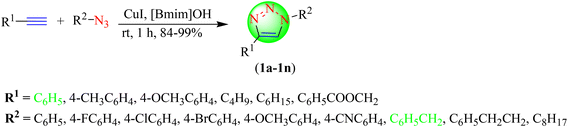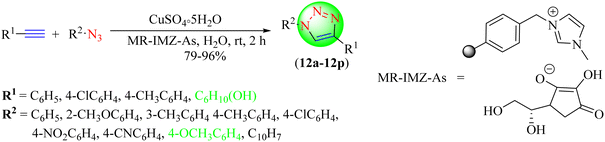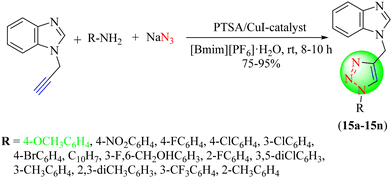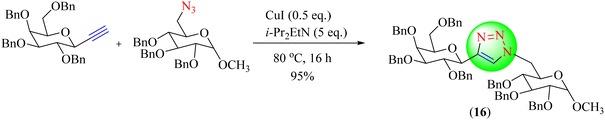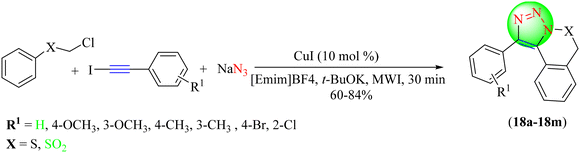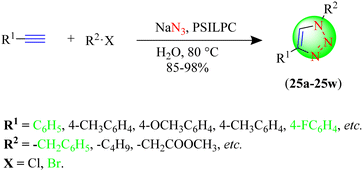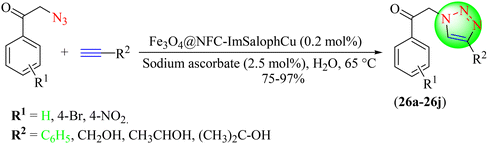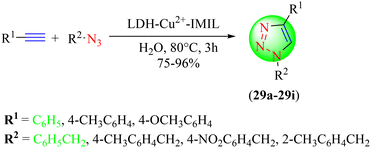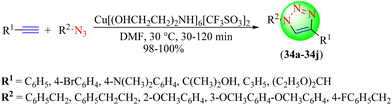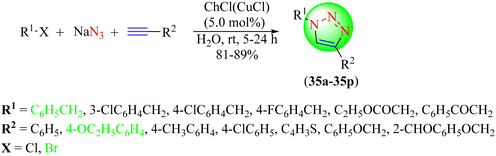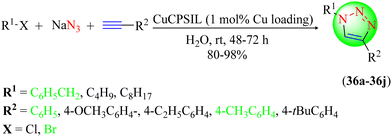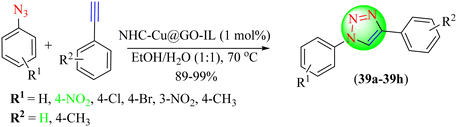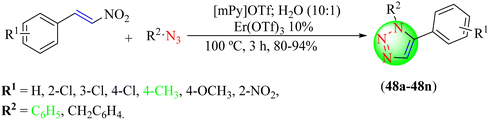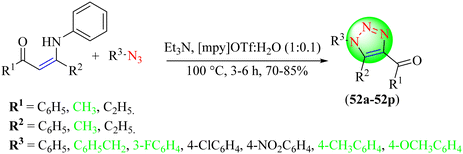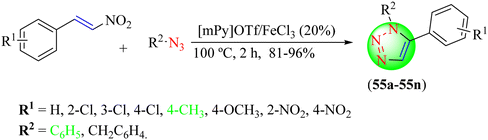Ionic liquids for the green synthesis of 1,2,3-triazoles: a systematic review
Aman
Kumar
ad,
Vijay
Kumar
a,
Prashant
Singh
b,
Ram Kumar
Tittal
 *c and
Kashmiri
Lal
*c and
Kashmiri
Lal
 *a
*a
aDepartment of Chemistry, Guru Jambheshwar University of Science & Tech., Hisar, Haryana 125001, India. E-mail: klal_iitd@yahoo.com
bDepartment of Chemistry, Atma Ram Sanatan Dharma College, University of Delhi, Dhaula Kuan, New Delhi 110021, India
cDepartment of Chemistry, National Institute of Technology, Kurukshetra, Haryana 136119, India. E-mail: rktittaliitd@nitkkr.ac.in
dDepartment of Chemistry, Manipal University Jaipur, Dehmi kalan, Jaipur 303007, Rajasthan, India
First published on 21st February 2024
Abstract
The exponentially increasing use of hazardous traditional organic solvents and catalysts for more accessible synthesis of important heterocycles useful for humankind in various fields, such as medicine, materials science, and agrochemicals, is having devastating impacts on the environment, are directly or indirectly affecting the structure and productivity of ecosystems. This has shifted the focus of the scientific community towards using eco-friendly and greener solvents, catalysts, materials, and methods for sustainable growth. As a result, there is a drive to change traditional techniques to environmentally sustainable processes by following the principle's of green chemistry. In this connection, the use of hazardous organic solvents and catalysts for synthesizing 1,2,3-triazoles is a global concern. Thus, newer methodologies were designed using eco-friendly catalysts and benign solvents such as ionic liquids (ILs) or water. The use of ILs in organic synthesis as a solvent and catalyst has attracted tremendous attention in recent years owing to their superior physiochemical properties, such as low vapor pressure, non-volatility, non-flammability, excellent conductivity, and electrochemical and thermal stability, thereby, increasing the reactivity, selectivity, catalyst recyclability, and other properties. In this review, authors have critically screened, divided, and summarized research articles into two significant sections: firstly, the synthesis of 1,2,3-triazoles from alkynes using ILs and secondly, the synthesis of 1,2,3-triazoles from substrate molecules other than alkynes using ILs. It is hoped that this review will stimulate scientists to adopt environmentally sustainable ILs as green solvents and catalysts for the synthesis of 1,2,3-triazole-based compounds.
1. Introduction
Heterocyclic compounds are among the most important classes of organic compounds as these are an integral part of various biologically significant molecules, including deoxyribonucleic acid (DNA), ribonucleic acid (RNA), hemoglobin, vitamins, and steroids.1–4 Further, heterocyclic compounds play a significant role in the synthesis of several drugs useful for the treatment of various diseases in humans, animals, and plants.5,6 However, protocols followed for their synthesis are of global concern as they utilize hazardous chemicals, solvents, and catalysts. For the less hazardous synthesis of these significant heterocycles, the scientific community has been working hard in recent years to create new methodologies that are less harmful to the environment and human health.7,8 Researchers have paid much attention to new synthetic “green chemistry” strategies. The concept of green chemistry was first put forth in the 1990s; since then it become one of the most promising fields that works at the molecular level to achieve sustainability.9 This approach reduces the risks to human health and environmental concerns that emerge from synthesizing, processing, and using hazardous chemicals. Many efforts are underway to replace harmful traditional solvents and catalysts with eco-friendly substitutes in observance of two levels of assessment, namely environmental, health, and safety (EHS) and life cycle assessment (LCA). It is obligatory to find green solvents that are non-hazardous, eco-friendly, safe, renewable, prevent waste, non-flammable, easy to handle, reusable, etc. This may also provide easier synthetic access to heterocyclic scaffolds.10–14 In this regard, ILs may be a suitable medium/ catalyst that adheres to green chemistry principles, as illustrated in Fig. 1.15ILs are compounds entirely made up of ions with a melting point of less than 373 K and these have drawn extensive attention in recent years from researchers and industry because of their distinctive properties, including low vapor pressure, low toxicity, outstanding thermal stability, and recyclability and are, therefore, recognized as sustainable liquids or solvents.16,17 Utilizing ILs as alternative reaction media is attracting chemists’ attention as it can circumvent the issues associated with many of the classic volatile organic solvents and catalysts. The term “designer solvent” has been used for ILs as they comprise cationic and anionic components and can be designed according to the reaction conditions.18,19 Besides their uses as a solvent and catalyst, ILs have a lot of potential for use in several fields, including biological, physical chemistry, engineering, analytics, and electrochemistry.20–34
2. Green synthesis of 1,2,3-triazoles
Synthetic protocols utilizing ILs as a solvent/catalyst under microwave irradiation (MI) instead of conventional heating are more useful as both ILs and MI adhere to the principles of green chemistry and provide a way to achieve excellent yields with short reaction times and effortless purification.35–42 In this review article, we summarize various reports available on synthesis of 1,2,3-triazoles utilizing ILs as a greener solvent or potential catalyst under thermal conditions or microwave irradiation. 1,2,3-Triazole is a significant scaffold with promising applications in different fields such as medicinal chemistry,43–55 biochemistry,56 dendrimers,57–62 materials chemistry,63–67 chemical sensing,68–70 ionic recognition,71,72 peptidomimetics,73,74 and bioconjugation.75 1,2,3-Triazoles came into the mainstream of synthesis after the great work by Huisgen in this field in 1960, who provided a more straightforward laboratory synthetic pathway for the synthesis of 1,2,3-triazoles that involves the 1,3-dipolar cycloaddition of terminal alkynes with organic azides under thermal conditions.76 After some time, it was found that Huisgen's protocol for synthesizing 1,2,3-triazoles has some disadvantages, such as requiring high temperature, more time for reaction completion, and it gives products in a low yield. To overcome these demerits of Huisgen's methodology, Meldal77 and Sharpless’78 teams in 2002 independently modified the process into a more straightforward and novel prize-winning form known as Cu(I)-catalyzed azide–alkyne cycloaddition (CuAAC). This CuAAC protocol has many advantages over the traditional method, such as high selectivity, mild reaction conditions, low reaction temperature, high yield, and broad substrate scope. However, the use of volatile organic solvents and catalysts in this methodology is of environmental concern. Consequently, many researchers are working on overcoming this situation and developing more eco-friendly methods for the greener synthesis of 1,2,3-triazoles.79,80Recently, Tittal et al. published a review entitled “Green synthesis of 1,4-disubstituted 1,2,3-triazoles: a sustainable approach”.81 The review article was focused on the synthesis of only 1,4-disubstituted 1,2,3-triazoles by making use of green catalysts, solvents, microwave and ultrasonic irradiation assistance. However, the present review highlights the use of ILs as a solvent and as a catalytic system or both for the synthesis of a diverse range of 1,2,3-triazole derivatives. This review may help to develop new synthetic strategies for ILs based on green catalytic or reaction media adhering to the green chemistry principles for obtaining 1,2,3-triazoles. This review summarizes various reports on the synthesis of 1,2,3-triazoles using ILs, and also highlights the results of the best catalytic system or reaction media. Further, the substituents shown in green colors in all the schemes represent the highest yield of the 1,2,3-triazole. The classification of this work is in two parts, as mentioned below:
(i) Synthesis of 1,2,3-triazoles from terminal alkyne substrates using: (a) ILs as a solvent, (b) ILs as a catalyst system, and (c) ILs as both solvent as well as catalyst;
(ii) Synthesis of 1,2,3-triazoles from substrates other than alkynes using: (a) ILs as a solvent, (b) ILs as a catalyst system, and (c) ILs as both solvent as well as catalyst.
