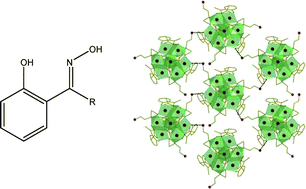
The synthesis and magnetic properties of the compounds [HNEt3][Fe2(OMe)(Ph-sao)2 (Ph-saoH)2]·5MeOH (1·5MeOH), [Fe3O(Et-sao)(O2CPh)5(MeOH)2]·3MeOH (2·3MeOH), [Fe4(Me-sao)4(Me-saoH)4] (3), [HNEt3]2[Fe6O2(Me-sao)4(SO4)2(OMe)4(MeOH)2] (4), [Fe8O3(Me-sao)3(tea)(teaH)3(O2CMe)3] (5), [Fe8O3(Et-sao)3(tea)(teaH)3(O2CMe)3] (6), and [Fe8O3(Ph-sao)3(tea)(teaH)3(O2CMe)3] (7) are reported (Me-saoH2 is 2′-hydroxyacetophenone oxime, Et-saoH2 is 2′-hydroxypropiophenone oxime and Ph-saoH2 is 2-hydroxybenzophenone oxime). 1–7 are the first FeIII compounds synthesised using the derivatised salicylaldoxime ligands, R-saoH2. 1 is prepared by treatment of Fe2(SO4)3·6H2O with Ph-saoH2 in the presence of NEt3 in MeOH; 2 prepared by treatment of Fe(ClO4)2·6H2O with Et-saoH2 and NaO2CPh in the presence of NEt4OH in MeOH; 3 prepared by treatment of Fe(ClO4)2·6H2O with Me-saoH2 and NaO2CCMe3 in the presence of NEt4OH in MeOH; and 4 prepared by treatment of Fe2(SO4)3·6H2O with Me-saoH2 in the presence of NEt3 in MeOH. 4 is a rare example of a polynuclear iron complex containing a coordinated SO42− ion. Compounds 5–7 are prepared by treatment of Fe(O2CMe)2 with Me-saoH2 (5), Et-saoH2 (6), Ph-saoH2 (7) in the presence of H3tea (triethanolamine) in MeOH, and represent the largest nuclearity FeIII clusters containing salicyladoxime-based ligands, joining a surprisingly small family of characterised octanuclear Fe complexes. Variable temperature magnetic susceptibilty measurements of 1,3 and 5–7 reveal all five complexes possess S = 0 spin ground states; 2 possesses an S = 1/2 spin ground state, while 4 has an S = 4 ± 1 spin ground state.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...