Structure-function correlations in graphene screen-printed electrodes: capacitive and faradaic behaviour
Tharinda
Kasemphong
a,
Monchai
Jitvisate
b,
Chanida
Jakkrawhad
a,
Wichayaporn
Kamsong
c,
Chanpen
Karuwan
c,
Pachanuporn
Sunon
d,
Supinya
Nijpanich
 e,
Phanawan
Whangdee
f,
Theeranun
Siritanon
e,
Phanawan
Whangdee
f,
Theeranun
Siritanon
 a and
Kamonwad
Ngamchuea
a and
Kamonwad
Ngamchuea
 *a
*a
aSchool of Chemistry, Institute of Science, Suranaree University of Technology, 111 University Avenue, Suranaree, Muang, Nakhon Ratchasima 30000, Thailand. E-mail: kamonwad@g.sut.ac.th; Tel: +66 (0) 44 224 637
bSchool of Physics, Institute of Science, Suranaree University of Technology, 111 University Avenue, Suranaree, Muang, Nakhon Ratchasima 30000, Thailand
cNational Nanotechnology Center (NANOTEC), National Science and Technology Development Agency (NSTDA), Pathum Thani 12120, Thailand
dInstitute of Research and Development, Suranaree University of Technology, 111 University Avenue, Suranaree, Muang, Nakhon Ratchasima 30000, Thailand
eSynchrotron Light Research Institute, 111 University Avenue, Suranaree, Muang, Nakhon Ratchasima 30000, Thailand
fDepartment of Applied Physics, Faculty of Sciences and Liberal Arts, Rajamangala University of Technology Isan, Nakhon Ratchasima, 30000, Thailand
First published on 24th December 2025
Abstract
How the origin of graphene influences interfacial behaviour once it is formulate and screen-printed has been unclear. Herein, we fabricate four graphene screen-printed electrodes (commercial, combustion-derived, exfoliated, and CVD-grown) using an identical ink and printing protocol and combine conventional electroanalysis with step-potential electrochemical spectroscopy to obtain assumption-light differential capacitance C(E) and charging timescales τ. All electrodes show U-shaped C(E) with tightly clustered PZCs (0.35–0.40 V vs. Ag/AgCl), indicating that the charge-neutral potential is set primarily by surface chemistry and electronic structure, rather than morphology. However, the extent and speed of interfacial charging vary. The double-layer capacitance (Cdl) scales with wetting and mesoporosity, while the charging time (τ = 15–25 ms across the set) reflects the trade-off between ionic access resistance and capacitance. For the partially inner-sphere redox couple [Fe(CN)6]4−/3−, heterogeneous electron transfer follows edge, defect, and oxygen functionality, rather than film conductivity, with k0 spanning (0.76–1.99) × 10−5 m s−1. These interfacial electrochemical metrics, both capacitive and faradaic, map directly onto physical features such as porosity, defect density, and interfacial chemistry, providing initial criteria for selecting graphene precursors in sensing, catalysis, and energy storage.
1 Introduction
Screen-printed electrodes (SPEs) have emerged as indispensable components in electrochemical sensing and energy technologies, owing to their low production cost, high throughput, and suitability for miniaturized, disposable, and flexible platforms.1,2 Among carbon-based materials, graphene has garnered considerable attention for SPE fabrication due to its excellent electrical conductivity, large specific surface area, and tunable surface chemistry.3 Despite these advantages, a fundamental and unresolved question persists regarding the extent to which the method of graphene synthesis dictates its electrochemical performance when integrated into printed devices.Graphene can be synthesized by a wide range of approaches, from bottom-up chemical vapour deposition (CVD) to top-down exfoliation and even low-cost combustion-derived routes.4 Each method yields materials with distinct structural, chemical, and morphological features e.g. layer number, defect density, oxygen functionality, and stacking order that are known to influence double-layer charging and heterogeneous electron transfer (HET).5 Notably, several studies report that single- and few-layer graphene do not necessarily outperform graphite in electroanalytical measurements as the higher edge density and interlayer defects in multilayer/graphite systems provide abundant active sites, stronger analyte adsorption, and lower noise/ohmic artifacts.6
Beyond layer count, porosity alters the apparent kinetics by creating thin-layer or confinement conditions. Overlapping diffusional fields in pores increase local residence time and near-surface concentration, which can sharpen peaks, reduce peak-to-peak separation (ΔEp), and lower overpotentials, even when the intrinsic electron-transfer barrier is unchanged.7 Defects such as vacancies, Stone–Wales motifs, and sp2/sp3 discontinuities introduce localized electronic states that enhance electronic coupling and adsorption for partially inner-sphere probes. However, excessive oxidation or disorder can increase charge-transfer resistance and promote site blocking.8–10 Surface functionalization tunes work function, wettability, potential of zero charge (PZC), and specific adsorption; for example, oxygenated groups improve hydrophilicity and ion access but can form passivating surface complexes (e.g. with [Fe(CN)6]3−/4−), thereby slowing HET.11,12 The edge-to-basal-plane ratio further governs density of states at the Fermi level and chemisorption propensity, with edge-rich carbons generally exhibiting faster HET than basal-plane-dominated materials.13,14
However, existing reports on graphene SPEs remain fragmented, often focusing on a single graphene source or emphasizing only either capacitive behaviour for supercapacitors or faradaic responses for redox probes, without systematically linking the two regimes. As a result, there is no unified understanding of how the intrinsic properties of graphene, dictated by its synthesis method, affect both the baseline capacitive properties and the electron transfer kinetics when integrated into practical SPE platforms. Furthermore, the dynamic evolution of electrode surface wetting under electrolyte exposure, which directly affects electrochemical responses, is rarely considered in the context of printed graphene electrodes. This knowledge gap carries important implications for both fundamental understanding and practical deployment.
Herein, we present a systematic assessment of the electrochemical behaviour of SPEs fabricated from four distinct graphene sources, combustion-derived, electrochemically exfoliated, CVD-grown, and commercial graphene, using an identical ink formulation and printing process. These sources reflect the range of graphene materials commonly used in printed electrochemical devices, including (bio)sensors, where ink formulation and graphene type affect sensitivity and reproducibility,3,15 and energy devices such as flexible conductors and micro-/planar supercapacitors, where accessible surface area and interfacial resistance govern capacitance and rate performance.16–19 Prior studies have further highlighted that graphene source can introduce significant variability, through factors such as binder interactions and residual metal impurities, which impact electron-transfer kinetics and capacitive behaviour in SPEs.20
By combining structural/chemical characterization with electrochemical analyses and contact-angle measurements in water and concentrated electrolytes over repeated use, our study links microscopic material features and wettability evolution to macroscopic electrode descriptors relevant to practical printed devices (e.g. ion access, double-layer charging, and redox kinetics), providing guidance for selecting graphene sources for sensing and energy-related SPE applications.
