Decatungstate-photocatalyzed direct acylation of N-heterocycles with aldehydes†
Zhiyang
Zhang‡
a,
Fukun
Cheng‡
a,
Xinyu
Ma
a,
Kai
Sun
 *a,
Xianqiang
Huang
*a,
Xianqiang
Huang
 b,
Jiangzhen
An
ac,
Mei
Peng
d,
Xiaolan
Chen
b,
Jiangzhen
An
ac,
Mei
Peng
d,
Xiaolan
Chen
 a and
Bing
Yu
a and
Bing
Yu
 *a
*a
aCollege of Chemistry, Zhengzhou University, Zhengzhou 450001, China. E-mail: sunkaichem@zzu.edu.cn; bingyu@zzu.edu.cn
bSchool of Chemistry & Chemical Engineering, Liaocheng University, Liaocheng, Shandong 252059, China
cNational Engineering Research Center of Low-Carbon Processing and Utilization of Forest Biomass, Nanjing Forestry University, Nanjing 210037, China
dSchool of Pharmacy, Hengyang Medical School, University of South China, Hengyang, Hunan 421001, China
First published on 30th May 2024
Abstract
A novel photocatalytic acylation strategy was developed harnessing tetrabutylammonium decatungstate (TBADT) as a hydrogen atom transfer (HAT) photocatalyst to facilitate the direct coupling of aldehydes with N-heterocycles at ambient temperature. This approach realizes the direct acylation of N-hetrerocycles without the need for noble metals, acids, or external oxidants, thereby marking a significant advance in green chemistry protocols. Utilizing this photocatalyzed method, we successfully synthesized a diverse array of 3-acylated azauracils and quinoxalin-2(1H)-ones, adorned with a variety of functional groups, achieving yields ranging from good to excellent. This research opens avenues for the rapid and efficient modification of heterocyclic compounds, which are foundational structures in medicinal chemistry and materials science.
Introduction
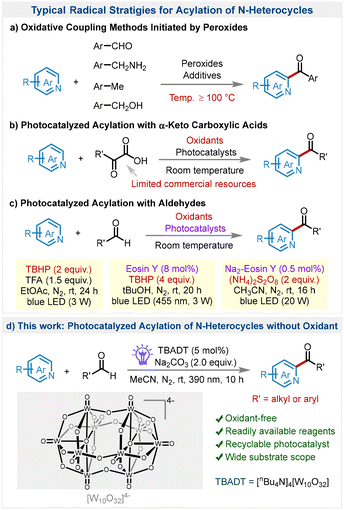
The ubiquitous presence of nitrogen-containing heterocycles within bioactive entities underscores their indispensable contributions to the fields of pharmaceutical development and agrochemical invention.1 These heterocyclic frameworks are the cornerstone of myriad therapeutic agents and agrochemical products, necessitating the advancement of selective and efficient methodologies for their functionalization.2 Acylation, in particular, is essential for altering the chemical and pharmacokinetic properties of heterocycles and has been a fundamental transformation in organic synthesis.3 Historically, the Friedel–Crafts acylation has been a cornerstone transformation within organic chemistry, enabling the incorporation of acyl groups into aromatic and heteroaromatic systems.3c,4 However, Friedel–Crafts acylation generally requires electron-rich substrates. In this context, the Minisci reaction serves as a valuable complement, enabling the radical substitution of electron-deficient aromatic heterocycles.5To date, numerous Minisci-type reactions, which involve the substitution of basic heterocycles by carbon-centered radicals, have been developed.6 Particularly, for the acylation reaction of heterocycles, the oxidative coupling methods are commonly employed but typically require the use of peroxides to initiate the reaction at elevated temperatures (≥100 °C) (Scheme 1a).7 These strategies leverage the radical initiation capability of peroxides to establish C–C bonds. However, a key challenge in the field of green chemistry is the minimization of stoichiometric oxidant usage and the avoidance of severe reaction conditions.
