the pico-molar concentration level in human serum in only one run was developed in this study. This method allows the simultaneous separation and quantitation of individual phytosterols, cholesterol precursors and oxidized derivatives of cholesterol without a derivatization step in a single run, thus providing a more confident quantitation of sterols in serum. After saponification, solid-phase extraction (SPE) used as a clean-up step and HPLC separation, detection by MS was developed using APCI and multiple ion monitoring modes. This method employs reversed-phase C18 SPE cartridges and serum calibrators, as well as isotopically labelled cholesterol as an internal standard added before sample processing. The time consumed for a single sample is reduced from the 4 hours of conventional sterol analysis to 1 hour including the chromatographic run time. The method has been evaluated by analyzing a certified cholesterol sample as well as by comparison to other two methods used as reference, based on GC and enzymatic reaction, respectively. Serum from 14 individuals was successfully analyzed. Detection limits for oxysterols, phytosterols and non-cholesterol sterols, all determined in a single run in small serum volumes, were between 0.47 and 1.69 pM. Intra-day precision was <7% for all sterols in in-house-made lipoprotein-deficient serum. The limits of detection showed high sensitivity with very good intraday- and interday-precision for all sterols. The recoveries of the phytosterols, cholesterol precursors and cholestanol ranged from 77% to 92%. These analytical parameters provide a reliable and reproducible method for the identification and quantitation of human sterols.

 Please wait while we load your content...
Please wait while we load your content...