2 Experimental
2.1 Chemical reagents
All chemical reagents were used as received without further purification: potassium chloride (KCl, ≥99.0%, Sigma-Aldrich), potassium hexacyanoferrate(II) trihydrate (K4[Fe(CN)6]·3H2O, ≥98.5%, Sigma-Aldrich), potassium hexacyanoferrate(III) (K3[Fe(CN)6], ≥99.0%, Sigma-Aldrich), silver/silver chloride ink (C61003P7, Sun Chemical), carbon paste ink (C2030519P4, Sun Chemical), insulator ink (D2070423P5, Sun Chemical). Deionized water (18.2 MΩ cm) was obtained from an Elga Purelab Ultra system (Elga Labwater, UK). All experiments were conducted at 25 °C unless stated otherwise.2.2 Graphene sources and synthesis
Four distinct types of graphene were investigated: commercial, combustion-derived, electrolytically exfoliated, and CVD-grown. Commercial graphene powder was purchased from UC Bacon Co. Ltd and used as received. Combustion-derived graphene was synthesized following an adaptation of Chen et al.21 Briefly, 3.00 g of magnesium ribbon was placed on a slab of solid CO2 (dry ice), ignited, and immediately covered with a second slab. Combustion in CO2 produced MgO and a carbonaceous by-product. The black solid was collected and stirred in 100.0 mL of 1.00 M HCl at room temperature overnight to remove Mg/MgO, then filtered and rinsed with deionized water to neutral pH. The purified carbon was dried under high vacuum at 100 °C overnight to yield the combustion-derived graphene.21Exfoliated graphene powder was prepared by one-step electrolytic exfoliation.22 Two graphite rods served as electrodes in aqueous polystyrene sulfonate (PSS). A constant 8.0 V bias was applied for 24 h to exfoliate graphite into graphene sheets. The dispersion was washed with ethanol and deionized water to remove excess PSS/ions, then dried at 80 °C for 2 h.22
CVD graphene was grown on FeNi catalyst in a thermal CVD reactor. The FeNi catalyst was prepared by impregnation and reduced at 500 °C for 1 h under 1500 sccm H2 to convert the oxide to its metallic form. The catalyst and a triglyceride-based lipid feedstock (used as the carbon source) were then loaded into a quartz tube furnace and heated to 800 °C at 10 °C min−1 under 1500 sccm N2 flow. Growth proceeded for 1 h at 800 °C, after which the system was cooled to room temperature under 1500 sccm N2. The resulting graphene was collected as powder for ink formulation (see below).
2.3 Fabrication of graphene screen-printed electrodes (SPEs)
A conductive graphene ink was prepared by blending 2.50 g of each type of graphene powder (commercial, combustion, exfoliated, or CVD) with 10.0 g of carbon paste ink in a ball mill (Retsch model Emax, Germany) at 1000 rpm for 60 min. Graphene screen-printed electrodes (SPEs) were fabricated on polyethylene terephthalate (PET) substrates using a screen printer (DEK model 03ix, USA). First, Ag/AgCl ink was screen-printed and dried at 60 °C for 5 min to form the reference electrode (RE). The formulated graphene ink was then printed and dried at 60 °C for 5 min to produce the working (WE) and counter (CE) electrodes. Finally, an insulator ink was printed to define the active electrode area and dried at 120 °C for 5 min. For each batch, a total of 600 SPEs were printed. The corresponding graphene loading is 4.17 mg per SPE.2.4 Characterization
Surface morphology was imaged by field-emission scanning electron microscopy (FE-SEM, AURIGA, Zeiss; 3.0 kV). Crystallinity and stacking order were assessed by powder X-ray diffraction (XRD, D8 ADVANCE, Bruker; Cu Kα, λ = 1.5406 Å). Graphitic and defect signatures (D, G, 2D) were evaluated by Raman spectroscopy (VERTEX 70 with RAM II, Bruker). Surface composition was determined by X-ray photoelectron spectroscopy (XPS, PHI 5000 VersaProbe II, ULVAC-PHI; Al Kα, hν = 1486.6 eV) at the SUT-NANOTEC-SLRI Joint Research Facility (SLRI, Thailand); spectra were processed in PHI MultiPak with Shirley backgrounds and mixed Gaussian–Lorentzian line shapes, using the C 1s peak at 284.8 eV for charge referencing. Textural properties were obtained from nitrogen adsorption–desorption at 77 K (Autosorb 6100, Anton Paar). The specific surface area was calculated by Brunauer–Emmett–Teller (BET) analysis. The pore volume was obtained by non-local density functional theory (NLDFT) physisorption analysis, a statistical-mechanics adsorption model in which the experimental isotherm is represented as a linear combination of theoretical adsorption isotherms computed for pores of different widths.23–25 The mesopore size distribution was evaluated using the Barrett–Joyner–Halenda (BJH) analysis (desorption branch). Samples were degassed at 250 °C for 5 h before measurement.2.5 Electrochemical measurements
Electrochemical studies were performed with an Autolab PGSTAT302N potentiostat (Metrohm, Netherlands). All potentials are reported vs. the on-chip Ag/AgCl unless stated otherwise. Electrochemical impedance spectroscopy (EIS) was performed in a 5.0 mM solution of [Fe(CN)6]3−/4− containing 0.10 M KCl. Measurements were conducted across a frequency range of 1–105 Hz with an applied AC amplitude of 5 mV. Nyquist spectra were fitted to a Randles equivalent circuit to extract the charge-transfer resistance Rct.Time-resolved analysis of double-layer charging employed step-potential electrochemical spectroscopy (SPECS). Prior to measurements, electrodes were conditioned by 50 cyclic voltammetry (CV) cycles in 1.0 M KCl within a non-faradaic window.26 A staircase of ±10 mV potential steps was then applied, and the resulting current–time transients were recorded for all steps and used to determine the double-layer capacitance (Cdl) and the differential capacitance C(E) over the window range. The potential of zero charge (PZC) was identified as the potential at which C(E) reached its minimum value.
Faradaic behaviour was evaluated by CV in a 1.0 mM [Fe(CN)6]4− solution with 0.10 M KCl as the supporting electrolyte at scan rates of 10–200 mV s−1. Peak current–scan-rate (Randles–Ševčík) analysis provided the electroactive area. The kinetic parameters were obtained from Nicholson analysis of ΔEp for k0 and mass-transport-corrected Tafel slopes for β, as detailed in the Results and discussion section.
3 Results and discussion
The graphene synthesis route imprints a distinct combination of structural order, defect density, surface chemistry and mesoscale connectivity, and these features jointly govern the electrochemical behaviour of SPEs. To resolve these links, we examined four representative materials, commercial, combustion-derived, exfoliated graphene, and CVD-grown graphene formulated and printed under identical conditions. By integrating materials characterization and electrochemical analysis, we map how layer stacking, defect/edge architecture and oxygen functionality affect electrolyte wetting and accessibility, the magnitude and potential-dependence of interfacial capacitance, and heterogeneous electron-transfer rates.3.1 Structure, defects, and surface chemistry
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, 0.15% O–C
O, 0.15% O–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O).29 Combustion-derived graphene retains high sp2 (80.95%) with notable oxygen (10.98% C–O, 2.80% C = O, 5.27% O–C
O).29 Combustion-derived graphene retains high sp2 (80.95%) with notable oxygen (10.98% C–O, 2.80% C = O, 5.27% O–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) and a distinctive O 1s feature (∼531.0 eV) consistent with quinone-like groups.30 Exfoliated graphene shows lower sp2 (73.23%) with substantial oxygen functionalities (17.96% C–O, 3.07% C
O) and a distinctive O 1s feature (∼531.0 eV) consistent with quinone-like groups.30 Exfoliated graphene shows lower sp2 (73.23%) with substantial oxygen functionalities (17.96% C–O, 3.07% C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, 5.74% O–C
O, 5.74% O–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), consistent with partial oxidation. CVD graphene exhibits the highest sp2 carbon fraction (85.38%) and minimal oxygen (12.70% C–O, 1.92% C
O), consistent with partial oxidation. CVD graphene exhibits the highest sp2 carbon fraction (85.38%) and minimal oxygen (12.70% C–O, 1.92% C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, 0% O–C
O, 0% O–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O).31 These data therefore indicate how synthesis governs functional group distribution, which are the key determinants of interfacial and electrochemical behaviour.