In the past decade, photocatalysis has emerged as a pivotal force,8 transforming the landscape with its unique capability to capture and utilize photon energy as an abundant and clean energy source.9 The application of photocatalysis embodies the core principles of green chemistry and has substantially advanced the direct C–H acylation of N-heterocycles. For example, α-keto carboxylic acids have been widely applied as efficient acyl radical sources for the photocatalytic acylation of N-heterocycles (Scheme 1b).10 However, the range of commercially available α-keto carboxylic acids remains somewhat limited, and their synthesis frequently entails laborious and intricate procedures. On the contrary, aldehydes have historically been recognized as a fundamental component in the history of synthetic chemistry due to their widespread presence.11 They are particularly beneficial in the generation of acyl radicals, offering direct and effective routes that align with green chemistry's objectives to reduce waste and decrease the number of steps in synthesis (Scheme 1c).12 Nevertheless, these methodologies still generally demand the use of additional stoichiometric oxidants.13 Therefore, the development of new catalytic systems with reduced reliance on oxidants is of significance.
Based on our ongoing interest in photocatalytic organic synthesis, we herein present a photoinduced acylation of N-heterocycles with commercially available aldehydes as acylating agents (Scheme 1d). To the best of our knowledge, this is the first successful demonstration of the photoinduced acylation of N-heterocycles with aldehydes without the use of harsh oxidizing conditions. The method's versatility is significantly improved by its ability to be used with a diverse array of aldehydes, including both alkyl and aromatic aldehydes. Furthermore, the unique physicochemical properties of TBADT confer upon the catalyst an outstanding ability for repeated use. This durable reusability not only enhances economic efficiency but also echoes the principles of environmental sustainability by minimizing the generation of chemical waste.
Results and discussion
Azauracil, a derivative of azapyrimidinone with a structural analogy to uracil, is increasingly recognized for its antimicrobial inhibitory potential.14 The scientific discourse surrounding this compound has intensified, as evidenced by the heightened investigative efforts from both chemists and pharmacologists. Advancements in the synthesis of azauracil derivatives, which aim to diversify their structural attributes, are anticipated to significantly accelerate progress in this domain, thus enriching the arsenal of microbiological treatments.15 In the context of these scientific advancements, our research initiated an exploratory synthesis involving the reaction of 2,4-dibenzyl-1,2,4-triazine-3,5(2H,4H)-dione (1a) with 3-phenylpropanal (2a), delineated in Table 1. After extensive optimization, it was observed that employing TBADT as a photocatalyst, with 2 equivalents of Na2CO3 as a base, and MeCN as the solvent under 390 nm wavelength light irradiation, the desired azauracil derivative 3a was isolated in 74% yield (entry 1). A solvent screen revealed that the reaction did not proceed effectively when MeCN was substituted with water, DMSO, or dimethyl carbonate (DMC) (entries 2–4). Alternative solvents such as acetone or dichloromethane (DCM) also led to diminished yields in comparison with MeCN (entries 5 and 6). Further base optimization trials demonstrated that 2 equivalents of Na2CO3 were integral for the successful progression of the reaction (entries 7–10). In the absence of base, light irradiation, or TBADT, the reaction was significantly hindered, underscoring the important role of these reaction components (entries 11–13). Moreover, when the reaction assembly was exposed to an open-air atmosphere, a significant inhibition of the reaction was observed, suggesting that atmospheric oxygen may act detrimentally upon the reaction's efficiency (entry 14). Finally, when the reaction was conducted with Eosin Y as the photocatalyst under white light irradiation—a catalyst heralded for its adeptness in hydrogen atom transfer (HAT) catalysis16—the anticipated transformation was impeded, failing to yield the product (entry 15). This pronounced disparity in reactivity highlights the superior catalytic performance of our TBADT-based photocatalytic system, showcasing the unique capabilities of our strategy.| Entry | Variation from the conditions | Yield (%) |