2.1. Synthesis of 1,2,3-triazoles from terminal alkyne substrates using ILs
The synthesis of 1,2,3-triazoles utilizing ILs is presented and it was observed that ILs have been used either as a solvent or as a catalyst system. Further, there are some reports, where ILs have played a role as both a solvent as well as catalyst. Therefore, the literature has been classified further in to three subsections in this paper: Synthesis of 1,2,3-triazoles from terminal alkyne substrates using ILs as a solvent; Synthesis of 1,2,3-triazoles from terminal alkyne substrates using ILs as a catalyst system; and Synthesis of 1,2,3-triazoles from terminal alkyne substrates using ILs as both a solvent as well as a catalyst. These three subsections are discussed below.Javaherian and co-workers synthesized a series of 1,2,3-triazoles (2a–2l) via an eco-friendly CuAAC reaction of various organic azides with phenylacetylene in IL![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) water as a solvent system in the presence of several Cu(I)-catalyst (Scheme 2).83 Analysis with different reaction conditions showed that the best yield (63%–92%) of 1,2,3-triazoles was obtained when IL and water were taken in a 1
water as a solvent system in the presence of several Cu(I)-catalyst (Scheme 2).83 Analysis with different reaction conditions showed that the best yield (63%–92%) of 1,2,3-triazoles was obtained when IL and water were taken in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio, in the presence of CuSO4·5H2O and sodium ascorbate. The IL used in this reaction was tetra-ethylene-glycol bis-(1-methyl-3-imidazolium)tosylate, and the co-solvent taken was water. This protocol was associated with an advantage that led to the formation of 1,2,3-triazoles within a short period without using toxic solvents. CuSO4 with sodium ascorbate in IL, b gave an excellent yield (95%) in a short reaction time (30 min) compared to the tested CuCl, CuI, and CuSO4-sodium ascorbate systems in a and b.
1 ratio, in the presence of CuSO4·5H2O and sodium ascorbate. The IL used in this reaction was tetra-ethylene-glycol bis-(1-methyl-3-imidazolium)tosylate, and the co-solvent taken was water. This protocol was associated with an advantage that led to the formation of 1,2,3-triazoles within a short period without using toxic solvents. CuSO4 with sodium ascorbate in IL, b gave an excellent yield (95%) in a short reaction time (30 min) compared to the tested CuCl, CuI, and CuSO4-sodium ascorbate systems in a and b.
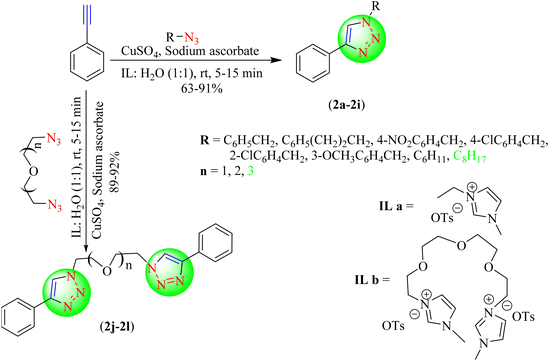
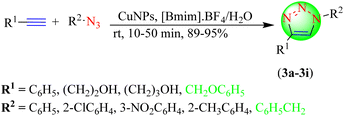
Raut et al. reported a novel and greener approach for obtaining 1,4-disubstituted 1,2,3-triazoles (3a–3i) from terminal alkynes and azides using copper nanoparticles (CuNPs) in an IL![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) water solution in an excellent yield (Scheme 3).84 Initially, the IL-based NPs were obtained from copper acetate by reduction in the presence of hydrazine hydrate in the IL
water solution in an excellent yield (Scheme 3).84 Initially, the IL-based NPs were obtained from copper acetate by reduction in the presence of hydrazine hydrate in the IL![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) water solvent system and were then stabilized by the addition of PVA/PVP. The study summarized that obtaining triazoles from electron-deficient azides required a longer duration. CuNPs catalyst in IL, [Bmim]BF4, and water as a solvent system gave excellent yields (89%–95%) in short reaction times (10–15 min) among the tested ILs, i.e., [Bmim]·BF4 and [Bmim]·PF6.
water solvent system and were then stabilized by the addition of PVA/PVP. The study summarized that obtaining triazoles from electron-deficient azides required a longer duration. CuNPs catalyst in IL, [Bmim]BF4, and water as a solvent system gave excellent yields (89%–95%) in short reaction times (10–15 min) among the tested ILs, i.e., [Bmim]·BF4 and [Bmim]·PF6.
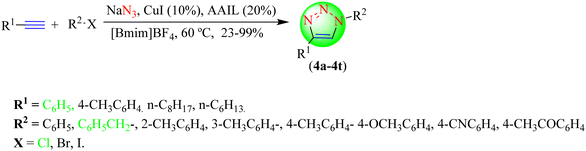
Yan et al. established a novel green protocol for synthesizing 1,2,3-triazoles (4a–4t) by reacting phenylacetylene with in situ-generated phenyl azides in the presence of CuI in [Bmim]BF4 and Bu4NOAc solvents in high yields (Scheme 4).85 Additionally, without suffering a substantial loss of activity, CuI, Bu4NOAc, and [Bmim]BF4 could be utilized for six runs. The amino acid-based IL (AAIL) used in the synthesis was obtained by treating tetrabutylammonium hydroxide with L-proline. It was found that when [Bmim]BF4 was not used in the reaction, the yield of triazoles decreased sharply. The CuI catalyst (10%) in the [Bmim]BF4 solvent system provided an 88% yield in a shorter reaction time (6 h) compared to the other catalysts, such as Cu(OAc)2, CuI, CuBr, and CuCl in different solvents, i.e., EtOH, MeOH, [Bmim]BF4, and Bu4NOAc.
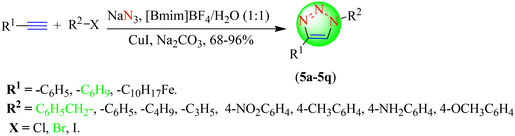
Zhao et al. reported a series of 1,2,3-triazoles via a promising, one-pot, three-component CuAAC reaction (Scheme 5).86 To synthesize various triazoles (5a–5q), in situ-obtained organic azides from halides of sp3- or sp2-hybridized carbon atoms and terminal alkynes were reacted together in [Bmim]BF4 with H2O in 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 solvent system in the presence of CuI. This method emerged as an eco-friendly procedure for the synthesis of 1,2,3-triazoles. The reaction medium [Bmim]BF4
1 solvent system in the presence of CuI. This method emerged as an eco-friendly procedure for the synthesis of 1,2,3-triazoles. The reaction medium [Bmim]BF4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (in 1
H2O (in 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) could be reused for five consecutive reactions without significant loss in activity. It was found that when benzyl azides and alkyl azides were used in the synthesis reaction, it proceeded smoothly at room temperature, required less time, and gave excellent yields. However, in the case of azides obtained in situ from the sp2-hybridized carbon atom of aromatic halides, like iodobenzene, the reaction required more time and a higher temperature for completion using L-proline. The CuI catalyst in the [Bmim]BF4
1) could be reused for five consecutive reactions without significant loss in activity. It was found that when benzyl azides and alkyl azides were used in the synthesis reaction, it proceeded smoothly at room temperature, required less time, and gave excellent yields. However, in the case of azides obtained in situ from the sp2-hybridized carbon atom of aromatic halides, like iodobenzene, the reaction required more time and a higher temperature for completion using L-proline. The CuI catalyst in the [Bmim]BF4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (1
H2O (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) solvent system provided an excellent yield of 94% in a shorter reaction time (4 h) compared to the other tested catalyst systems, including CuCN, CuBr, CuI, CuCl, and CuSO4/Vc in DMSO, THF, [Bmim][BF4]/H2O (1
1) solvent system provided an excellent yield of 94% in a shorter reaction time (4 h) compared to the other tested catalyst systems, including CuCN, CuBr, CuI, CuCl, and CuSO4/Vc in DMSO, THF, [Bmim][BF4]/H2O (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4), [Bmim][BF4], and DMSO/H2O (1
4), [Bmim][BF4], and DMSO/H2O (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4).
4).
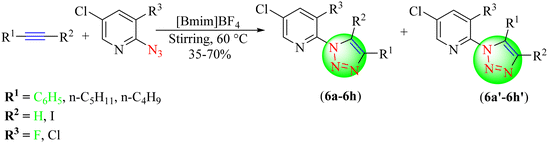
Zhong and co-workers reported a series of 1,4,5-trisubstituted 1,2,3-triazoles (6a–6h, 6a′–6h′) by reacting alkynes with the corresponding azides (2-azido-5-chloro-3-fluoropyridine or 2-azido-3,5-dichloropyridine), which in turn were obtained from 2,3,5-trichloropyridine, 5-chloro-2,3-difluoropyridine or 3,5-dichloro-2-fluoropyriridine and NaN3 in an IL [Bmim]BF4 (Scheme 6).87 The gray side of the reaction showed the longer duration, i.e., 2–7 days, and lack of regioselectivity in the products. The [Bmim]BF4 green catalyst provided considerable yields (35%–70%) in a short reaction time.