O).31 These data therefore indicate how synthesis governs functional group distribution, which are the key determinants of interfacial and electrochemical behaviour.
3.2 Surface area and porosity characteristics
The surface area and porosity of the graphene SPEs were characterized by nitrogen adsorption–desorption at 77 K. Specific surface areas were determined using the Brunauer–Emmett–Teller (BET) method, total pore volumes from density functional theory (DFT) analysis, and mesopore size distributions from the Barrett–Joyner–Halenda (BJH) method. The resulting parameters are summarized in Table 1.| Isotherm | A BET (m2 g−1) | V total (cm3 g−1) | r pore (nm) | |
|---|---|---|---|---|
| a A BET reported as mean ± standard deviation. b V total and rpore reported as modal values. | ||||
| Commercial | Type II | 29.2 ± 0.4 | 0.088 | 3.725 |
| Combustion | Type IV | 383.6 ± 2.4 | 0.856 | 3.838 |
| Exfoliation | Type II | 5.8 ± 0.1 | 0.012 | 3.789 |
| CVD | Type II | 14.3 ± 0.2 | 0.036 | 3.773 |
The combustion-derived graphene exhibits a Type IV isotherm, two orders of magnitude larger ABET, and an order-of-magnitude larger Vtotal than the other samples, consistent with a mesoporous network created by disrupted stacking and inter-flake voids during high-temperature formation. In contrast, the commercial, exfoliated, and CVD materials show Type II isotherms with low ABET and small Vtotal, indicative of largely nonporous or macroporous solids with limited accessible mesoporosity.
Across all samples, BJH returns similar mesopore radii (Table 1). For the combustion material, this reflects a dense population of small mesopores distributed throughout the network (hence high ABET and Vtotal). For the Type II samples, the comparable rpore should be interpreted as sparse slit-like gaps between restacked flakes. Although the radius is similar, the density of such features and therefore the accessible surface is much lower, as captured by ABET and Vtotal.
However, only a fraction of ABET contributes to the electrochemically active area, as access is limited by electrolyte penetration, wetting state, and electronic percolation to the current-carrying network. In concentrated electrolytes (e.g. 1.0 M KCl), the electrical double layer is sub-nanometer in thickness, enabling efficient charge storage in mesopores. In contrast, micropores are often underutilized in aqueous media owing to desolvation barriers and steric constraints. Studies have reported a threshold potential at which partial desolvation enables ion adsorption in these pores, which increases the capacitance Cdl.32,33 The potential-dependent capacitance is further discussed in Section 3.4.
3.3 Wetting behaviour and contact-angle evolution
All graphene SPEs exhibited similarly large water contact angles (θH2O = 118–121°, Fig. 3), essentially independent of graphene type, indicating that initial wettability is dominated by the binder-rich surface which limits water affinity. Exposure to an electrolyte alters this interfacial state rapidly. In 1.0 M KCl, the apparent contact angle (θKCl) decreases upon reuse for every material, with cumulative first-to-second-use changes of 38.5° for combustion-derived ((118.1 ± 1.0)° → (79.6 ± 3.4)°), 34.5° for exfoliated graphene ((118.8 ± 1.3)° → (84.3 ± 3.9)°), 34.1° for commercial ((119.4 ± 0.6)° → (85.3 ± 1.8)°), and 25.5° for CVD graphene ((118.5 ± 2.0)° → (93.0 ± 5.6)°), refer to Fig. 3. In contrast, θH2O changes only marginally between the first and second use, highlighting that wettability is governed primarily by ion–surface interactions rather than water.The dependence of the macroscopic angle on interfacial tensions is given by Young's equation,
γSV − γSL = γLVcos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θY, θY, | (1) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θY increases and the apparent angle decreases.34 In 1
θY increases and the apparent angle decreases.34 In 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 electrolytes such as KCl, the electrostatic screening (Debye) length is extremely short (κ−1 ≈ 0.3 nm at 1.0 M KCl and 298 K), which promotes close approach of ions/water to polar/oxygenated sites and thus further reduces γSL. In addition, the intrinsic roughness and inter-flake voids of printed films favour capillary infiltration. As trapped air is replaced by liquid, the real solid–liquid contact fraction increases, and the macroscopic angle falls accordingly.35,36
1 electrolytes such as KCl, the electrostatic screening (Debye) length is extremely short (κ−1 ≈ 0.3 nm at 1.0 M KCl and 298 K), which promotes close approach of ions/water to polar/oxygenated sites and thus further reduces γSL. In addition, the intrinsic roughness and inter-flake voids of printed films favour capillary infiltration. As trapped air is replaced by liquid, the real solid–liquid contact fraction increases, and the macroscopic angle falls accordingly.35,36
The material-dependent ranking in changes in contact angle (ΔθKCl) is consistent with their structural and chemical attributes. The combustion-derived graphene combines abundant oxygenated sites with a dense, accessible mesopore population, enabling extensive electrolyte infiltration and therefore most effectively increases the true solid–liquid contact fraction. Exfoliated and commercial graphene, which are oxygen-rich and edge-dense, provides numerous polar sites (C–O, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, O–C
O, O–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) that lower the solid–liquid interfacial free energy via hydrogen bonding and specific adsorption of hydrated ions (particularly K+ at negatively polarized/oxygenated sites), therefore also producing a large drop in θKCl. In contrast, CVD graphene, dominated by cleaner basal planes with minimal oxygen functionality and fewer capillary pathways, offers fewer sites for ion–surface interactions and exhibits the smallest wetting response, and hence the most angle-stable.
O) that lower the solid–liquid interfacial free energy via hydrogen bonding and specific adsorption of hydrated ions (particularly K+ at negatively polarized/oxygenated sites), therefore also producing a large drop in θKCl. In contrast, CVD graphene, dominated by cleaner basal planes with minimal oxygen functionality and fewer capillary pathways, offers fewer sites for ion–surface interactions and exhibits the smallest wetting response, and hence the most angle-stable.