|---|---|---|
| a Reaction conditions: 1a (0.2 mmol), 2a (0.6 mmol), MeCN (2.0 mL) at room temperature for 10 h under the irradiation of purple LED (390 nm, 10 W) under N2 atmosphere. Isolated yields were given based on 1a. N.D. = not detected. | ||
| 1 | None | 74 |
| 2 | H2O instead of MeCN | N.D. |
| 3 | DMSO instead of MeCN | N.D. |
| 4 | DMC instead of MeCN | N.D. |
| 5 | Acetone instead of MeCN | 60 |
| 6 | DCM instead of MeCN | 30 |
| 7 | NaHCO3 instead of Na2CO3 | 50 |
| 8 | DABCO instead of Na2CO3 | 35 |
| 9 | Et3N instead of Na2CO3 | N.D. |
| 10 | 1.0 equiv. of Na2CO3 | 45 |
| 11 | No base | 30 |
| 12 | In the dark | N.D. |
| 13 | Without TBADT | N.D. |
| 14 | Under open-air atmosphere | N.D. |
| 15 | Eosin Y instead of TBADT with white light | N.D. |
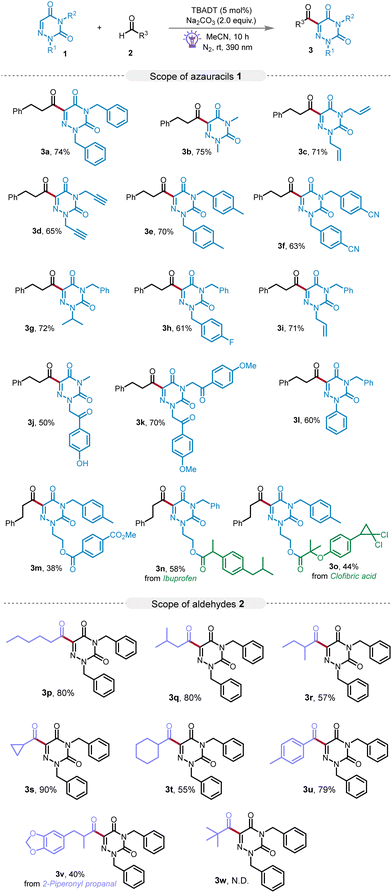
After obtaining the optimized reaction conditions, we embarked on an exhaustive examination to ascertain the system's versatility across a diverse range of substrates. Utilizing 3-phenylpropanal (2a) as our benchmark aldehyde, we engaged in reactions with a variety of 1,2,4-triazine-3,5(2H,4H)-diones as illustrated in Scheme 2. Our experimental results were encouraging, revealing that 1,2,4-triazine-3,5(2H,4H)-diones, featuring a range of substituents, encompassing alkyl, aryl, benzyl, allyl, propargyl, and acetophenone groups, were all amenable to the photocatalyzed alkylation process. This process culminated in a suite of acylation products, identified as 3a–3m, being synthesized with consistently satisfactory yields ranging from 38% to 74%. Significantly, the derivatives of 1,2,4-triazine-3,5(2H,4H)-diones, which incorporated structural motifs of pharmaceutical relevance, such as those found in Ibuprofen or Clofibric acid, also underwent smooth reactions with 3-phenylpropanal giving the expected compounds 3n and 3o, with respectable yields of 58% and 44%, respectively. These results highlight the robustness and flexibility of our photocatalytic system. Further broadening our investigation, we assessed the reactivity profile of various aldehydes 2 with compound 1a. Primary alkyl aldehydes, such as hexanal and isovaleraldehyde, were identified as compatible substrates, furnishing the target products 3p and 3q with high efficiency. Secondary alkyl aldehydes, including 2-methylbutanal, cyclobutanecarbaldehyde, and cyclohexanecarbaldehyde, were also well accommodated, generating products 3r–3t with yields ranging from 55% to 90%. Furthermore, the protocol proved amenable to aryl aldehydes, as demonstrated by the reaction with 4-methyl benzaldehyde, which resulted in the formation of product 3u with a 79% yield. Intriguingly, when 2-piperonyl propanal, a natural product aldehyde, was introduced to the reaction with 1a, the resulting product 3v was achieved with a moderate yield (40%). These findings provide evidence for the effectiveness of our approach, which can facilitate direct acylation reactions with a wide variety of aldehyde substrates, ranging from simple to complex in structure. Regrettably, the use of trimethylacetaldehyde, a tertiary alkyl aldehyde, as a substrate under our optimized reaction conditions did not result in the detection of the anticipated product 3w. This lack of formation could be attributed to the intrinsic instability of tertiary aldehyde radicals that are susceptible to decarbonylation, leading to the production of tertiary carbon radicals.