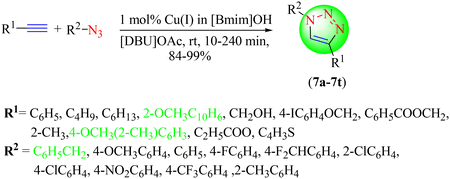
Garg et al. synthesized amino acid-containing 1,2,3-triazole hybrids (7a–7t) employing 1,8-diazabicyclo[5.4.0]undec-7-ene acetate and using an IL as a green solvent (Scheme 7).88 The synthesis of these 1,2,3-triazoles was also performed in several solvents, such as H2O, [Bmim]OH, and [Omim]OH, with different catalysts. However, the best results were obtained when 1 mol% of CuBr was used as a catalyst for reacting various terminal alkynes with azides in [DBU]OAc solvent. CuBr (1 mol%) in [Bmim]OH provided an excellent yield (90%) in a short reaction time of 1 h, among the other green solvents tested, i.e. [DBU]OAc, [Omim]Br, [Omim]OH, [Omim]NTf2, [Bmim]OH, and H2O/t-butanol.
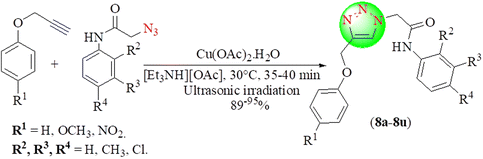
Akolkar et al. synthesized some 1,4-disubstituted 1,2,3-triazole (8a–8u) compounds from various terminal alkynes by reacting with diversified in situ-generated acetamide azides under ultrasonic irradiation in the presence of Cu(OAc)2·H2O in [Et3NH][OAc] via click chemistry (Scheme 8).89 This method has many advantages, including obtaining a high yield within a short time, reduced use of toxic solvents, and the recyclable and reusable solvent [Et3NH][OAc]. Cu(OAc)2·H2O in [Et3NH][OAc] provided an excellent yield (97%) in a short reaction time (35 min) among all the used catalysts, i.e. Cu(OAc)2·H2O, CuSO4·5H2O with sodium ascorbate, CuI and CuCl in t-BuOH![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O, THF, [Et3NH][OAc] and [Et3NH][HSO4].
H2O, THF, [Et3NH][OAc] and [Et3NH][HSO4].
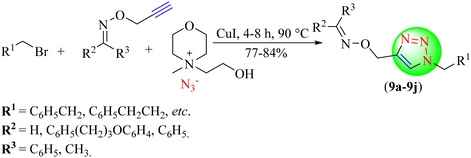
Soltani Rad and co-workers reported a novel procedure for the synthesis of 1,2,3-triazole-based hybrids (9a–9j), in which an hydroxyethyl methyl morpholinium azide-based IL (HEM Morph)N3 was used as a dual applicant, i.e., reaction media and azide source (Scheme 9).90 For targeted triazole synthesis, various terminal alkynes and aryl halides were reacted in the presence of Cu(I)-catalyst in the IL. Herein, (HEM Morph)N3 was used as an efficient, environmentally friendly, affordable, and simple substrate. The Cu(I)-catalyst (HEM Morph)N3 IL (3 mmol) provided an excellent yield (85%) in a short reaction time (4 h) among all the tested loadings of ILs.
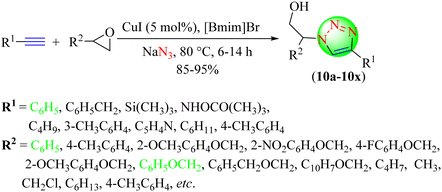
Singh and co-workers reported a facile, eco-compatible, and reliable protocol for synthesizing various β-hydroxy 1,2,3-triazole hybrids (Scheme 10).91 In this practically reliable approach, various epoxide derivatives, terminal alkynes, and sodium azide were reacted together in the presence of CuI immersed in an IL [Bmim]Br. This envisaged methodology was more effective as it provided triazoles (10a–10x) in high yields under mild reaction conditions. The catalyst was reusable for three more runs without any significant loss in efficacy. The CuI catalyst (10 mol%) with the [Bmim]Br IL solvent system provided an excellent yield (90%) in a short reaction time (15 h) among the tested catalysts with different loadings of CuI and CuCl2 in various ILs.
Narsimha et al. synthesized various 1,2,3-triazoles (11a–11o) through a one-pot CuAAC approach in which various 1-iodoalkynes were reacted with different in situ-generated organic azides in the catalytic presence of CuI in [Bmim]PF6 under microwave irradiation (Scheme 11).92 A simple workup, high yield, and short reaction time make this protocol highly beneficial. CuI catalyst (5 mol%) with [Bmim]PF6 and t-BuOK as a base provided a high yield (81%) in a short reaction time (30 min) compared to various loadings of catalyst in [Emim]BF4 and [Bmim]PF6 ILs as well as bases.
Patil et al. reported a benign and regioselective synthesis of 1,2,3-triazoles (12a–12p) by reacting terminal alkynes with in situ-generated organic azides in water using CuSO4·5H2O and a polymer-supported ascorbate-functionalized IL (MR-IMZ-As) (Scheme 12).93 This method emerged as the most promising due to its easier synthetic accessibility at room temperature, high yield, and regioselectivity of the products. The polymer-supported ascorbate-functionalized IL (MR-IMZ-As) gave good to high yields (79%–96%) in a short reaction time (2 h).
Babu et al. reported an eco-friendly protocol for synthesizing some benzoxazoles-based 1,2,3-triazoles (13a–13j) from several terminal alkynes and diversified organic azides in different ILs using CuI under microwave irradiation for 30–50 min, while using conventional method the products were obtained after continuing stirring for 8–12 h (Scheme 13).94 The maximum yield (87%) was obtained when the reaction was carried out in [Bmim]PF6 IL under microwave irradiation at 120 °C for 30 min.
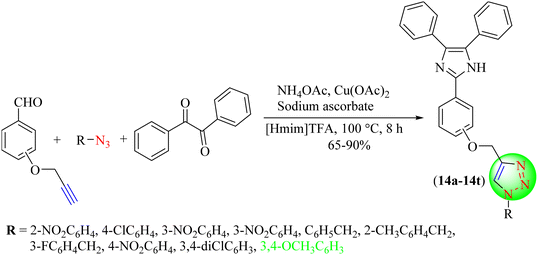
Dabiri and co-workers reported a one-pot reaction for synthesizing imidazole based on 1,2,3-triazole hybrids (Scheme 14).95 This method involves merging of the CuAAC reaction with multicomponent condensation reactions. To synthesize the desired hybrids (14a–14t), various ortho-, meta-, and para-propargylated hydroxy benzaldehydes, organic azides, benzil, and ammonium acetate were stirred together in [Hmim]TFA in the presence of Cu(OAc)2 and sodium ascorbate for 8 h at 100 °C. Reaction condition optimization studies showed that the Cu(OAc)2 (10 mol%)/sodium ascorbate (20 mol%)/[Hmim]TFA (50 mol%) IL provided excellent yield (89%) compared to the other catalysts, i.e., Cu(OAc)2/sodium ascorbate, Cu(OAc)2/sodium ascorbate/p-TsOH, CuI/IL and CuSO4/sodium ascorbate/IL.
Seeka et al. synthesized a novel series of benzimidazole-based 1,2,3-triazoles (15a–15n) from N-propargyl benzimidazole, p-TsOH hydrate, aryl amines, and NaN3 in [BMIM][PF6]·H2O at room temperature via CuAAC (Scheme 15).96 This method represents an highly efficient and benign approach for synthesizing 1,2,3-triazoles in good to excellent yields of 75%–95% using the [Bmim][PF6]·H2O solvent system.
Marra and co-workers reported a CuAAC synthesis using several ILs for reacting sugar azide with a terminal alkyne linked to sugar using CuI, i-Pr2EtN. Among the used ILs, N-octyl-dabco-cation-based dicyanamide [C8DABCO][N(CN)2] provided the highest yield (95%) of 1,2,3-triazole (16). The latter solvent was recovered without reducing the effectiveness of the reactions in four successive steps (Scheme 16).97 ILs, Ammoeng 110, and [C8DABCO][N(CN)2] furnished triazoles in excellent yields (95%) in minimum reaction times (16 h) compared to the other tested solvent systems.
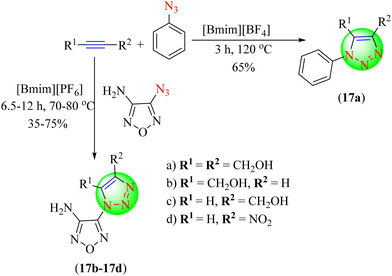
Seregin and co-workers reported that ILs are capable of being successfully utilized as a greener medium for the cycloaddition reaction of aromatic and heterocyclic azides with various acetylenes and enamines to produce 1-aryl(hetaryl)-1,2,3-triazoles (17a–17d).98 Upon comparing the performance with analogous reactions in regular organic solvents, both the rate and the regioselectivity of the responses were found to be improved. The potential of utilizing an IL again was demonstrated by reusing it three successive times in the same portion of IL with nearly no change in yield (Scheme 17). [Bmim]PF6 IL in ethanol gave product in good yield (75%) and in a short reaction time (7.5 h) compared to other ILs.
Sucharitha and co-workers reported an efficient approach for the synthesis of 1,2,3-triazoles (Scheme 18).99 To obtain the desired triazole hybrids (18a–18m), in situ-generated diversified organic azides were treated with various alkynes using ILs [Emim]BF4 and CuI under microwave irradiation. This strategy's advantages involved forming triazole hybrids in a high yield within a short time. To optimize the reaction conditions, different bases, like t-BuOLi, t-BuOK, and K2PO3, in [Emim]BF4 solvent were used, with the t-BuOK base providing excellent yield (81%) in the shortest time (2 h).