To isolate ion-identity effects beyond graphene type, we further measured contact angles for additional 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 electrolytes using the combustion-derived graphene SPE. The first use yields similarly high contact angles across all salts (θ = 119–121°), reinforcing that the initial state is binder-dominated. Upon reuse, however, the wetting evolution becomes strongly ion-specific. In the cation series at fixed Cl− (LiCl, NaCl, KCl, RbCl), the cumulative decrease over repeated use is modest for Li+ (Δθ = 24.5°) and Na+ (Δθ = 25.0°), but becomes larger for K+ (Δθ = 38.5°) and Rb+ (Δθ = 47.8°), consistent with enhanced specific-ion contributions for larger, more weakly hydrated and more polarizable cations that can more effectively lower the effective γSL and promote greater penetration into interflake voids during repeated wetting. In the anion series at fixed K+ (KCl, KBr, KI), the corresponding decreases follow Cl− (Δθ = 38.5°) > Br− (Δθ = 24.5°) > I− (Δθ = 5.9°), indicating that anion identity also strongly controls how readily the interface transitions to a more wetted state upon reuse. Notably, prior studies on hydrophobic interfaces emphasise that specific-ion trends can be system- and mechanism-dependent and may not follow a single universal ordering, reflecting a combination of ion-specific interfacial affinity and dynamic interfacial processes. Practically, these data show that while the initial contact angle is set by the printed binder layer, the evolution of wetting with reuse is governed by ion-specific interactions and interfacial partitioning previously reported for hydrophobic/aqueous interfaces.37–39
1 electrolytes using the combustion-derived graphene SPE. The first use yields similarly high contact angles across all salts (θ = 119–121°), reinforcing that the initial state is binder-dominated. Upon reuse, however, the wetting evolution becomes strongly ion-specific. In the cation series at fixed Cl− (LiCl, NaCl, KCl, RbCl), the cumulative decrease over repeated use is modest for Li+ (Δθ = 24.5°) and Na+ (Δθ = 25.0°), but becomes larger for K+ (Δθ = 38.5°) and Rb+ (Δθ = 47.8°), consistent with enhanced specific-ion contributions for larger, more weakly hydrated and more polarizable cations that can more effectively lower the effective γSL and promote greater penetration into interflake voids during repeated wetting. In the anion series at fixed K+ (KCl, KBr, KI), the corresponding decreases follow Cl− (Δθ = 38.5°) > Br− (Δθ = 24.5°) > I− (Δθ = 5.9°), indicating that anion identity also strongly controls how readily the interface transitions to a more wetted state upon reuse. Notably, prior studies on hydrophobic interfaces emphasise that specific-ion trends can be system- and mechanism-dependent and may not follow a single universal ordering, reflecting a combination of ion-specific interfacial affinity and dynamic interfacial processes. Practically, these data show that while the initial contact angle is set by the printed binder layer, the evolution of wetting with reuse is governed by ion-specific interactions and interfacial partitioning previously reported for hydrophobic/aqueous interfaces.37–39
3.4 Electrical transport
Four-point probe measurements establish a clear conductivity hierarchy for the printed graphene films: combustion (6.50 ± 1.21 kS m−1) > CVD (4.46 ± 1.65 kS m−1) > commercial (3.42 ± 1.34 kS m−1) > exfoliated (2.72 ± 1.18 kS m−1). These values are in the same order of magnitude with other reported graphene/carbon inks.16,40–43 The high conductivity of the combustion graphene suggests an efficient percolation network with strong interflake coupling, likely aided by broad flake sizes and partial graphitization that maximize lateral contacts and minimize resistive junctions. CVD graphene, although intrinsically highly conductive, shows lower conductivity possibly due to folds and grain boundaries interrupt long-range pathways. The mesoscale assembly and interflake contact quality therefore dominate macroscopic transport in printed films,36 outweighing the intrinsic properties of the graphene source. Commercial and exfoliated graphene deliver moderate conductivities, consistent with smaller lateral domains, limited π-conjugation, higher edge fraction, and oxygenated groups that raise tunneling barriers.3.5 Charge transfer resistance
Electrochemical impedance spectroscopy (EIS) of graphene SPEs was recorded in 5.0 mM [Fe(CN)6]3−/4− with 0.10 M KCl (AC amplitude 5 mV; 1–105 Hz). Nyquist plots (Fig. 4a) show a small high-frequency intercept (solution resistance, Rs), a semicircle at intermediate frequencies associated with interfacial charge transfer, and a low-frequency diffusional tail. Spectra were fitted with a Randles-type equivalent circuit (Fig. 4a, inset). The extracted charge-transfer resistances (Rct) are 295 ± 14 Ω for combustion, 354 ± 6 Ω for commercial, 368 ± 36 Ω for exfoliated, and 474 ± 13 Ω for CVD graphene.Two points follow directly from these data. First, there is no direct correlation between Rct and the bulk film conductivity (Section 3.4). For example, the CVD electrode exhibits relatively high electrical conductivity yet the largest Rct, indicating that through-film transport does not dictate interfacial charge-transfer rates. Second, the Rct ranking mirrors trends in wetting behaviour and porosity (Section 3.3), with the combustion-derived sample showing the lowest Rct due to a combination of high mesoporosity, good electrolyte accessibility, and a surface enriched in edge and defect sites. In contrast, the CVD sample, composed largely of smooth basal planes with low oxygen functionality and minimal capillary infiltration, displays the highest Rct.
These observations therefore indicate that charge-transfer performance in printed graphene electrodes is governed by interfacial site chemistry and electrolyte accessibility rather than bulk conductivity. Related parameters such as double-layer capacitance (Section 3.6) and kinetic constants (Section 3.7) further quantify these interfacial effects.
3.6 Capacitive behaviour
The electrochemical double-layer (EDL) behaviour of the graphene SPEs was evaluated by CV in 1.0 M KCl across a non-faradaic window. After 50 consecutive scans to stabilize the interfacial structure, the voltammograms retained an ideal rectangular shape (Fig. 4b), confirming that double-layer charging dominates under these conditions. These CV data provide an overview of the interfacial capacitance and motivate a transient analysis to resolve how charge is stored in space and time.To deconvolute the interfacial processes, we employed step potential electrochemical spectroscopy (SPECS), where a staircase of small potential steps (±10 mV) yields current–time transients, each fitted to ref. 26
| IT = Idl + Id + Ir, | (2) |
The double-layer term captures capacitive charging immediately after each step, and is described by:
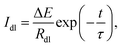 | (3) |
| τ = RdlCdl, | (4) |
The slower relaxation of the near-surface concentration field is captured by a semi-infinite diffusion term:
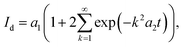 | (5) |
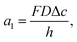 | (6) |
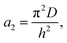 | (7) |
A direct comparison of the I–t transients in Fig. 4(c)–(f) provides insight into how each printed graphene electrode stores and releases charge under identical perturbations. The initial current immediately after each step reflects the magnitude of interfacial charging (i.e. electrolyte-accessible capacitance/area), while the decay rate reflects the longer-range charging/ion-access and hence how rapidly the interface stabilizes, which is relevant to rate performance in energy storage and response times in sensing. Any persistent offsets or drift indicate non-ideal background contributions that can compromise baseline stability in low-signal sensing. These qualitative differences motivate the quantitative fitting described below to extract Cdl, Rdl, and τ (and their potential dependence) for comparison across graphene sources.