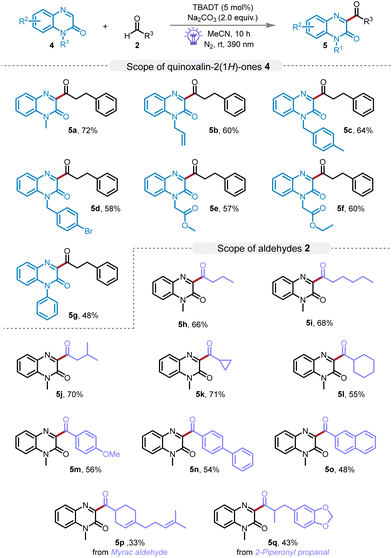
To further showcase the breadth of our novel catalytic system, we applied our catalytic system to the acylation of quinoxalin-2(1H)-ones, as portrayed in Scheme 3. These heteroaromatic compounds garner significant interest due to their pivotal roles in the discovery of antimicrobial, antitumor agents, and materials with semiconducting properties.17 The reactions proceeded with remarkable site specificity and reactivity, yielding the corresponding acylation products, denoted 5a–5g with 48–72% yields. This adaptable acylation technique highlights the wide applicability of our method in synthesizing diverse quinoxalin-2(1H)-one derivatives. Particularly, the mild reaction conditions of our system exemplify its ability to meticulously preserve sensitive functional groups, such as alkenyl groups (5b). Furthermore, we investigated the reactivity of various aldehydes with quinoxalinones. Analogously, both primary and secondary aliphatic aldehydes, along with aromatic aldehydes, were amenable to the acylation reaction, producing the desired products 5h–5o in 48%–71% yields. Notably, the natural essence Myrac aldehyde and 2-piperonyl propanal were also successfully transformed, yielding the expected products 5p and 5q with yields of 33% and 43%, respectively. Unfortunately, when employing quinoline, isoquinoline, or pyridine as substrate under the optimized reaction conditions, we failed to detect the formation of the desired product (for details see the ESI†). This absence of reactivity can plausibly be attributed to the requisite acid activation in the typical Minisci reaction, which is not provided under the basic conditions of our methodology. Consequently, our approach may not be appropriately extendable to substrates such as quinoline, isoquinoline, or pyridine.
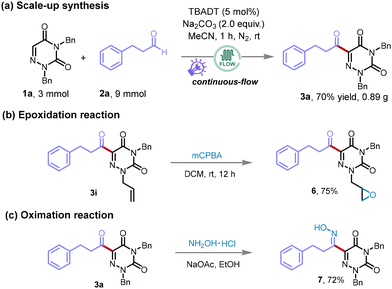
Following the completion of initial exploratory experiments, we sought to extend the model reaction to a larger scale, employing 7.5 mmol of the starting materials and adopting a continuous-flow methodology to assess the scalability and practical feasibility of our synthetic approach (Scheme 4a). It was indeed encouraging to observe that the amplification of the reaction scale did not impinge on its efficiency. The reaction proceeded with notable smoothness, culminating in the synthesis of the target compound 3a, with only a slight reduction in yield to 70% (0.89 g). To further substantiate the method's reliability and its applicability in practical settings, we performed additional transformations as illustrated in Scheme 4b and c. Among these, the terminal olefin present in compound 3i was effectively oxidized by m-chloroperbenzoic acid (mCPBA), yielding the oxidized product 6 with an excellent yield of 75% (Scheme 4b). Additionally, product 3a could react with hydroxylamine hydrochloride (NH2OH·HCl) to generate the oxime derivative 7 with a substantial yield of 72% (Scheme 4c). These results not only affirm the high efficiency and scalability of the reaction but also illuminate the method's capacity for facilitating diverse chemical modifications.
Moreover, the TBADT catalyst was successfully recovered through precipitation. Upon completion of the reaction, the introduction of diethyl ether to the reaction mixture led to the full precipitation of the TBADT. Subsequently, the precipitated catalyst was filtered, dried, and prepared for reuse in the next cycle. Notably, the recycled TBADT demonstrated the capacity for at least four cycles of reuse. This reduction in catalytic performance is conjectured to arise from the cumulative loss of catalyst during the recovery process (for details see the ESI†). This aspect of catalyst reusability further underscores the sustainable nature of the process, marking an advancement in the development of environmentally conscious synthetic protocols.
