Efficient and convenient entry to β-hydroxy-β-trifluoromethyl-β-substituted ketones and 2,6-disubstituted 4-trifluoromethylpyridines based on the reaction of trifluoromethyl ketones with enamines or imines
Abstract
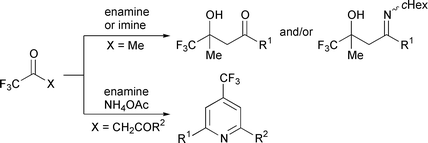
The reactions of trifluoromethyl ketones with enamines or imines are described. The reaction of trifluoroacetone with enamines or imines followed by hydrolysis gave the corresponding β-hydroxy-β-trifluoromethyl-β-methyl ketones in good yields. The reaction of trifluoromethylated β-diketones with enamines in the presence of ammonium acetate gave 4-trifluoromethylated pyridines exclusively in good yields, without any detectable amount of regioisomers.

 Please wait while we load your content...
Please wait while we load your content...