Hydrogen bonded supramolecular polymers in protic solvents: role of multitopicity†
Abstract
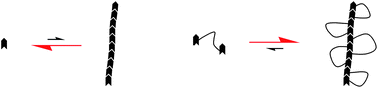
We describe the synthesis of macromolecular
- This article is part of the themed collection: Supramolecular and dynamic covalent polymers
* Corresponding authors
a
UPMC Univ Paris 06, UMR 7610, Chimie des Polymères, 75005 Paris, France
E-mail:
laurent.bouteiller@upmc.fr
b CNRS, UMR 7610, Chimie des Polymères, 75005 Paris, France
c BASF ISIS, 8 Allée Gaspard Monge, 67083 Strasbourg, France
d Laboratoire Léon Brillouin, UMR 12 CNRS-CEA, 91191 Gif-sur-Yvette Cedex, France
We describe the synthesis of macromolecular

 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?
M. Tharcis, T. Breiner, J. Belleney, F. Boué and L. Bouteiller, Polym. Chem., 2012, 3, 3093 DOI: 10.1039/C2PY20287E
To request permission to reproduce material from this article, please go to the Copyright Clearance Center request page.
If you are an author contributing to an RSC publication, you do not need to request permission provided correct acknowledgement is given.
If you are the author of this article, you do not need to request permission to reproduce figures and diagrams provided correct acknowledgement is given. If you want to reproduce the whole article in a third-party publication (excluding your thesis/dissertation for which permission is not required) please go to the Copyright Clearance Center request page.
Read more about how to correctly acknowledge RSC content.
 Fetching data from CrossRef.
Fetching data from CrossRef.
This may take some time to load.
Loading related content