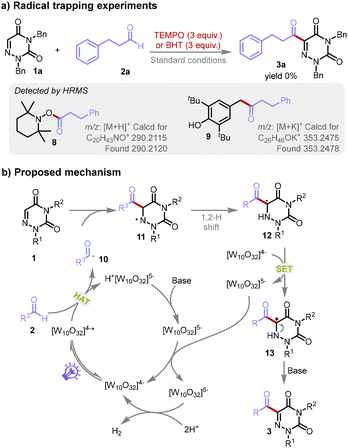
For a deep understanding of the reaction, we executed control experiments by introducing known radical scavengers, 2,2,6,6-tetramethylpiperidin-1-yl-oxidanyl (TEMPO) or 2,6-di-tert-butyl-4-methylphenol (BHT), into the model reaction, while maintaining all other experimental parameters constant. The introduction of these scavengers resulted in the complete inhibition of the target product 3a formation, which indicates the involvement of a radical mechanism in this synthetic process (Scheme 5a). To further validate these findings, high-resolution mass spectrometry (HRMS) was employed to analyze the reaction mixture. The HRMS spectra displayed distinct peaks at m/z = 290.2115 and m/z = 353.2475, which were attributed to the protonated molecule [C20H43NO]+ and the potassium-adduct [C26H46OK]+, respectively. The identification of these entities offers evidence for an acyl radical intermediate participating in the reaction sequence (for details, see the ESI†).
Integrating our experimental findings with relevant reports,18 we propose a plausible reaction mechanism. As depicted in Scheme 5b, the polyoxometalate anion [W10O32]4− is photo-excited to its excited state [W10O32]4−*. This active species instigates a hydrogen atom transfer (HAT) with aldehyde 2, engendering the acyl radical 10 and H+[W10O32]5−. The acyl radical 10 then reacts with substrate 1, resulting in the radical intermediate 11. The intermediate 11 experiences an intramolecular 1,2-hydrogen shift, generating the radical intermediate 12, which in turn participates in a single electron transfer with [W10O32]4−. This process yields [W10O32]5− and the cationic intermediate 13, which, upon deprotonation, furnishes the acylated product 3. Simultaneously, the species H+[W10O32]5− is transformed back to [W10O32]5− in the presence of a base. Thereafter, [W10O32]5− can return to the initial photocatalyst [W10O32]4− along with the formation of [W10O32]6−via disproportionation.19 Ultimately, the catalytic action of [W10O32]6− facilitates the production of H2 from protons, thereby regenerating the photocatalyst [W10O32]4−. The production of H2 was distinctly evidenced by a characteristic peak in the gas chromatography analysis, identified by a thermal conductivity detector (TCD) detector, lending strong support to our proposed reaction mechanism (for details see the ESI†).
Conclusions
In conclusion, we have successfully established a photocatalytic acylation protocol for the direct coupling of alkyl and aromatic aldehydes with N-heterocycles at room temperature employing tetrabutylammonium decatungstate (TBADT) as a photocatalyst. This newly established protocol sidesteps the traditional reliance on noble metals, acidic environments, and oxidative reagents, aligning with the objectives of sustainable and eco-friendly chemical practices. The efficacy and adaptability of this photocatalytic system have been demonstrated through the successful synthesis of an array of 3-acylated azauracils and quinoxalin-2(1H)-ones, adorned with a plethora of functional groups, and consistently delivering good yields. Furthermore, the specialized physicochemical properties of TBADT impart to the catalyst an extraordinary capacity for multiple cycles of use. This quality not only fosters economic advantage but also aligns with green chemistry's ethos by markedly diminishing the production of chemical waste.Conflicts of interest
There are no conflicts to declare.Acknowledgements
We acknowledge the financial support from the National Natural Science Foundation of China (21971224 and 22171249), the Natural Science Foundation of Henan Province (232300421363), and the Science & Technology Innovation Talents in Universities of Henan Province (23HASTIT003).References
-
(a) Z. Qu, T. Tian, G.-J. Deng and H. Huang, Chin. Chem. Lett., 2023, 34, 107565 CrossRef CAS
; (b) H. H. Nguyen, M. B. Kim, R. J. Wilson, C. J. Butch, K. M. Kuo, E. J. Miller, Y. A. Tahirovic, E. Jecs, V. M. Truax, T. Wang, C. S. Sum, M. E. Cvijic, G. M. Schroeder, L. J. Wilson and D. C. Liotta, J. Med. Chem., 2018, 61, 7168 CrossRef CAS PubMed
; (c) M. Sharique, J. Majhi, R. K. Dhungana, L. M. Kammer, M. Krumb, A. Lipp, E. Romero and G. A. Molander, Chem. Sci., 2022, 13, 5701 RSC
.