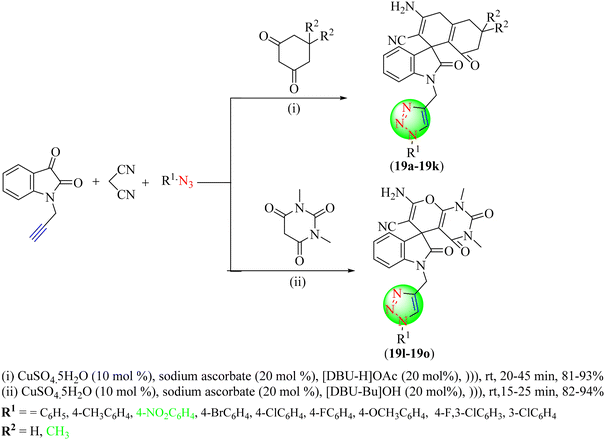
Singh et al. synthesized fluorescent triazolyl spirocyclic oxindoles (19a–19o) from 1-(prop-2-ynyl)indoline-2,3-dione, malononitrile, cyclic 1,3-diketones, and aryl azides employing DBU-derived ILs, i.e., [DBU-Bu]OH and [DBU-H]OAc, under ultrasonication (Scheme 19).100 Among the two ILs, [DBU-Bu]OH was found to be more effective for synthesizing triazole hybrids by reaction, as it required a low completion time and higher yield. Optimization of the reaction conditions showed that [DBU-H]OAc and [DBU-Bu]OH IL in several solvents (ethanol, methanol, water) provided very high yields (94%) of products through ultrasonic irradiation.
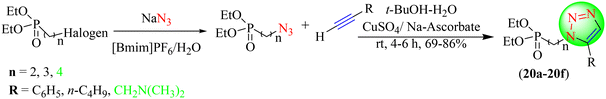
Artyushin et al. reported that 1-butyl-3-methylimidazolium hexafluorophosphate ([Bmim][PF6]) is an appropriate reusable medium for the practical synthesis of azidoalkyl phosphonates, which are valuable intermediates to feed the synthesis of CuAAC using different alkynes to yield 4-substituted (1H-1,2,3-triazol-1-yl) alkyl phosphonates (20a–20f) as a drug contender (Scheme 20).101 The [Bmim][PF6]/H2O system was quickly used to achieve 95%–98% yields of desired azides useful for triazoles synthesis without producing any side product during the reaction.
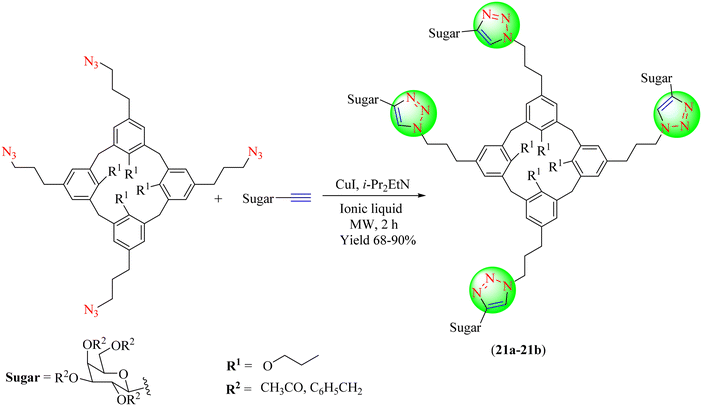
Vecchi and co-workers synthesized some sugar based 1,2,3-triazoles using CuI, i-Pr2EtN, a tetra-azido calix[4]arene derivative and ethynyl tetra-O-benzyl-C-galactoside in three distinct ILs, namely [C8DABCO][N(CN)2], [C8DABCO][Br] and Ammoeng 110. Heating and MW dielectric heat were used to carry out the processes. Multiple cycloadditions occurred in every instance, yielding triazole-linked tetra-C-galactosyl-calix[4]arenes (21a–21b) in 68%–90% yields. Multi-click reactions with propargyl O-lactose and S-seaside were also carried out using the [C8DABCO][N(CN)2] IL (Scheme 21).102 Among the three ILs, [C8DABCO][N(CN)2] IL furnished a high yield of products in a short time of 2 h.
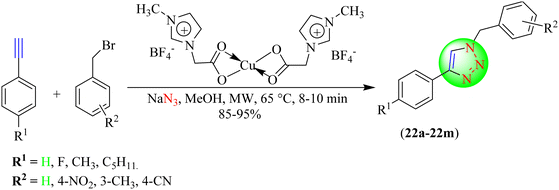
Sharma et al. developed a newer, eco-friendly, shorter time-consuming strategy for synthesizing 1,2,3-triazoles (Scheme 23).104 This strategy involved a 1,3-dipolar cycloaddition reaction between a synthesized alkyne and in situ-generated organic azides using water in the presence of an IL-based copper catalyst. The catalyst was synthesized from 1-butly-4-methylpyridinium tetrafluoroborate upon treatment with CuFe2O4 and L-tyrosine functionalized titania nanospheres (TiTCIL). This protocol yielded triazoles (23a–23l) in a good to excellent amount, needed less time for reaction completion, and the catalyst could be used for 6–7 runs without any notable decrease in activity. The IL@CuFe2O4-L-Tyr-TiO2/TiTCIL catalyst in water provided an excellent yield (95%) in a short reaction time (8 min) among the tested catalytic systems, such as Cu2O octahedra, CuFe2O4, CuI, CuFe2O4@starch and L@CuFe2O4-L-Tyr-TiO2 in various solvents, including EtOH, water![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) acetone (1
acetone (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) and water.
1) and water.
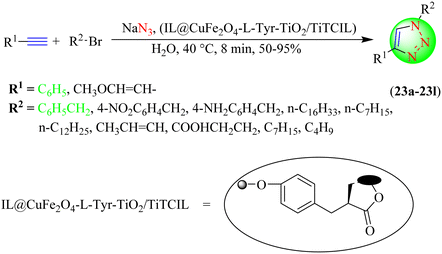
Daraie et al. reported a biocompatible process for the synthesis of 1,2,3-triazoles (24a–24m), in which phenyl-based terminal alkynes were treated with several in situ-generated phenacyl azides/benzyl azides in several solvents in the presence of chitosan IL-based copper iodide nanoparticles (Cu@SMA-FA-CS-IL) (Scheme 24).105 The results of a reaction optimization study revealed that the triazoles were obtained rapidly (8 min) in a very high yield (97%) when the reaction was performed under ultrasonic conditions at ambient temperature using the Cu@SMA-FA-CS-IL catalyst in water.
Sonawane et al. described a novel eco-friendly method for the synthesis of 1,2,3-triazoles with 85%–98% yields (Scheme 25; 25a–25w) from various in situ-generated organic azides and terminal alkynes in water using ascorbate-functionalized copper-bound polymer-supported IL-based catalyst (PSILPC).106 PSILPC-6 catalyst (10%) in a water solvent system provided an excellent yield (98%) in a short reaction time (20 min) compared to the other tested catalysts, such as PSIL 1, PSIL 2, PSIL 3, PSIL 4, PSIL 5, and PSILPC 6, in various solvents i.e., EtOH, MeOH, water, water![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ethanol (1
ethanol (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), water
1), water![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ethanol (3
ethanol (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7) and Bu4NOAc. As discussed in Scheme 26, the mechanism of the reaction involves initial π-bond coordinated complex (B) formation by the polymer-supported IL-based ascorbate-functionalized copper catalyst followed by dinuclear Cu-acetylide complex (C) formation with the propargyl unit. This dinuclear Cu-acetylide complex is later attacked by the in situ-generated organic azide to form complex D. The next step of the mechanism involves the nucleophilic attack of C-4 of the propargyl unit at N-3 of the organic azide, thus developing the first C–N covalent bond formation to establish intermediate E. The intermediate readily undergoes ring contraction to form a triazolyl-Cu complex (F), which, upon protonation, creates a 1,4-disubstituted 1,2,3-triazole ring. From the environmental and economic perspectives, this protocol emerges as the most significant as it only requires water as a solvent, a shorter reaction time, and mild reaction conditions, and with minimal effort, the catalyst could be retrieved and utilized seven more times without significantly losing its catalytic activity.
7) and Bu4NOAc. As discussed in Scheme 26, the mechanism of the reaction involves initial π-bond coordinated complex (B) formation by the polymer-supported IL-based ascorbate-functionalized copper catalyst followed by dinuclear Cu-acetylide complex (C) formation with the propargyl unit. This dinuclear Cu-acetylide complex is later attacked by the in situ-generated organic azide to form complex D. The next step of the mechanism involves the nucleophilic attack of C-4 of the propargyl unit at N-3 of the organic azide, thus developing the first C–N covalent bond formation to establish intermediate E. The intermediate readily undergoes ring contraction to form a triazolyl-Cu complex (F), which, upon protonation, creates a 1,4-disubstituted 1,2,3-triazole ring. From the environmental and economic perspectives, this protocol emerges as the most significant as it only requires water as a solvent, a shorter reaction time, and mild reaction conditions, and with minimal effort, the catalyst could be retrieved and utilized seven more times without significantly losing its catalytic activity.
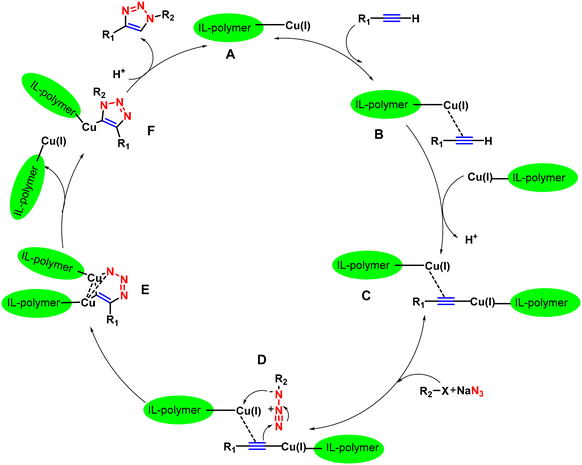 | ||
| Scheme 26 Possible mechanism of triazole formation via an IL-based Cu-catalyst.106 | ||
Kargar et al. described a green one-pot methodology for synthesizing 1,2,3-triazoles (26a–26j) from in situ-obtained phenacyl azides and various alkynes using IL-based copper nanoparticles and sodium ascorbate in water in good to high yields within a short time (Scheme 27).107 The catalyst used in the synthesis was obtained from a Cu(II)-Schiff base complex derived from the imidazolium ionic phase and immobilized on core–shell Fe3O4@NFC magnetic nanoparticles (Fe3O4@NFC-ImSalophCu). Additionally, the catalyst was recyclable and could be used four times without activity loss. Using the Fe3O4@NFC-ImSalophCu catalyst in water provided a 97% yield in 30 min compared to other solvents, i.e., EtOH, MeOH, CH3CN, DMF, as well as to solvent-free conditions.