Analyzing the measured charging current with eqn (2) results in fitting parameters that are used for surface charge calculation in the following procedure. The surface charge of the jth potential step can be determined by
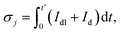 | (8) |
The cumulative summation of the surface charge at each potential step yields the potential-dependent surface charge:
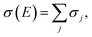 | (9) |
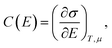 | (10) |
The differential capacitance curves C(E) are shown in Fig. 5. These capacitance values are reported as measured from SPECS transients and are not normalized by surface area; both BET-derived area and electroactive surface area metrics are provided in Table 2 for reference. The overall appearance of the curves in most cases is U-like shape, which can be expected from carbon/aqueous electrolyte systems. Note that we scanned the electrode potential in both cathodic and anodic directions to observe whether the hysteresis occurs in our studied systems. It is clear from the plots that our C(E) data exhibits, if not identical, similar trend for both scan directions in all cases, indicating very small hysteresis. It must also be noted here that the measured capacitance is the total capacitance of the series combination of the electrolyte double layer capacitance and the electrode capacitance. Since carbon, unlike perfect metal, is known to have limited density of states. This gives rise to the penetration of an electric field into the materials, generating the space charge polarization. With this in mind, if one of the capacitances of the two components is considerably smaller than another one, it will be pronounced in the total capacitance.
 | ||
| Fig. 5 Interfacial double-layer capacitance as a function of potential extracted from SPECS in 1.0 M KCl for (a) commercial, (b) combustion-derived, (c) exfoliated, and (d) CVD-grown graphene. | ||
| Graphene type | Morphology | Edge exposure | Layers | Defect level | Surface functionality | A BET (m2 g−1) | |||
|---|---|---|---|---|---|---|---|---|---|
| sp2 C (%) | C–O (%) | C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O (%) O (%) |
O–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O (%) O (%) |
||||||
| Note: All SPE types have the same graphene mass loading per electrode (4.17 mg graphene per SPE). | |||||||||
| (A) Structural and chemical properties | |||||||||
| Commercial | Stacked sheets | High | Multi-layer | High | 75.36 | 15.81 | 3.03 | 0.15 | 29.2 ± 0.4 |
| Combustion | Mesoporous | High | Few-layer | Moderate | 80.95 | 10.98 | 2.80 | 5.27 | 383.6 ± 2.4 |
| Exfoliation | Stacked sheets | High | Multi-layer | Low | 73.23 | 17.96 | 3.07 | 5.74 | 5.8 ± 0.1 |
| CVD | Sheet | Low, mostly basal | Few-layer | Moderate | 85.38 | 12.70 | 1.92 | 0.00 | 14.3 ± 0.2 |
| Graphene type | σ (kS m−1) | θ KCl (°) | PZC (V) | C dl (µF) | R ct (Ω) | A CV,rev (10−6 m2) | k 0 (10−5 m s−1) | β | |
|---|---|---|---|---|---|---|---|---|---|
| 1st | 2nd | ||||||||
| (B) Electronic, wetting, and interfacial electrochemical properties | |||||||||
| Commercial | 3.42 ± 1.34 | 119.4 ± 0.6 | 85.3 ± 1.8 | 0.40 ± 0.06 | 0.83 ± 0.02 | 354 ± 6 | 8.02 ± 0.08 | 1.03 ± 0.06 | 0.86 ± 0.01 |
| Combustion | 6.50 ± 1.21 | 118.1 ± 1.0 | 79.6 ± 3.4 | 0.38 ± 0.01 | 2.47 ± 0.17 | 295 ± 14 | 8.71 ± 0.03 | 1.20 ± 0.09 | 0.83 ± 0.01 |
| Exfoliation | 2.72 ± 1.18 | 118.8 ± 1.3 | 84.3 ± 3.9 | 0.39 ± 0.01 | 1.14 ± 0.03 | 368 ± 36 | 8.43 ± 0.10 | 1.99 ± 0.26 | 0.81 ± 0.01 |
| CVD | 4.46 ± 1.65 | 118.5 ± 2.0 | 93.0 ± 5.6 | 0.35 ± 0.04 | 0.64 ± 0.02 | 474 ± 13 | 7.82 ± 0.04 | 0.76 ± 0.03 | 0.76 ± 0.01 |
From the C(E) profile, the potential of zero charge (PZC) is located at the minimum of C(E). PZCs cluster narrowly at 0.38–0.40 V vs. Ag/AgCl for all the graphene SPEs (commercial 0.40 ± 0.06 V, exfoliated 0.39 ± 0.01 V, combustion 0.38 ± 0.01 V, CVD 0.35 ± 0.04 V). The relatively invariant PZC reflects the fact that it is primarily dictated by the surface chemical composition, such as functional groups and specific ion adsorption.
Although the PZC varies modestly, the capacitance at the PZC differs markedly: commercial 0.83 ± 0.02 µF, combustion 2.47 ± 0.17 µF, exfoliated 1.14 ± 0.03 µF, and CVD 0.64 ± 0.02 µF. Normalized by the electroactive surface area (determined using [Fe(CN)6]4−/3− in the next section), the average specific capacitances are 10.3, 32.3, 13.5, and 8.3 µF cm−2 for commercial, combustion, exfoliated, and CVD, respectively, typical of well-wetted carbons in aqueous electrolyte.45 The commercial and exfoliated electrodes show comparable capacitance, consistent with their similar surface structures, whereas combustion graphene exhibits over two-fold increase in capacitance, indicating a distinct surface structure that enhances electrolyte accessibility, and this result may indicate the role of surface structure in modulating capacitive behaviour in carbon materials.46
In the high-ionic-strength limit (Debye length ≪ pore size), pores larger than a few nanometres host non-overlapping double layers. Under these conditions, Cdl scales with the electrolyte-accessible surface area rather than with the total surface area measured by gas adsorption (ABET).47 Improved wetting increases the contact area between electrolyte and electrode but does not alter the intrinsic surface potential at which net charge vanishes. Consequently, significant changes in Cdl can occur without appreciable shifts in PZC.
The charging resistances at the PZC further differentiate the electrodes: Rdl (PZC) = 6.1 ± 0.4 kΩ (combustion), 22.1 ± 0.6 kΩ (exfoliated), 28.6 ± 0.7 kΩ (commercial), and 30.5 ± 0.9 kΩ (CVD). Therefore, the combustion graphene couples high Cdl with low Rdl, indicative of a percolated, well-wetted pore network, whereas the CVD film combines the smallest Cdl with the largest Rdl, consistent with limited electrolyte accessibility and high access resistance.
The corresponding charging time constant (τ) lies in the 15–25 ms range across electrodes (Fig. 6), explaining why CVs appear nearly rectangular at relatively slow scan rates (t ≫ τ). τ(E) exhibits shallow U-shaped profiles that mirror C(E) with minima occur near the PZC. Considering C(E) alongside τ(E) separates how much charge is stored from how fast it is stored. Combustion graphene exhibits high Cdl with the smallest Rdl and τ, consistent with rapid, well-accessed charging. Thus, porosity does not necessarily slow charging. While microporous or poorly wetted carbons can produce long, multi-exponential relaxations, a mesoporous, well-wetted architecture, as in this combustion material, can reduce Rdl enough to shorten τ even as Cdl increases. Commercial and exfoliated electrodes show intermediate τ, consistent with comparable surface structures and moderate Rdl. CVD graphene with the smallest Cdl but the largest Rdl shows comparatively slow charging due to limited accessibility.