-
(a) L. Wei, C. Shen, Y.-Z. Hu, H.-Y. Tao and C.-J. Wang, Chem. Commun., 2019, 55, 6672 RSC
; (b) Y. Liu, G. Zeng, Y. Cheng, L. Chen, Y. Liu, Y. Wei and G. Yang, Chin. Chem. Lett., 2024, 35, 108480 CrossRef CAS
; (c) H.-Y. Song, F. Xiao, J. Jiang, C. Wu, H.-T. Ji, Y.-H. Lu, K.-L. Wang and W.-M. He, Chin. Chem. Lett., 2023, 34, 108509 CrossRef CAS
.
-
(a) X.-F. Wu, Chem. – Eur. J., 2015, 21, 12252 CrossRef CAS PubMed
; (b) H. Y. Hou, Y. Y. Cheng, B. Chen, C. H. Tung and L. Z. Wu, Chin. J. Org. Chem., 2023, 43, 1012 CrossRef CAS
; (c) C. Raviola, S. Protti, D. Ravelli and M. Fagnoni, Green Chem., 2019, 21, 748 RSC
; (d) K. Xie, M. Jiang, X. Chen, Q. Lv and B. Yu, Chin. J. Org. Chem., 2021, 41, 4575 CrossRef CAS
; (e) G. S. Lee and S. H. Hong, Acc. Chem. Res., 2023, 56, 2170 CrossRef CAS PubMed
.
- M. Bandini, J. Am. Chem. Soc., 2010, 132, 7821 CrossRef CAS
.
-
(a) L. Désaubry and J.-J. Bourguignon, Tetrahedron Lett., 1995, 36, 7875 CrossRef
; (b) Q.-Q. Wang, K. Xu, Y.-Y. Jiang, Y.-G. Liu, B.-G. Sun and C.-C. Zeng, Org. Lett., 2017, 19, 5517 CrossRef CAS PubMed
; (c) F. Fontana, F. Minisci, M. C. Nogueira Barbosa and E. Vismara, J. Org. Chem., 1991, 56, 2866 CrossRef CAS
.
-
(a) R. S. J. Proctor and R. J. Phipps, Angew. Chem., Int. Ed., 2019, 58, 13666 CrossRef CAS PubMed
; (b) B. Bieszczad, L. A. Perego and P. Melchiorre, Angew. Chem., Int. Ed., 2019, 58, 16878 CrossRef CAS PubMed
; (c) Z. Wang, X. Ji, J. Zhao and H. Huang, Green Chem., 2019, 21, 5512 RSC
; (d) Z. Wang, Q. Liu, X. Ji, G.-J. Deng and H. Huang, ACS Catal., 2020, 10, 154 CrossRef CAS
; (e) J. Dong, Z. Wang, X. Wang, H. Song, Y. Liu and Q. Wang, Sci. Adv., 2019, 5, eaax9955 CrossRef CAS PubMed
.
-
(a) J. Chen, M. Wan, J. Hua, Y. Sun, Z. Lv, W. Li and L. Liu, Org. Biomol. Chem., 2015, 13, 11561 RSC
; (b) Y. Siddaraju, M. Lamani and K. R. Prabhu, J. Org. Chem., 2014, 79, 3856 CrossRef CAS PubMed
; (c) W. Ali, A. Behera, S. Guin and B. K. Patel, J. Org. Chem., 2015, 80, 5625 CrossRef CAS PubMed
; (d) R. Sharma, M. Abdullaha and S. B. Bharate, J. Org. Chem., 2017, 82, 9786 CrossRef CAS PubMed
.