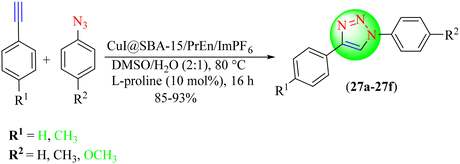
Hosseini and co-workers reported the synthesis of 1,2,3-triazoles (27a–27f) using an IL-supported heterogeneous copper catalyst, i.e., CuI@SBA-15/PrEn/ImPF6 (Scheme 28).108 They found that this heterogeneous catalyst was highly efficient for the CuAAC reaction. These studies showed that the maximum yield of triazoles was obtained with a catalyst loading of 3.5–4.0 mol% in a water/DMSO/H2O–DMSO mixture. Diversified 1,2,3-triazoles were obtained by treating various phenylacetylene derivatives with in situ-generated organic azides in a suitable solvent in the presence of an IL-supported Cu(I) catalyst. CuI@SBA-15/PrEn/ImPF6 in DMSO/H2O (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) provided an excellent yield (93%) in a short reaction time (16 h) among the tested solvents, like DMSO, H2O, EtOH, dioxane, and DMSO/H2O (2
1) provided an excellent yield (93%) in a short reaction time (16 h) among the tested solvents, like DMSO, H2O, EtOH, dioxane, and DMSO/H2O (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1).
1).
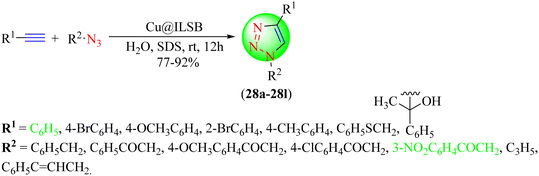
Thakur et al. reported a newer protocol for synthesizing 1,2,3-triazole hybrids (28a–28l) using copper complexes of Schiff bases (Cu@ILSB) based on benzimidazole IL as a catalyst (Scheme 29).109 To synthesize the targeted 1,2,3-triazoles, various in situ-generated organic azides were reacted with several phenylacetylene derivatives in water via CuAAC. Sodium dodecyl sulfate (SDS) was used as an additive in the reaction. The Cu@ILSB catalyst could be reused without significant loss in its catalytic activity. Cu@ILSB with SDS in water provided an excellent yield (89%) in a short reaction time (12 h) at room temperature among the tested catalysts, i.e., Cu@ILSB, catalyst-free, Cu(OAc)2·H2O, Cu(OAc)2·2H2O, CuCl2, CuI in DMF, H2O, and solvent-free conditions with the additives SDS and TBAB.
Afzali and co-workers synthesized a series of 1,2,3-triazole hybrids (29a–29i) from various terminal alkynes and diversified organic azides in water using a modified layered doublehydroxide with a Cu(II)-cation-immobilized IL-based nanocatalyst (LDH-Cu2+-IMIL) (Scheme 30).110 The IL was synthesized from LDH-IMIL and CuCl2. Its remarkable catalytic potential was attributed to the regular dispersion of copper ions in the LDH interlayer. However, the inner layer of the functionalized double-layered hydroxide nanostructure contained an IL, which provided a favorable environment for the leaching protection and stability of the immensely dispersed copper ions, leading to high efficiency and good reusable features of the nanocatalyst. Also, the nanocatalyst's increased surface area and appropriate pore size were other merits for its improved efficiency. LDH-Cu2+-IMIL in water at 80 °C provided an excellent yield (90%) in a short reaction time (3 h) among all the tested solvent systems.
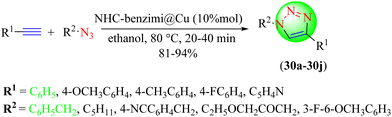
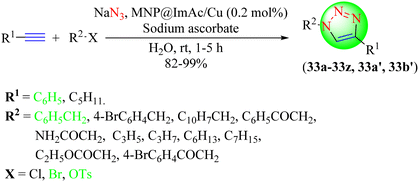
Pawar et al. developed a heterogeneous silica-coated NHC-benzimi@Cu(I)-catalyst for the eco-friendly synthesis of 1,2,3-triazole hybrids via CuAAC reaction (30a–30j) from terminal alkynes and in situ-generated organic azides obtained from alkyl or aryl halides on reacting with with sodium azide (Scheme 31).111 The remarkable performance of the catalyst and its easy separation from the reaction mixture made it more promising among other catalysts. The nano-magnetite NHC-benzimi@Cu catalyst (10 mol%) provided an excellent yield (94%) in a short reaction time (20 min) among the tested nano-magnetite NHC-benzimi@Cu, [nano-magnetite-benzimi]Cl and nano-magnetite with different catalyst loadings. The heterogeneous nanocatalyst was also reusable without any loss of catalytic potential. The mechanism for the formation of the target triazoles (Scheme 32) involved an initial Cu(I)-complex as intermediate (I) with an organic azide, which upon attack by a terminal alkyne form a pseudo Cu-acetylide complex as intermediate (II), followed by a six-membered Cu-complex formation as intermediate (III). This six-membered Cu-complex undergoes ring contraction to form a five-membered triazole Cu-complex formation to yield the final 1,2,3-triazole after Cu(I) removal.
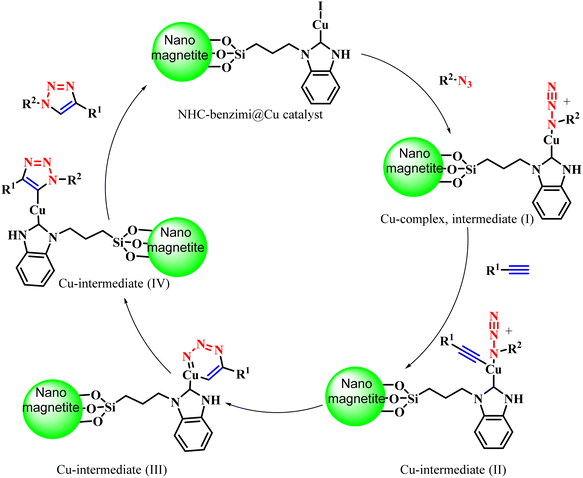 | ||
| Scheme 32 Possible mechanism for the synthesis of 1,2,3-triazoles using a heterogeneous silica-coated NHC-benzimi@Cu(I)-catalyst.111 | ||
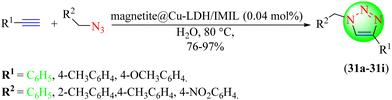
Leilan et al. developed a new IL-based catalyst to synthesize 1,2,3-triazoles (Scheme 33).112 Herein, a new nanoreactor catalyst comprising copper-doped bi-layer hydroxide magnetite as a novel nanocatalyst was strategically produced using a hydrothermal method followed by postmodification. The catalyst surface was further modified with an imidazole-based IL to increase its dispersion ability in reaction media. The newly synthesized catalyst, i.e., magnetite@Cu-LDH/IMIL, proved to be an effective catalyst for obtaining1,2,3-triazoles (31a–31i) from the cycloaddition of various terminal alkynes with organic azides in water. Further, this catalyst was easily recoverable due to its magnetic character and could be reused for up to five consecutive runs without loss in activity. The magnetite@Cu-LDH/IMIL (0.04) catalyst in water provided a very high yield (90%) in a short reaction time (3 h) among the evaluated catalysts, i.e., magnetite@Cu-LDH/IMIL, CuMgAl-LDH, IMIL and magnetite nanospheres in water, ethanol, chloroform, acetonitrile, and toluene.
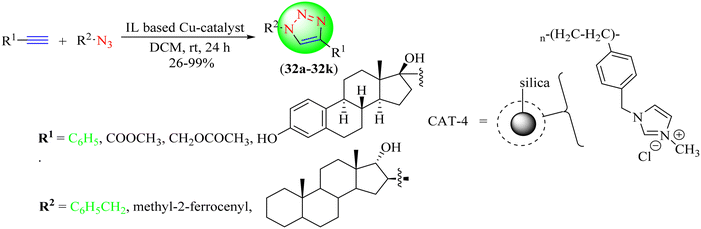
Fehér and co-workers developed a newer catalyst for Huisgen's cycloaddition of terminal alkynes with organic azides to yield 1,2,3-triazoles (32a–32k) from various terminal alkynes and organic azides in DCM at room temperature (Scheme 34).113 The heterogeneous catalyst was obtained by depositing an IL obtained from N-methyl imidazole and 4-vinylbenzyl chloride on silica, and then by copper deposition using cuprous iodide. The catalyst could be recovered and recycled multiple times without considerable loss of efficacy. The study showed that bulky substituents, such as a steroid or ferrocene ring, resisted the cycloaddition with bulky azides. The CAT-2 catalyst with minimum loading provided excellent yields (26%–99%) in a short reaction time (24 h) among the tested CAT-1, CAT-2, CAT-3, and CAT-4 samples.
Pourjavadi et al. designed and synthesized a novel poly-IL-based copper catalyst, which was found to be helpful for the 1,3-dipolar cycloaddition reaction of terminal alkynes with organic azides to yield 1,2,3-triazoles (33a–33z, 33a′, 33b′) at room temperature in aqueous solution (Scheme 35).114 To synthesize the catalyst, 3-carboxymethyl-1-vinylimidazolium was polymerized using surface-modified magnetic nanoparticles. Further, the carboxylate units of the polymer chains were coordinated with copper sulfate. The catalysts could be recycled and reused without any loss in activity. Many catalysts, including MNP, CuSO4, CuSO4/ImAc, MNP@ImAc, and MNP@ImAc/Cu with different amounts of catalyst loading in H2O–EtOH, H2O, hexane, CH3OH, THF, could be used for triazoles synthesis. However, MNP@ImAc/Cu with 0.2 mol% in a water solvent system provided the best yield (99%) in a short reaction time (2 h).
Mohan and co-workers synthesized an IL-based copper catalyst Cu[(OHCH2CH2)2NH]6[CF3SO3]2, which proved to be an effective, inexpensive, and green catalyst for the synthesis of 1,2,3-triazoles (Scheme 36).115 To synthesize 1,4-disubstituted 1,2,3-triazoles (34a–34j), various terminal alkynes and organic azides were reacted together in the presence of this IL-based catalyst in dimethylformamide. Triazole hybrids were obtained with excellent yields without reducing agents, bases, or an inert atmosphere, etc. It was found that the Cu[(OHCH2CH2)2NH]6[CF3SO3]2 IL catalyst in DMF solvent furnished a high yield (96%) of triazoles in a short reaction time (30 min) compared to other catalysts, like Cu(OTf)2 + DEA, Cu(OTf)2, etc., in different solvents.