 | ||
| Fig. 6 Charging time constant (τ) as a function of potential extracted from SPECS in 1.0 M KCl for (a) commercial, (b) combustion-derived, (c) exfoliated, and (d) CVD-grown graphene. | ||
3.7 Faradaic behaviour
The [Fe(CN)6]4−/3− redox couple, while formally reversible, is well recognized to proceed via a partially inner-sphere electron transfer pathway on carbon-based electrodes, rendering its kinetics highly sensitive to surface chemistry and local microstructure.11,13 This property makes it an ideal probe for elucidating interfacial differences among printed graphene electrodes. Representative cyclic voltammograms collected at 50 mV s−1 (Fig. 7a) revealed subtle but reproducible variations in peak shape and separation across the different types of graphene, suggesting differences in their ability to facilitate electron exchange. Further investigation of the scan rate dependence (10–200 mV s−1), illustrated in Fig. 7b for the combustion-derived system, showed linear increases in peak current with the square root of the scan rate (Fig. 7b, inset), confirming a diffusion-controlled process for all electrode types.The electroactive surface area (A) was initially estimated using the reversible Randles–Ševčík relationship (eqn (11)),48 giving comparable values across the electrodes: (8.02 ± 0.08) × 10−6 m2 for commercial, (8.71 ± 0.03) × 10−6 m2 for combustion, (8.43 ± 0.10) × 10−6 m2 for exfoliated, and (7.82 ± 0.04) × 10−6 m2 for CVD graphene. Estimates using the totally irreversible limiting form (eqn (12), with β = 0.5) are larger in all cases: (1.44 ± 0.01) × 10−5 m2 for commercial, (1.57 ± 0.01) × 10−5 m2 for combustion, (1.52 ± 0.02) × 10−5 m2 for exfoliated, and (1.41 ± 0.01) × 10−5 m2 for CVD graphene. To evaluate these limiting assumptions, we performed numerical simulations (refer to SI) and found that the experimental response lies between the reversible and totally irreversible limits, consistent with quasi-reversible behaviour of [Fe(CN)6]4−/3−. Therefore, neither limiting form is appropriate for accurate determination of A. Nonetheless, applying a single expression consistently across all electrodes provides a useful comparative (‘apparent’) electroactive area for benchmarking between SPE types.
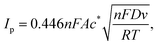 | (11) |
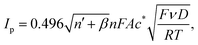 | (12) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 485 C mol−1), D is the diffusion coefficient, R is the universal gas constant (8.314 J K−1 mol−1), T is the absolute temperature, n′ is the number of electron transfer before the rate-determining step, and β is the anodic transfer coefficient of the rate-determining step.
485 C mol−1), D is the diffusion coefficient, R is the universal gas constant (8.314 J K−1 mol−1), T is the absolute temperature, n′ is the number of electron transfer before the rate-determining step, and β is the anodic transfer coefficient of the rate-determining step.
To quantify kinetic differences, the apparent heterogeneous electron-transfer rate constant (k0) was extracted using Nicholson's approach:49,50
| ψ = (−0.6288 + 0.021ΔEp)/(1 − 0.017ΔEp), | (13) |
| ψ = k0[πDnvF/RT]−1/2, | (14) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 485 C mol−1), υ is the potential scan rate, R is the universal gas constant (8.314 J K−1 mol−1), and T is the temperature.
485 C mol−1), υ is the potential scan rate, R is the universal gas constant (8.314 J K−1 mol−1), and T is the temperature.
The exfoliated graphene electrode exhibited the highest rate constant, at (1.99 ± 0.26) × 10−5 m s−1, significantly outperforming the CVD-based electrode, which showed the lowest value of (0.76 ± 0.03) × 10−5 m s−1. The combustion-derived and commercial electrodes demonstrated intermediate behaviour, with k0 values of (1.20 ± 0.09) × 10−5 m s−1 and (1.03 ± 0.06) × 10−5 m s−1, respectively. These values fall within the established range for graphene-based electrodes and are consistent with prior reports demonstrating that electron transfer kinetics for [Fe(CN)6]4−/3− vary significantly with graphene type and structure.5,52–54 Notably, literature comparisons show k0 values of 1.81 × 10−5, 3.25 × 10−5, 19.3 × 10−5, and 53.2 × 10−5 m s−1 for monolayer graphene, quasi-graphene, basal-plane pyrolytic graphite (BPPG), and edge-plane pyrolytic graphite (EPPG), respectively.13 This well-established trend confirms that k0 scales with the abundance of edge sites and surface defects that facilitate inner-sphere pathways.
The superior performance of the exfoliated and combustion-derived graphene electrodes can be rationalized by their structural features: both materials comprise multilayer graphene55 with a high density of exposed edge planes, defect sites, and surface functionalities; factors known to enhance electronic coupling and enable more facile redox interactions.54 In contrast, CVD-grown graphene predominantly consists of few-layer structures with extensive basal-plane coverage and minimal edge exposure. This leads to significantly slower kinetics, as basal planes are intrinsically less reactive toward partially inner-sphere redox couples like [Fe(CN)6]4−/3−.
The underlying reason for the kinetic trends lies in how activation energy for electron transfer is modulated by the electrode's surface features. According to Butler–Volmer and transition state theory, the activation energy for the anodic process of [Fe(CN)6]4−/3− (n = 1) under applied overpotential (η) is:
| ΔG‡ = ΔG‡0 − βFη, | (15) |
We also estimated β using mass-transport-corrected Tafel analysis:56
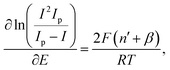 | (16) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 485 C mol−1), and T is the absolute temperature.
485 C mol−1), and T is the absolute temperature.
The apparent anodic transfer coefficients (β) derived from this treatment were 0.86 ± 0.01 for commercial, 0.83 ± 0.01 for combustion, 0.81 ± 0.01 for exfoliated, and 0.76 ± 0.01 for CVD graphene. However, the experimental voltammograms are highly symmetric and simulations that reproduce the peak positions and overall wave shape are best described by β ≈ 0.5 for all electrodes (refer to SI). We therefore treat the Tafel-derived β values as apparent parameters, noting that surface heterogeneity, uncompensated resistance, and deviations from the ideal assumptions can bias the extracted slopes. In contrast, the observed variation in k0 is robust and is attributed primarily to differences in electronic coupling and the density/distribution of electroactive sites, rather than changes in β.
Table 2 consolidates the structural, wetting, capacitive, and kinetic parameters for the four graphene SPEs, enabling direct comparison of trends discussed in Sections 3.3–3.7 and Fig. 3–7. This synopsis highlights the co-variation of interfacial electrochemical metrics with porosity, edge and defect density, and surface chemistry.
4 Conclusions
Using a common ink formulation and printing protocol, we have shown that the graphene synthesis route dictates route-specific electrochemical behaviour in SPEs. All materials exhibit U-shaped C(E) with narrowly clustered PZCs (0.35–0.40 V vs. Ag/AgCl), indicating that surface chemistry and electronic structure, rather than morphology, sets the charge-neutral potential. Nevertheless, the magnitude and kinetics of charging diverge: combustion-derived graphene combines the highest Cdl with the lowest, Rdl, yielding the fastest interfacial charging, whereas CVD shows the opposite, consistent with limited electrolyte accessibility. In sufficiently supported electrolyte (Debye length ≪ pore size), Cdl scales with electrolyte-accessible area rather than ABET, and improved wetting increases Cdl without shifting PZC. For the partially inner-sphere couple [Fe(CN)6]4−/3−, heterogeneous electron transfer follows edge and defect abundance: exfoliated ≳ combustion > commercial ≫ CVD, while bulk film conductivity is a poor predictor of interfacial kinetics. Overall, C(E) and τ(E) quantify charge storage and charging timescales, and k0 captures kinetic facility, providing rigorous criteria to prioritize open, well-wetted mesoporosity for rapid charging and edge-rich, moderately functionalized surfaces for inner-sphere sensing, principles directly transferable to printed carbon electrodes for sensing, energy storage, and electrocatalysis.Conflicts of interest
The authors declare no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Data availability
The data used to support the findings of this study are included within the manuscript.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5cp03833b.
