Spatially heterogeneous dynamics of cells in a growing tumor spheroid: comparison between theory and experiments
Abstract
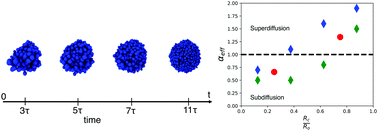
Collective cell movement, characterized by multiple cells that are in contact for substantial periods of time and undergo correlated motion, plays a central role in cancer and embryogenesis. Recent imaging experiments have provided time-dependent traces of individual cells, thus providing an unprecedented picture of tumor spheroid growth. By using simulations of a minimal cell model, we analyze the experimental data that map the movement of cells in a fibrosarcoma tumor spheroid embedded in a collagen matrix. Both simulations and experiments show that cells in the core of the spheroid exhibit subdiffusive glassy dynamics (mean square displacement, Δ(t) ≈ tα with α < 1), whereas cells in the periphery exhibit superdiffusive motion, Δ(t) ≈ tα with α > 1. The motion of most of the cells near the periphery is highly persistent and correlated directional motion due to cell doubling and apoptosis rates, thus explaining the observed superdiffusive behavior. The α values for cells in the core and periphery, extracted from simulations and experiments, are in near quantitative agreement with each other, which is surprising given that no parameter in the model was used to fit the measurements. The qualitatively different dynamics of cells in the core and periphery is captured by the fourth order susceptibility, introduced to characterize metastable states in glass forming systems. Analyses of the velocity autocorrelation of individual cells show remarkable spatial heterogeneity with no two cells exhibiting similar behavior. The prediction that α should depend on the location of the cells in the tumor is amenable to experimental testing. The highly heterogeneous dynamics of cells in the tumor spheroid provides a plausible mechanism for the origin of intratumor heterogeneity.


 Please wait while we load your content...
Please wait while we load your content...