Liu et al. developed a reusable bifunctional IL-based (choline chloride – CuCl) valuable catalyst for obtaining 1,2,3-triazole hybrids (35a–35p) from terminal alkynes, organic halides, and sodium azide in water via a CuAAC reaction (Scheme 37).116 The desirable characteristics of this synthesis strategy were the use of green and moderate reaction conditions, avoiding the isolation of hazardous azides, and ease of handling. It was observed that the (ChCl)-based CuCl-IL catalyst (5.0 mol%) furnished the highest yield (89%) in a short reaction time (5 h) compared to different catalyst loadings.
Wang et al. synthesized two IL-based copper catalysts, i.e. Cu-CPSIL and Cu-PSIL, which were found to be very useful for the synthesis of 1,2,3-triazole hybrids (Scheme 38).117 To synthesize the target hybrids (36a–36j), various terminal alkynes were treated with in situ-generated organic azides in water with the catalytic presence of these IL-based copper catalysts. These catalysts could be quickly recovered and reused several times without loss of activity. Among the two catalysts, Cu-CPSIL was the most appropriate for synthesizing the targeted triazole hybrids with excellent yields (80%–98%). The Cu-PSIL catalyst (1 mol%) provided triazoles with excellent yields (98%) compared with the other tested catalysts with different copper loadings, like Cu-CPSIL, Cu-PSIL, and CuO/SiO2.
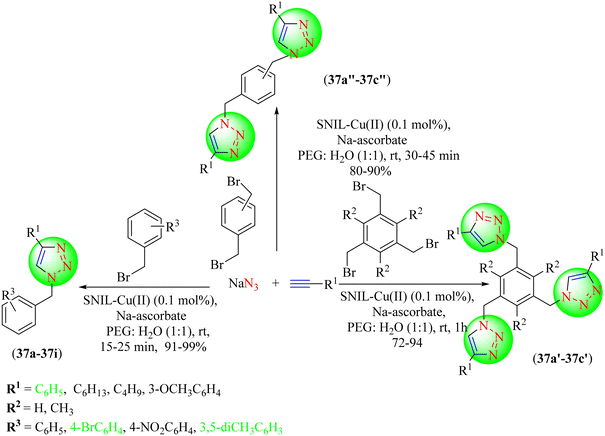
Tavassoli et al. synthesized a silica-supported copper-containing IL that was useful for the click synthesis of 1,2,3-triazoles (Scheme 39).118 To obtain the various 1,2,3-triazole hybrids (37a–37i, 37a′–37c′, 37a′′–37c′′), various aryl halides, alkyl/phenyl acetylenes, sodium azide, and catalyst, i.e., SNIL-Cu(II), and Na ascorbate were treated together in a PEG-400/H2O solvent system. The synthesized IL-based catalyst was found to be an active, reusable, solid-phase catalyst and highly stable, and could be reused several times without significant loss of efficacy. The SNIL-Cu(II) catalyst with Cu(OTf)2 copper salt gave an excellent yield (99%) in a short reaction time (25 min) compared to CuCl2, Cu(OTf)2, CuCl and CuI.
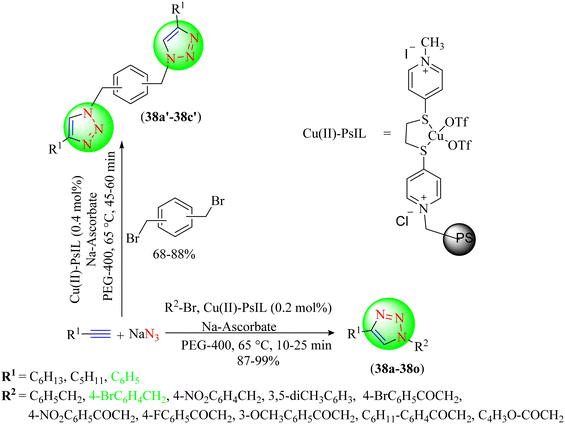
Tavassoli et al. also prepared an IL-based copper catalyst by the immobilization of a copper(II) complex of 1,2-bis(4-pyridylthio)ethane onto polystyrene (Cu(II)-PsIL) (Scheme 40).119 The catalyst was then used to synthesize 1,2,3-triazoles (38a–38o, 38a′–38c′) from organic halides, sodium azide, and terminal alkynes via click chemistry. The synthesized catalyst was highly active, stable, and recyclable for the green synthesis of triazoles. The catalyst was reusable after recycling it seven times without losing catalytic efficacy. The reaction optimization strategy showed that the Cu(II)-PsIL-based catalyst in PEG-400 solvent provided a very high yield (99%) compared to other solvents in a short reaction time (15 min).
Dabiri et al. reported an IL supported on graphene that provided efficient and potent support for the immobilization and stabilization of the copper complex. This was achieved by anchoring an N-heterocyclic carbene-copper complex over graphene oxide transformed with an IL. The Cu-NHC/GO-IL composite showed excellent catalytic ability during the Huisgen 1,3-dipolar cycloaddition involving aryl azides and terminal alkynes (Scheme 41).120 Triazoles (39a–39h) were obtained with a good to excellent yields of 89%–99%. Cu-NHC/GO-IL in EtOH![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (1
H2O (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) provided a high yield (93%) in a short reaction time (8 h) compared to when the reaction was performed in MeOH, MeCN, EtOH, H2O, and EtOH
1) provided a high yield (93%) in a short reaction time (8 h) compared to when the reaction was performed in MeOH, MeCN, EtOH, H2O, and EtOH![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O.
H2O.
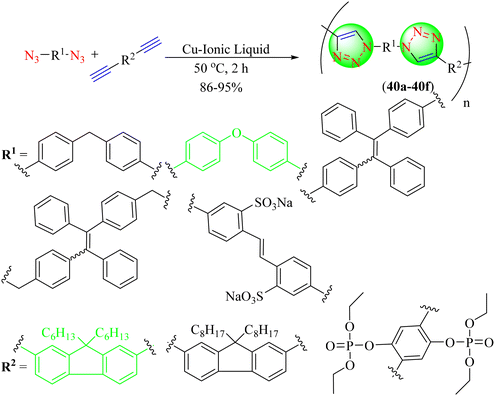
Li and co-workers discovered a new and effective Cu-IL-catalyzed azide–alkyne click polymerization (AACP) reaction. The polymerization proceeded efficiently under benign conditions, yielding soluble and thermally stable 1,4-regioregular poly triazoles (40a–40f) with large molecular weights. Because of its excellent water solubility and high fluorescent emission, the benefits of this catalyst included the absence of external ligands and reducing agents and its ease of synthesis and storage (Scheme 42).121 Cu-IL, CuBr, CuI, and Cu(PPh3)3Br catalysts were used for click polymerization, but Cu-IL gave an excellent yield (94%) in the shortest time (2 h).
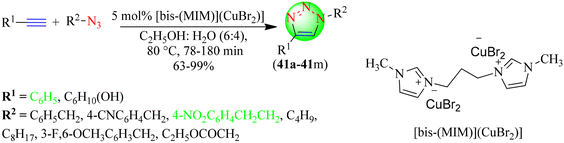
Dige et al. reported a novel green methodology for synthesizing 1,2,3-triazoles (41a–41m) following Huisgen's 1,3-dipolar cycloaddition reaction (Scheme 43).122 To synthesize the targeted triazole hybrids various terminal alkynes were reacted with in situ-obtained organic azides in an ethanol–water solvent (6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4) system in the presence of an IL-based copper catalyst, i.e. 1,3-bis(1-methyl-1H-imidazol-3-ium)propane copper(I)dibromate [bis-(MIM)](CuBr2), in good to excellent yields (63%–99%) within 78–120 min. The advantages of this synthetic strategy were the reusability of the catalyst and operational simplicity. The IL-based copper catalyst [bis-(MIM)](CuBr2) at 5 mol% gave a good to excellent yield of triazoles in the shortest time (1.4 h).
4) system in the presence of an IL-based copper catalyst, i.e. 1,3-bis(1-methyl-1H-imidazol-3-ium)propane copper(I)dibromate [bis-(MIM)](CuBr2), in good to excellent yields (63%–99%) within 78–120 min. The advantages of this synthetic strategy were the reusability of the catalyst and operational simplicity. The IL-based copper catalyst [bis-(MIM)](CuBr2) at 5 mol% gave a good to excellent yield of triazoles in the shortest time (1.4 h).
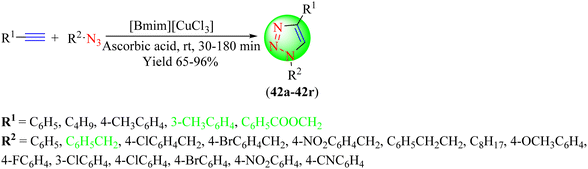
Phukan et al. synthesized a series of 1,2,3-triazoles (42a–42r) using a straightforward and cost-effective protocol, in which a low-copper-loaded IL [Bmim][CuCl3] was used as a solvent and catalyst (Scheme 44).123 This protocol was more effective as it reduced the use of additional ligands, bases, and solvents for the cycloaddition of terminal alkynes with organic azides to yield 1,2,3-triazoles. The IL in the reaction was synthesized from CuCl2 upon treatment with 1-butyl-3-methylimidazolium chloride (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) in acetonitrile. The catalyst demonstrated its efficacy as a non-cytotoxic substance due to its low copper loading; also extending its adaptability for other synthetic applications. Copper-loaded IL [Bmim][CuCl3] along with the additive ascorbic acid under solvent-free conditions provided a 94% yield in 30 min compared to when the reaction was carried out in different solvents using ascorbic acid, urea, hydrazine, and glucose as additives.
1) in acetonitrile. The catalyst demonstrated its efficacy as a non-cytotoxic substance due to its low copper loading; also extending its adaptability for other synthetic applications. Copper-loaded IL [Bmim][CuCl3] along with the additive ascorbic acid under solvent-free conditions provided a 94% yield in 30 min compared to when the reaction was carried out in different solvents using ascorbic acid, urea, hydrazine, and glucose as additives.