-
(a) X. Li, W. Cui, Q. Deng, X. Song, J. Lv and D. Yang, Green Chem., 2022, 24, 1302 RSC
; (b) F. Yue, H. Ma, P. Ding, H. Song, Y. Liu and Q. Wang, ACS Cent. Sci., 2023, 9, 2268 CrossRef CAS PubMed
; (c) J. Qi, F. L. Zhang, J. K. Jin, Q. Zhao, B. Li, L. X. Liu and Y. F. Wang, Angew. Chem., Int. Ed., 2020, 59, 12876 CrossRef CAS PubMed
; (d) H. Bao, L. Zheng, Q. Liu, M. Han, Y. Li, M. Bao, Y. Li, P. Yan and Y. Liu, Org. Chem. Front., 2023, 10, 5551 RSC
; (e) D. Xu, F. Huang, L. Tang, X. Zhang and W. Zhang, Chin. J. Org. Chem., 2022, 42, 1493 CrossRef CAS
.
-
(a) C. Ma, X. Li, X. Chen, X. He, S.-T. Zhang, Y.-Q. Jiang and B. Yu, Org. Lett., 2023, 25, 8016 CrossRef CAS PubMed
; (b) L. Wang, M. Zhang, Y. Zhang, Q. Liu, X. Zhao, J.-S. Li, Z. Luo and W. Wei, Chin. Chem. Lett., 2020, 31, 67 CrossRef CAS
; (c) Y. Lv, P. Bao, H. Yue, J.-S. Li and W. Wei, Green Chem., 2019, 21, 6051 RSC
; (d) G. Li, Q. Yan, X. Gong, X. Dou and D. Yang, ACS Sustainable Chem. Eng., 2019, 7, 14009 CrossRef CAS
; (e) L. Wang, H. Zhang, C. Zhu and C. Feng, Chin. J. Chem., 2022, 40, 59 CrossRef CAS
; (f) Z. Qu, T. Tian, Y. Tan, X. Ji, G.-J. Deng and H. Huang, Green Chem., 2022, 24, 7403 RSC
.
-
(a) X.-Y. Zhang, W.-Z. Weng, H. Liang, H. Yang and B. Zhang, Org. Lett., 2018, 20, 4686 CrossRef CAS PubMed
; (b) H.-L. Zhu, F.-L. Zeng, X.-L. Chen, K. Sun, H.-C. Li, X.-Y. Yuan, L.-B. Qu and B. Yu, Org. Lett., 2021, 23, 2976 CrossRef CAS PubMed
; (c) L. Guillemard, F. Colobert and J. Wencel-Delord, Adv. Synth. Catal., 2018, 360, 4184 CrossRef CAS
; (d) L.-N. Guo, H. Wang and X.-H. Duan, Org. Biomol. Chem., 2016, 14, 7380 RSC
.
-
(a) A. Banerjee, Z. Lei and M.-Y. Ngai, Synthesis, 2019, 303 CAS
; (b) Y. Wang, X.-F. Liu and W.-M. He, Org. Chem. Front., 2023, 10, 4198 RSC
; (c) J. Xue, Y.-S. Zhang, Z. Huan, J.-D. Yang and J.-P. Cheng, J. Org. Chem., 2022, 87, 15539 CrossRef CAS PubMed
; (d) C. Ma, H. Meng, J. Li, X. Yang, Y. Jiang and B. Yu, Chin. J. Chem., 2022, 40, 2655 CrossRef CAS
; (e) C.-H. Ma, Y. Ji, J. Zhao, X. He, S.-T. Zhang, Y.-Q. Jiang and B. Yu, Chin. J. Catal., 2022, 43, 571 CrossRef CAS
.
-
(a) Y.-L. Liu, Y.-J. Ouyang, H. Zheng, H. Liu and W.-T. Wei, Chem. Commun., 2021, 57, 6111 RSC
; (b) W. Lee, S. Jung, M. Kim and S. Hong, J. Am. Chem. Soc., 2021, 143, 3003 CrossRef CAS PubMed
.