Acknowledgements
This work is supported by (i) Suranaree University of Technology (SUT), (ii) Thailand Science Research and Innovation (TSRI) and (iii) National Science, Research and Innovation Fund (NSRF) (NRIIS number 215530).References
- J. P. Metters, R. O. Kadara and C. E. Banks, New directions in screen printed electroanalytical sensors: an overview of recent developments, Analyst, 2011, 136(6), 1067–1076 RSC.
- P. Fanjul-Bolado, D. Hernández-Santos, P. J. Lamas-Ardisana, A. Martín-Pernía and A. Costa-García, Electrochemical characterization of screen-printed and conventional carbon paste electrodes, Electrochim. Acta, 2008, 53(10), 3635–3642 CrossRef.
- E. P. Randviir, D. A. Brownson, J. P. Metters, R. O. Kadara and C. E. Banks, The fabrication, characterisation and electrochemical investigation of screen-printed graphene electrodes, Phys. Chem. Chem. Phys., 2014, 16(10), 4598–4611 RSC.
- M. S. A. Bhuyan, M. N. Uddin, M. M. Islam, F. A. Bipasha and S. S. Hossain, Synthesis of graphene, Int. Nano Lett., 2016, 6(2), 65–83 CrossRef.
- A. Ambrosi, C. K. Chua, A. Bonanni and M. Pumera, Electrochemistry of graphene and related materials, Chem. Rev., 2014, 114(14), 7150–7188 CrossRef PubMed.
- M. S. Goh and M. Pumera, Single-, few-, and multilayer graphene not exhibiting significant advantages over graphite microparticles in electroanalysis, Anal. Chem., 2010, 82(19), 8367–8370 CrossRef PubMed.
- C. Punckt, M. A. Pope, J. Liu, Y. Lin and I. A. Aksay, Electrochemical performance of graphene as effected by electrode porosity and graphene functionalization, Electroanalysis, 2010, 22(23), 2834–2841 CrossRef.
- P. Wei, J. Shen, K. Wu and N. Yang, Defect-dependent electrochemistry of exfoliated graphene layers, Carbon, 2019, 154, 125–131 CrossRef.
- Y. Jia, L. Zhang, A. Du, G. Gao, J. Chen, X. Yan, C. L. Brown and X. Yao, Defect graphene as a trifunctional catalyst for electrochemical reactions, Adv. Mater., 2016, 28(43), 9532–9538 CrossRef PubMed.
- S. K. Tiwari, S. K. Pandey, R. Pandey, N. Wang, M. Bystrzejewski, Y. K. Mishra and Y. Zhu, Stone–Wales defect in graphene, Small, 2023, 19(44), 2303340 CrossRef PubMed.
- X. Ji, C. E. Banks, A. Crossley and R. G. Compton, Oxygenated edge plane sites slow the electron transfer of the ferro-/ferricyanide redox couple at graphite electrodes, ChemPhysChem, 2006, 7(6), 1337–1344 CrossRef PubMed.
- R. Jain and S. Mishra, Electrical and electrochemical properties of graphene modulated through surface functionalization, RSC Adv., 2016, 6(33), 27404–27415 RSC.
- D. A. C. Brownson, S. A. Varey, F. Hussain, S. J. Haigh and C. E. Banks, Electrochemical properties of CVD grown pristine graphene: monolayer- vs. quasi-graphene, Nanoscale, 2014, 6(3), 1607–1621 RSC.
- C. C. Neumann, C. Batchelor-McAuley, C. Downing and R. G. Compton, Anthraquinone monosulfonate adsorbed on graphite shows two very different rates of electron transfer: surface heterogeneity due to basal and edge plane sites, Chem. – Eur. J., 2011, 17(26), 7320–7326 CrossRef PubMed.
- S. Cinti and F. Arduini, Graphene-based screen-printed electrochemical (bio) sensors and their applications: Efforts and criticisms, Biosens. Bioelectron., 2017, 89, 107–122 CrossRef PubMed.
- P. He, J. Cao, H. Ding, C. Liu, J. Neilson, Z. Li, I. A. Kinloch and B. Derby, Screen-Printing of a Highly Conductive Graphene Ink for Flexible Printed Electronics, ACS Appl. Mater. Interfaces, 2019, 11(35), 32225–32234 CrossRef PubMed.
- T. Sang Tran, N. K. Dutta and N. Roy Choudhury, Graphene-based inks for printing of planar micro-supercapacitors: a review, Materials, 2019, 12(6), 978 CrossRef PubMed.
- Y. Wang, X. Zhang, Y. Zhu, X. Li and Z. Shen, Planar micro-supercapacitors with high power density screen-printed by aqueous graphene conductive ink, Materials, 2024, 17(16), 4021 CrossRef PubMed.
- Y. Wang, X. Zhang, Y. Zhu, X. Li and Z. Shen, Planar Micro-Supercapacitors with High Power Density Screen-Printed by Aqueous Graphene Conductive Ink, Materials, 2024, 17(16), 4021 CrossRef.
- J. P. Smith, C. W. Foster, J. P. Metters, O. B. Sutcliffe and C. E. Banks, Metallic impurities in graphene screen-printed electrodes can influence their electrochemical properties, Electroanalysis, 2014, 26(11), 2429–2433 CrossRef.
- A. Chakrabarti, J. Lu, J. C. Skrabutenas, T. Xu, Z. Xiao, J. A. Maguire and N. S. Hosmane, Conversion of carbon dioxide to few-layer graphene, J. Mater. Chem., 2011, 21(26), 9491–9493 RSC.
- C. Karuwan, A. Wisitsoraat, D. Phokharatkul, C. Sriprachuabwong, T. Lomas, D. Nacapricha and A. Tuantranont, A disposable screen printed graphene–carbon paste electrode and its application in electrochemical sensing, RSC Adv., 2013, 3(48), 25792–25799 RSC.
- R. Bardestani, G. S. Patience and S. Kaliaguine, Experimental methods in chemical engineering: specific surface area and pore size distribution measurements—BET, BJH, and DFT, Can. J. Chem. Eng., 2019, 97(11), 2781–2791 CrossRef.
- J. Landers, G. Y. Gor and A. V. Neimark, Density functional theory methods for characterization of porous materials, Colloids Surf., A, 2013, 437, 3–32 CrossRef.
- N. A. Seaton, J. P. R. B. Walton and N. quirke, A new analysis method for the determination of the pore size distribution of porous carbons from nitrogen adsorption measurements, Carbon, 1989, 27(6), 853–861 CrossRef.
- S. B. Davey, A. P. Cameron, K. G. Latham and S. W. Donne, Electrical double layer formation on glassy carbon in aqueous solution, Electrochim. Acta, 2021, 386, 138416 CrossRef.
- H. Nishihara, H. Fujimoto, H. Itoi, K. Nomura, H. Tanaka, M. T. Miyahara, P. A. Bonnaud, R. Miura, A. Suzuki and N. Miyamoto, Graphene-based ordered framework with a diverse range of carbon polygons formed in zeolite nanochannels, Carbon, 2018, 129, 854–862 CrossRef.
- C. Rao, K. Biswas, K. Subrahmanyam and A. Govindaraj, Graphene, the new nanocarbon, J. Mater. Chem., 2009, 19(17), 2457–2469 RSC.