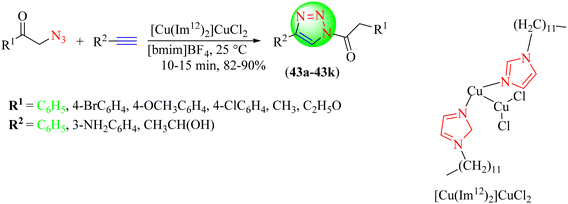
Ahmady et al. reported a new methodology for the synthesis of 1,2,3-triazoles (43a–43k), in which an IL-based copper complex was used as a catalyst to react alkynes with organic azides via Huisgen's 1,3-dipolar cycloaddition in a suitable solvent, preferably [Bmim]BF4 for better yields. [Cu(Im12)2]CuCl2 with the [Bmim]BF4 solvent system provided an excellent yield (90%) in a short reaction time (10 min) among the tested solvent systems, i.e., [Bmim]BF4, dioxane/H2O, THF/H2O, [Bmim]BF4/H2O, CH2Cl2, etc. (Scheme 45).124 This catalyst was more efficient and advantageous than the previously reported ones, as it was heterogeneous, green, and reusable for the click synthesis of 1,2,3-triazoles and could be used in a wide range of solvents. As discussed in Scheme 46, the mechanism of the reaction involved the formation of an initial acetylide complex through the IL-immobilized Cu(I) green catalyst [Cu(Im12)2]CuCl2, which was further attacked by organic azide to form a six-membered complex. Later, the six-membered complex yielded a five-membered complex upon ring contraction, which underwent protonation to create the targeted triazole hybrid.
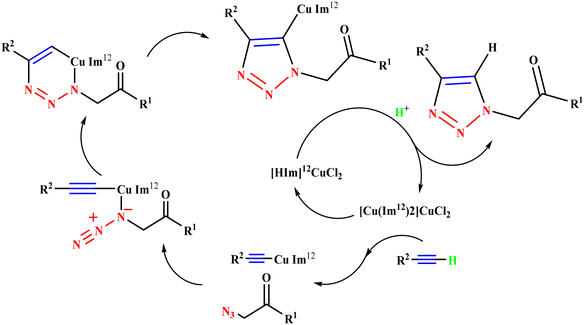 | ||
| Scheme 46 Possible mechanism for the synthesis of 1,2,3-triazoles.124 | ||
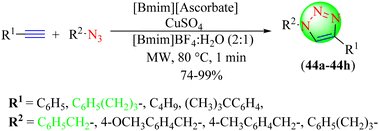
Koguchi et al. reported a new synthetic methodology for synthesizing 1,2,3-triazoles (Scheme 47).125 To synthesize the targeted hybrids (44a–44h), various alkynes were treated with diversified organic azides in different IL and water mixtures in the presence of ascorbate-functionalized IL catalysts under MW treatment. The triazoles were obtained in high yield when the reactions were carried out using [Bmim][ascorbate] and [Bmim]BF4 as the catalyst and solvent, respectively. This method provides easy access to recover the catalyst, which could be reused for five to six runs without any loss in activity. [Bmim]BF4 IL–H2O (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) gave an excellent yield of 99% in a short reaction time (1 min) compared to [Bmim]BF4, [Emim]BF4, [Emim]PF6, and [Bmim]MeSO4.
1) gave an excellent yield of 99% in a short reaction time (1 min) compared to [Bmim]BF4, [Emim]BF4, [Emim]PF6, and [Bmim]MeSO4.
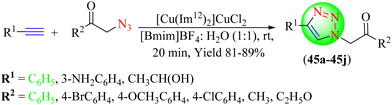
Keshavarz et al. described a green approach for synthesizing 1,2,3-triazoles (45a–45j) utilizing ILs as a catalyst and solvent (Scheme 48).126 To synthesize the targeted triazoles, several terminal alkynes were reacted with various organic azides at room temperature in the [Bmim]BF4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (1
H2O (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) solvent system in the presence of [Cu(Im12)2]CuCl2, an IL-based catalyst. The results of the study showed that this method was cost-effective and eco-friendly for the synthesis of 1,2,3-triazoles. The catalyst could be quickly recovered and reused several times without significant loss in catalytic efficacy. [Cu(Im12)2]CuCl2 IL in [Bmim]BF4/H2O 1
1) solvent system in the presence of [Cu(Im12)2]CuCl2, an IL-based catalyst. The results of the study showed that this method was cost-effective and eco-friendly for the synthesis of 1,2,3-triazoles. The catalyst could be quickly recovered and reused several times without significant loss in catalytic efficacy. [Cu(Im12)2]CuCl2 IL in [Bmim]BF4/H2O 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 led to an excellent yield (89%) in a shorter reaction time (20 min) compared to the other solvent systems tested.
1 led to an excellent yield (89%) in a shorter reaction time (20 min) compared to the other solvent systems tested.
Ketsomboon et al. described a one-pot glycosidation-CuAAC process for the synthesis of alkyl-chain triazolylglycosides (46a–46l, 46a′–46l′) by utilizing [bpy][Br] IL as a promoter and as a solvent. To produce the appropriate products with good yields, the reactions were performed with readily accessible substrates, such as glycosyl bromides, sodium azide, and different long alkyl-chain alkynes. Furthermore, they demonstrated little cytotoxicity to human endothelial cells, which makes them appropriate for use in consumer applications (Scheme 49).127 The [bpy][Br] IL/DMF co-solvent without any additive yielded triazoles in a high yield (85%) in short reaction times when compared to the other optimization conditions with different co-catalysts and ILs with and without an additive catalyst.
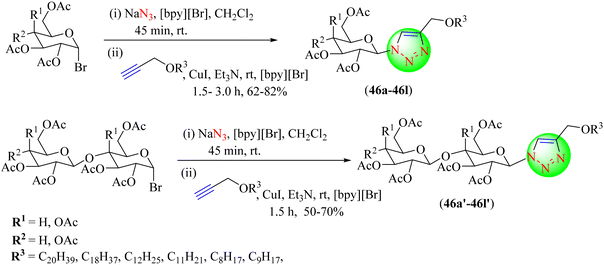 | ||
| Scheme 49 Synthesis of 1,2,3-triazoles (46a–46l, 46a′–46l′) using IL [bpy][Br] as a promoter and a solvent. | ||
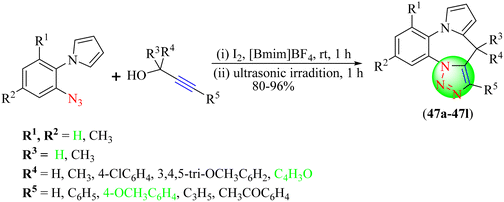
Saquib and co-workers developed an eco-friendly synthetic stratagem for the synthesis of various pyrrolobenzodiazepine-fused 1,2,3-triazole hybrids (47a–47l) via an intramolecular 1,3-dipolar cycloaddition reaction (Scheme 50).128 This reaction involved use of inexpensive, benign, recyclable molecular iodine-IL, i.e., iodine-[Bmim][BF4], as a catalyst and reaction medium. To achieve the targeted triazoles, alkynes were treated with diversified organic azides in the presence of iodine and various IL-based catalysts and reaction media. An excellent yield of triazoles (80%–96%) was obtained with the catalyst I2-[Bmim][BF4] under ultrasonic conditions. The iodine-[Bmim][BF4] catalyst gave a high yield (90%) in the shortest time (2 h). The order of catalytic efficacy was found to be [Bmim][BF4] > [Bmim]PF6 > [Bmim]Br > [Bmim]Cl > [Bmim]ClO4 > [Bmim]OAc > [Bmim]OH for the synthesis of 1,2,3-triazoles.
2.2. Synthesis of 1,2,3-triazoles from substrate molecules other than alkynes using ILs
This section describes synthetic strategies using various substrates other than a terminal alkyne and is further classified as section 2.2.1. Synthesis of 1,2,3-triazoles using ILs as a solvent; section 2.2.2. Synthesis of 1,2,3-triazoles using an IL-based catalyst system, and section 2.2.3. Synthesis of 1,2,3-triazoles using ILs as a solvent and catalyst system, as discussed below:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (1
H2O (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.1) solvent system at 100 °C. The mechanism, as shown in Scheme 52, involved the initial coordination of erbium(III) triflate to nitroolefin to generate an activated intermediate adopting a chair conformation, which was stabilized by H-bonding with H2O along with electrostatic interactions with the cation and anions of IL with proper alignment of the azide and nitroolefin in two parallel planes having a highly ordered arrangement. The 1,3-dipolar cycloaddition transition state was formed by stacking azide and nitroolefin moieties. Further, the highly ordered arrangement of these cation and anion units improved the HOMO
0.1) solvent system at 100 °C. The mechanism, as shown in Scheme 52, involved the initial coordination of erbium(III) triflate to nitroolefin to generate an activated intermediate adopting a chair conformation, which was stabilized by H-bonding with H2O along with electrostatic interactions with the cation and anions of IL with proper alignment of the azide and nitroolefin in two parallel planes having a highly ordered arrangement. The 1,3-dipolar cycloaddition transition state was formed by stacking azide and nitroolefin moieties. Further, the highly ordered arrangement of these cation and anion units improved the HOMO![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) LUMO:: dipole
LUMO:: dipole![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) dipolarophile interface. This transition state decomposed to a triazoline substrate, which yielded 1,5-disubstituted 1,2,3-triazole by eliminating HNO2. The reaction was optimized with various catalysts, like AgOTf, Zn(OTf)2, Er(OTf)3, Cu(OTf)2, Sc(OTf)3, etc. The Er(OTf)3 (10%) catalyst in an IL
dipolarophile interface. This transition state decomposed to a triazoline substrate, which yielded 1,5-disubstituted 1,2,3-triazole by eliminating HNO2. The reaction was optimized with various catalysts, like AgOTf, Zn(OTf)2, Er(OTf)3, Cu(OTf)2, Sc(OTf)3, etc. The Er(OTf)3 (10%) catalyst in an IL![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (1
H2O (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.1) solvent system gave the maximum yield (91%) in the shortest time (3 h).
0.1) solvent system gave the maximum yield (91%) in the shortest time (3 h).
