Design and synthesis of heterobimetallic donor–acceptor chemodosimetric ensembles for the detection of sulfhydryl-containing amino acids and peptides†
Abstract
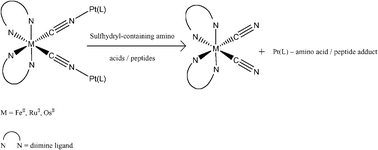
A competitive indicator displacement assay has been successfully developed for the ratiometric determination of sulfhydryl-containing amino acids and peptides using heterobimetallic donor–acceptor complexes as chemodosimetric ensembles. Chromotropic cis-[ML2(CN)2] (M = FeII, RuII, OsII; L = diimine) are used as signaling indicators and PtII(DMSO)Cl2 acceptor moiety is used as the receptor for the sulfhydryl-containing analytes. A series of three heterobimetallic donor–acceptor complexes: cis-FeII(bpy)2[CN–PtII(DMSO)Cl2]2 (1), cis-RuII(bpy)2[CN–PtII(DMSO)Cl2]2 (2) and cis-OsII(bpy)2[CN–PtII(DMSO)Cl2]2 (3) are synthesized and characterized by X-ray crystallography. All the three ensembles are able to produce specific colorimetric/fluorimetric responses to sulfhydryl-containing amino acids (cysteine, homocysteine and methionine) as well as the sulfhydryl-containing small peptide glutathione. The mechanism of the competitive displacement assay is evaluated by examining the thermodynamics of formation of the donor–acceptor linkage and adducts between the acceptor metal and the sulfhydryl-containing analytes as well as by systematic variation of the donor and acceptor metals in the chemodosimetric ensembles.

 Please wait while we load your content...
Please wait while we load your content...