- T. Reiss, K. Hjelt and A. Ferrari, Graphene is on track to deliver on its promises, Nat. Nanotechnol., 2019, 14, 907–910 CrossRef CAS.
- S. J. R. Prabakar, Y.-H. Hwang, B. Lee, K.-S. Sohn and M. Pyo, Graphene-Sandwiched LiNi0.5Mn1.5O4 Cathode Composites for Enhanced High Voltage Performance in Li Ion Batteries, J. Electrochem. Soc., 2013, 160(6), A832 CrossRef CAS.
- A. Shchukarev and D. Korolkov, XPS Study of group IA carbonates, Open Chem., 2004, 2(2), 347–362 CrossRef CAS.
- J. E. Zuliani, S. Tong, D. W. Kirk and C. Q. Jia, Isolating the effect of pore size distribution on electrochemical double-layer capacitance using activated fluid coke, J. Power Sources, 2015, 300, 190–198 CrossRef CAS.
- G. Sun, W. Song, X. Liu, D. Long, W. Qiao and L. Ling, Capacitive matching of pore size and ion size in the negative and positive electrodes for supercapacitors, Electrochim. Acta, 2011, 56(25), 9248–9256 CrossRef CAS.
- L. Zhao, Y. Li, M. Yu, Y. Peng and F. Ran, Electrolyte-wettability issues and challenges of electrode materials in electrochemical energy storage, energy conversion, and beyond, Adv. Sci., 2023, 10(17), 2300283 CrossRef CAS.
- M. Razavifar, A. Abdi, E. Nikooee, O. Aghili and M. Riazi, Quantifying the impact of surface roughness on contact angle dynamics under varying conditions, Sci. Rep., 2025, 15(1), 16611 CrossRef CAS.
- F. Torrisi, T. Hasan, W. Wu, Z. Sun, A. Lombardo, T. S. Kulmala, G.-W. Hsieh, S. Jung, F. Bonaccorso and P. J. Paul, Inkjet-printed graphene electronics, ACS Nano, 2012, 6(4), 2992–3006 CrossRef CAS.
- V. Craig, B. Ninham and R. M. Pashley, Effect of electrolytes on bubble coalescence, Nature, 1993, 364(6435), 317–319 CrossRef CAS.
- L. Meagher and V. S. Craig, Effect of dissolved gas and salt on the hydrophobic force between polypropylene surfaces, Langmuir, 1994, 10(8), 2736–2742 CrossRef CAS.
- C. L. Henry and V. S. Craig, The link between ion specific bubble coalescence and Hofmeister effects is the partitioning of ions within the interface, Langmuir, 2010, 26(9), 6478–6483 CrossRef CAS PubMed.
- Y. Lee, S. B. Park, K.-W. Kim, H. Jo, J. K. Kim, S. H. Kim, S. Lim, S. W. Lee and C.-H. Choi, Effective and Scalable Graphene Ink Production for Printed Microsupercapacitors, Ind. Eng. Chem. Res., 2025, 64(14), 7507–7515 CrossRef CAS.
- L. W. T. Ng, X. Zhu, G. Hu, N. Macadam, D. Um, T.-C. Wu, F. Le Moal, C. Jones and T. Hasan, Conformal Printing of Graphene for Single- and Multilayered Devices onto Arbitrarily Shaped 3D Surfaces, Adv. Funct. Mater., 2019, 29(36), 1807933 CrossRef.
- D. Wang, W. Zhang, J. Wang, X. Li and Y. Liu, A high-performance, all-solid-state Na+ selective sensor printed with eco-friendly conductive ink, RSC Adv., 2023, 13(24), 16610–16618 RSC.
- M. Wang, J. Wang, A. Wei, X. Li, W. Zhang and Y. Liu, Highly stable aqueous carbon-based conductive ink for screen-printed planar flexible micro-supercapacitor, J. Alloys Compd., 2024, 976, 173125 CrossRef CAS.
- A. J. Bard, L. R. Faulkner and H. S. White, Electrochemical methods: fundamentals and applications, John Wiley & Sons, 2022 Search PubMed.
- H. Ji, X. Zhao, Z. Qiao, J. Jung, Y. Zhu, Y. Lu, L. L. Zhang, A. H. MacDonald and R. S. Ruoff, Capacitance of carbon-based electrical double-layer capacitors, Nat. Commun., 2014, 5(1), 3317 CrossRef PubMed.
- J. Meyer, S. Hamwi, M. Kröger, W. Kowalsky, T. Riedl and A. Kahn, Transition metal oxides for organic electronics: energetics, device physics and applications, Adv. Mater., 2012, 24(40), 5408–5427 CrossRef CAS PubMed.
- G. Salitra, A. Soffer, L. Eliad, Y. Cohen and D. Aurbach, Carbon electrodes for double-layer capacitors I. Relations between ion and pore dimensions, J. Electrochem. Soc., 2000, 147(7), 2486 CrossRef CAS.
- R. G. Compton and C. E. Banks, Understanding voltammetry, World Scientific, 2018 Search PubMed.
- R. Agarwal, The Nicholson Method of Determination of the Standard Rate Constant of a Quasireversible Redox Couple Employing Cyclic Voltammetry: Everything One Needs to Know!, ACS Electrochem., 2025, 1(10), 1885–1894 CrossRef CAS.
- I. Lavagnini, R. Antiochia and F. Magno, An extended method for the practical evaluation of the standard rate constant from cyclic voltammetric data, Electroanalysis, 2004, 16(6), 505–506 CrossRef CAS.
- S. Konopka and B. McDuffie, Diffusion coefficients of ferri-and ferrocyanide ions in aqueous media, using twin-electrode thin-layer electrochemistry, Anal. Chem., 1970, 42(14), 1741–1746 CrossRef CAS.
- D. H. Angell and T. Dickinson, The kinetics of the ferrous/ferric and ferro/ferricyanide reactions at platinum and gold electrodes: Part I. Kinetics at bare-metal surfaces, J. Electroanal. Chem. Interfacial Electrochem., 1972, 35(1), 55–72 CrossRef CAS.
- J. Hui, X. Zhou, R. Bhargava, A. Chinderle, J. Zhang and J. Rodríguez-López, Kinetic Modulation of Outer-Sphere Electron Transfer Reactions on Graphene Electrode with a Sub-surface Metal Substrate, Electrochim. Acta, 2016, 211, 1016–1023 CrossRef CAS.
- M. Velický, D. F. Bradley, A. J. Cooper, E. W. Hill, I. A. Kinloch, A. Mishchenko, K. S. Novoselov, H. V. Patten, P. S. Toth, A. T. Valota, S. D. Worrall and R. A. W. Dryfe, Electron Transfer Kinetics on Mono- and Multilayer Graphene, ACS Nano, 2014, 8(10), 10089–10100 CrossRef.
- D. A. Brownson, L. J. Munro, D. K. Kampouris and C. E. Banks, Electrochemistry of graphene: not such a beneficial electrode material?, RSC Adv., 2011, 1(6), 978–988 RSC.
- D. Li, C. Lin, C. Batchelor-McAuley, L. Chen and R. G. Compton, Tafel analysis in practice, J. Electroanal. Chem., 2018, 826, 117–124 CrossRef CAS.
| This journal is © the Owner Societies 2026 |