-
(a) L. Zhang, G. Zhang, Y. Li, S. Wang and A. Lei, Chem. Commun., 2018, 54, 5744 RSC
; (b) X. Wang, X. Shao, Z. Cao, X. Wu and C. Zhu, Adv. Synth. Catal., 2022, 364, 1200 CrossRef CAS
; (c) V. J. Roy, P. P. Sen and S. R. Roy, Chem. Commun., 2022, 58, 1776 RSC
; (d) H. Ni, Y. Li, X. Shi, Y. Pang, C. Jin and F. Zhao, Tetrahedron Lett., 2021, 68, 152915 CrossRef CAS
; (e) H. Ni, Y. Li, J. Deng, X. Shi and Q. Pan, New J. Chem., 2021, 45, 22432 RSC
.
-
(a) K. Sun, A. Shi, Y. Liu, X. Chen, P. Xiang, X. Wang, L. Qu and B. Yu, Chem. Sci., 2022, 13, 5659 RSC
; (b) P. Ghosh, N. Y. Kwon, S. Kim, S. Han, S. H. Lee, W. An, N. K. Mishra, S. B. Han and I. S. Kim, Angew. Chem., Int. Ed., 2021, 60, 191 CrossRef CAS PubMed
.
- Y. Tan, W. Xuekun, Y.-P. Han, Y. Zhang, H.-Y. Zhang and J. Zhao, J. Org. Chem., 2022, 87, 8551 CrossRef CAS PubMed
.
-
(a) D.-M. Yan, J.-R. Chen and W.-J. Xiao, Angew. Chem., Int. Ed., 2019, 58, 378 CrossRef CAS PubMed
; (b) X.-Z. Fan, J.-W. Rong, H.-L. Wu, Q. Zhou, H.-P. Deng, J. D. Tan, C.-W. Xue, L.-Z. Wu, H.-R. Tao and J. Wu, Angew. Chem., Int. Ed., 2018, 57, 8514 CrossRef CAS PubMed
.
-
(a) S.-J. Wu, Y. Shi, K. Sun, X.-Y. Yuan, S. Tang and B. Yu, J. Catal., 2022, 415, 87 CrossRef CAS
; (b) Q. Yan, W. Cui, J. Li, G. Xu, X. Song, J. Lv and D. Yang, Org. Chem. Front., 2022, 9, 2653 RSC
; (c) X. Shi, Y. Cao, Y. Liu, K. Niu, H. Song, J. Zhang and Q. Wang, Org. Chem. Front., 2023, 10, 1296 RSC
; (d) H.-F. Liu, M.-X. He and H.-T. Tang, Org. Chem. Front., 2022, 9, 5955 RSC
; (e) C. Liang, Y. Guo, Y. Zhang, Z. Wang, L. Li and W. Li, Org. Chem. Front., 2023, 10, 611 RSC
.
-
(a) X.-Y. Yuan, Y.-F. Si, X. Li, S.-J. Wu, F.-L. Zeng, Q.-Y. Lv and B. Yu, Org. Chem. Front., 2022, 9, 2728 RSC
; (b) X. Yuan, G. Yang and B. Yu, Chin. J. Org. Chem., 2020, 40, 3620 CrossRef CAS
; (c) J. Xu, L. Liu, Z.-C. Yan, Y. Liu, L. Qin, N. Deng and H.-J. Xu, Green Chem., 2023, 25, 2268 RSC
; (d) S. A. Paveliev, O. O. Segida, O. M. Mulina, I. B. Krylov and A. O. Terent'ev, Org. Lett., 2022, 24, 8942 CrossRef CAS PubMed
; (e) W. Liu, C. Liu, M. Wang and W. Kong, ACS Catal., 2022, 12, 10207 CrossRef CAS
; (f) X. Wang, J. Dong, Y. Liu, H. Song and Q. Wang, Chin. Chem. Lett., 2021, 32, 3027 CrossRef CAS
; (g) Z.-Y. Dai, Z.-S. Nong, S. Song and P.-S. Wang, Org. Lett., 2021, 23, 3157 CrossRef CAS PubMed
; (h) L. Capaldo, D. Ravelli and M. Fagnoni, Chem. Rev., 2022, 122, 1875 CrossRef CAS PubMed
.
- R. F. Renneke, M. Kadkhodayan, M. Pasquali and C. L. Hill, J. Am. Chem. Soc., 1991, 113, 8357 CrossRef CAS
.
Footnotes |
| † Electronic supplementary information (ESI) available. See DOI: https://doi.org/10.1039/d4gc00534a |
| ‡ These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2024 |