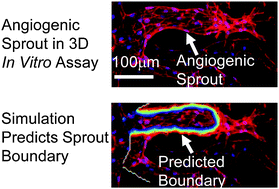
Recent angiogenesis studies have found nascent blood vessels to take on a wide diameter distribution (5–25 μm) during the first 14 days of growth in murine explant models [Nunes S, et al., Microvascular Research, 2010, 79, 1–20], but the mechanisms determining diameter and its reported variability have not been explained. Here, we investigated the dominant mechanisms governing nascent vessel diameter by combining a 3D microfluidic angiogenesis model with a computational reaction–diffusion model. Cultured primary human endothelial cells sprouted from a monolayer into a 3D type I collagen matrix in the microfluidic model, and we found that vessel diameters were inversely correlated with their elongation rates. Several key species of matrix metalloproteinases (MMPs) are known to play important roles in matrix remodelling and vessel growth. We found that selective inhibition of soluble MMP2, but not MMPs 9 and 1, significantly reduced vessel diameters, suggesting that diameter may be MMP2 mediated. Furthermore, immunofluorescent staining for membrane bound membrane type 1 (MT1)-MMP showed that it was localized to active tip cells. Since MT1-MMP is an important activator of MMP2, these findings suggest that MMP2 produced/activated at the tip cell may be a dominant mediator of matrix proteolysis and sprout diameter. To support this hypothesis, we built a computational reaction–diffusion model involving tip cell localized expression of soluble MMPs during sprout elongation. The model, consisting of three parameters based on published values for MMP2 and one quantitatively tuned parameter, reproduced the experimentally observed inverse relationship between elongation rate and vessel diameter. Our experimental findings coupled with our computational model suggest a dominant mechanism by which elongation rate and soluble MMPs govern nascent vessel diameter.

 Please wait while we load your content...
Please wait while we load your content...