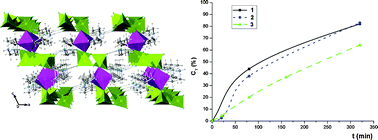
Three new hybrid vanadates have been synthesized under hydrothermal conditions with the formula M(C6H16N3)2(VO3)4, where M = Co(II), Ni(II) and Cu(II). The structural analyses show that the phases are isostructural and crystallize in the monoclinic space groupP21/c. These compounds show a two-dimensional crystal structure, with sheets composed of [VO3]nn− chains and metal centres octahedrally coordinated, chelated by two 1-(2-aminoethyl)piperazonium ligands. The thermal study reveals that the copper containing phase is less stable than the cobalt and nickel containing ones. The IR spectra of the three phases are very similar, with little differences in the inorganic bond region of the copper containing phase. The UV-visible spectra show that the cobalt(II) and the nickel(II) are in slightly distorted octahedral environments. The catalytic tests show that the phases act as heterogeneous catalysts for the selective oxidation of alkyl aryl sulfides, with both H2O2 and tert-butylhydroperoxide as oxidizing agents. The influence of the steric hindrance in the kinetic profile has been studied. The catalytic reactions induce the partial amorphization of the phases.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...