Selective oxidation of alcohols on hydrogen titanate nanotubes under visible light irradiation: relationship between nanostructure and catalytic activity†
Abstract
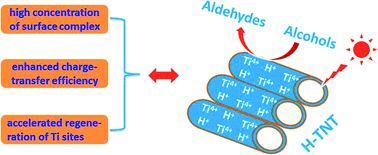
Nanostructured H-titanate materials with different morphologies, including nanotubes, nanosheets and nanofibers, are examined as active photocatalysts for the selective oxidation of alcohols to elucidate the relationship between the structure and the catalytic properties. H-titanate nanotubes (H-TNTs) exhibit superior photocatalytic performance under visible light irradiation to H-titanate nanosheets (H-TNS) and nanofibers (H-TNF) although they have similar crystal structures. N2 adsorption–desorption isotherms and low temperature electron spinning resonance (ESR) spectra indicate that H-TNTs possess a huge specific surface area and a large fraction of unsaturated surface Ti active sites. This facilitates the formation of a high concentration of surface complexes between unsaturated Ti atoms and adsorbed alcohols and consequently enhances the visible-light absorption, which play key roles in achieving high activity for ligand-to-metal charge transfer (LMCT) based photocatalytic systems. The unique structural features of 1D nanotubes promote the charge-transfer efficiency from the coordinated alcohol species to surface Ti atoms, which is evidenced by low temperature ESR under visible light and photoelectrochemical characterizations. Furthermore, pyridine-adsorbed Fourier transform infrared (FT-IR) spectra reveal that there are effective Brønsted acid sites on the surface of H-TNTs, which can accelerate the decomposition of Ti-peroxide species and the regeneration of Ti active sites. In brief, the features of 1D nanotubes, including large surface area, abundant unsaturated surface Ti sites, high concentration of surface complex, strong visible-light absorption, effective charge transfer and Brønsted acidity, contribute to the high photocatalytic activity of H-TNTs.

 Please wait while we load your content...
Please wait while we load your content...