Reactions of 1-(2-alkoxyphenyl)alkaniminyl radicals
Abstract
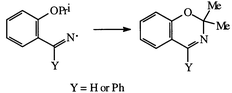
Genereration of the iminyls 1d and 1e by standard methods in the gas-phase by flash vacuum pyrolysis (FVP), or in solution, respectively, gives rise to 1,3-benzoxazines 18 and 22 by a mechanism which probably involves a 1,6-hydrogen atom translocation. Under FVP conditions, 2-cyanophenol 14 is a major product from 2-alkoxyphenylalkaniminyls 1b–d; the dealkylation is an integral part of the reaction mechanism and not a subsequent reaction of 2-alkoxybenzonitriles, which themselves are only very minor products of the pyrolyses.

 Please wait while we load your content...
Please wait while we load your content...