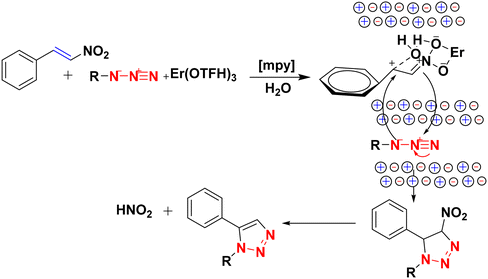 | ||
| Scheme 52 Possible mechanism for the synthesis of 1,5-disubstituted 1,2,3-triazole using Er(OTf)3, IL, and H2O.129 | ||
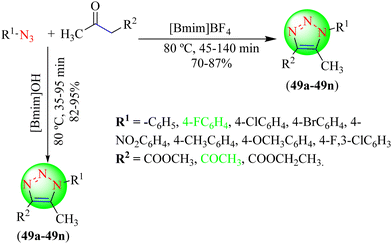
Singh and co-workers synthesized some 1,4,5-trisubstituted 1,2,3-triazoles (49a–49n) by stirring various aryl azides and acetylacetone or ethyl acetoacetate or methyl acetoacetate derivatives in ILs, i.e. [Bmim]BF4 and [Bmim]OH, at 80 °C (Scheme 53).130 When the reaction was performed in [Bmim]BF4 solvent, L-proline was used as a catalyst, but when the reaction was performed in [Bmim]OH, no catalyst was required as [Bmim]OH served as both the catalyst and solvent. Upon comparing the results of both reactions, it was found that triazoles obtained using [Bmim]OH gave a high yield of products in less time. The reaction was optimized with different catalysts in ILs [Bmim]BF4 and [Bmim]OH solvents, but L-proline (10 mol%) in IL [Bmim]BF4 at 80 °C gave high yields (82%–95%) in the shortest time.
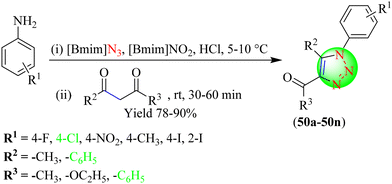
Valizadeh and co-workers synthesized a series of 1,2,3-triazoles (50a–50n) using ILs as a reagent and solvent for the reaction (Scheme 54).131 Initially, to synthesize triazoles, various aniline derivatives were mixed thoroughly with [Bmim]N3 (IL-2), [Bmim]NO2 (IL-1), and HCl to yield the in situ organic azides, which were treated with various 1,3-diketone derivatives at room temperature to produce the corresponding triazole hybrids. This protocol provided a more accessible pathway for synthesizing 1,2,3-triazoles in good yields (78%–90%) within a short time (30–60 min). Further, the use of IL-2 avoided the use of toxic NaN3.
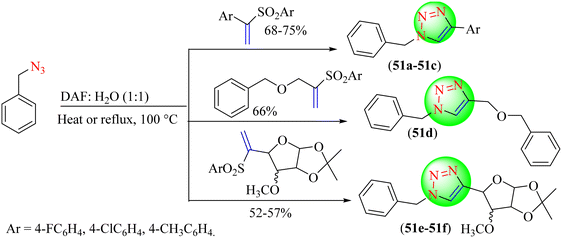
Sarkar et al. reported a new metal-free approach for the synthesis of 1,2,3-triazoles (51–51f), in which various vinyl sulfones were treated with diversified organic azides via an eliminative azide-olefinic cycloaddition (EAOC) reaction (Scheme 55).132 It was observed that the reaction medium played a significant role in synthesizing 1,2,3-triazoles as the reaction timing and product yield depended on it. The highest yield of triazoles (68%–75%) was obtained when phenyl-based vinyl sulfones were treated with benzyl azides in a DAF![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (1
H2O (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 v/v) solvent system at 100 °C. Among the tested solvents, the mixture of IL N,N-dimethyl ethanol ammonium formate with water was found to be the best.
1 v/v) solvent system at 100 °C. Among the tested solvents, the mixture of IL N,N-dimethyl ethanol ammonium formate with water was found to be the best.
De Nino et al. described the synthesis of 1,4,5-trisubstituted 1,2,3-triazoles (52a–52p) using an IL![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (1
H2O (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.1) solvent system (Scheme 56).133 To obtain the desired triazoles, various enaminone derivatives were treated with diverse organic azides using triethylamine in an [mpy]OTf
0.1) solvent system (Scheme 56).133 To obtain the desired triazoles, various enaminone derivatives were treated with diverse organic azides using triethylamine in an [mpy]OTf![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (1
H2O (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.1) solvent system. The reaction mechanism, as per the DFT calculations, showed that initially, the reaction started with a water-catalyzed 1,3-dipolar cycloaddition and then proceeded through a base-catalyzed retro-aza-Michael reaction. The reaction was optimized with different Lewis acids and bases in different solvents. Among the optimized bases, the presence of Et3N in the [mpy]OTf
0.1) solvent system. The reaction mechanism, as per the DFT calculations, showed that initially, the reaction started with a water-catalyzed 1,3-dipolar cycloaddition and then proceeded through a base-catalyzed retro-aza-Michael reaction. The reaction was optimized with different Lewis acids and bases in different solvents. Among the optimized bases, the presence of Et3N in the [mpy]OTf![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (1
H2O (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.1) solvent system provided an excellent yield (84%) in a short reaction time (5 h).
0.1) solvent system provided an excellent yield (84%) in a short reaction time (5 h).
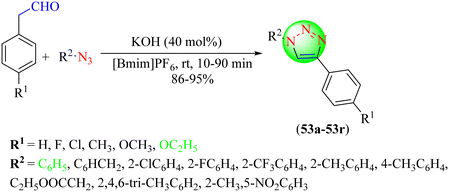
Jiang et al. reported a highly efficient, green approach for synthesizing 1,4-disubstituted 1,2,3-triazoles (53a–53r).134 This method involved triazole synthesis from the reaction of arylacetaldehydes with organic azides at room temperature using a [Bmim]PF6/KOH reaction medium. The mechanism of this reaction involved the [Bmim]PF6/KOH system producing a carbene which, upon reaction with arylacetaldehydes, converted them into enolates, which further reacted with organic azides to yield 1,2,3-triazoles, which subsequently were converted into 1,2,3-triazoles upon elimination (Scheme 57). KOH at 40 mol% provided the maximum yield (91%) in the shortest time (10 min) in [Bmim]PF6 among the used catalysts, like Na2CO3, K2CO3, KHCO3, and KOH with different catalyst loadings (mol%).
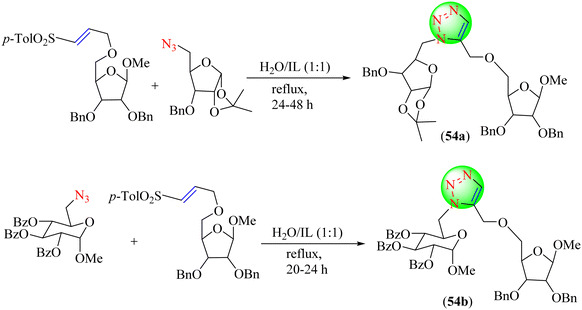
Kayet et al. exploited readily accessible vinyl sulfonyl methylene-modified furanosides and pyranosides to synthesize novel 1,5-disubstituted 1,2,3-triazolyl methylene-linked disaccharides (54a–54b). Especially compared to standard solvents, applying a biodegradable IL and water in combination significantly reduced the reaction time and improved the triazole yield (Scheme 58).135 Among the used solvent systems, i.e., water/N,N-dimethyl ethanol ammonium formate (DAF), dimethyl ethanol ammonium chloride (DAC) or N,N-dimethyl ethanol ammonium acetate (DAA), water![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) IL (1
IL (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), water–DAF furnished the triazoles in a high yield (80%) and short time (24 h).
1), water–DAF furnished the triazoles in a high yield (80%) and short time (24 h).
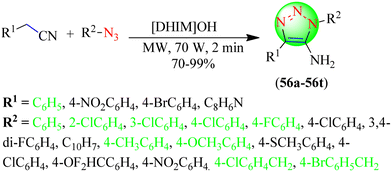
Dutta et al. reported an eco-friendly procedure for synthesizing 5-amino 1,2,3-triazole derivatives (Scheme 60).137 To synthesize the desired triazole hybrids (56a–56t), several benzyl cyanide and phenyl azide derivatives were treated in the bifunctional catalyst [DHIM]OH under microwave irradiation at 130 °C. This protocol emerges as an important one for synthesizing 1,2,3-triazoles as it does not require any metal catalyst and provides excellent yields (70%–99%) of triazoles. The reaction optimization disclosed that [DHIM][OH] (15 mol%) under solvent-free conditions at 70 W gave an excellent yield (99%) in the minimum time (2 min) compared to the other reaction conditions, i.e., KOH, NaOH, LiOH, [BMIM][OH], [DHIM][OH], etc. with different catalyst loadings with and without the solvent.
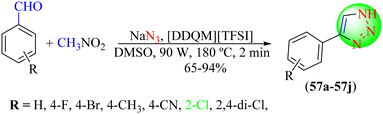
Dutta et al. also reported a newer and green approach for obtaining 1,2,3-triazoles (57a–57j), in which various benzaldehyde derivatives were reacted with nitromethane and sodium azide using1-dodecylquinolin-1-ium bis(trifluoromethane)sulfonimide ([DDQM][TFSI]) in dimethyl sulfoxide under microwave irradiation (Scheme 61).138 All the triazoles were obtained in good to high yields (65%–94%) within 2 min. As shown in Scheme 62, the plausible mechanism involved the initial formation of nitrostyrene from the reaction of aldehyde derivatives with nitroalkanes, which, in addition to the azide to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond, formed an intermediate (I). This intermediate undergoes an intramolecular nucleophilic cyclization reaction to produce the triazole intermediate (II) in a [DDQM][TFSI]/DMSO system, which leads to the formation of 1,2,3-triazoles after the elimination of HNO2.
C bond, formed an intermediate (I). This intermediate undergoes an intramolecular nucleophilic cyclization reaction to produce the triazole intermediate (II) in a [DDQM][TFSI]/DMSO system, which leads to the formation of 1,2,3-triazoles after the elimination of HNO2.
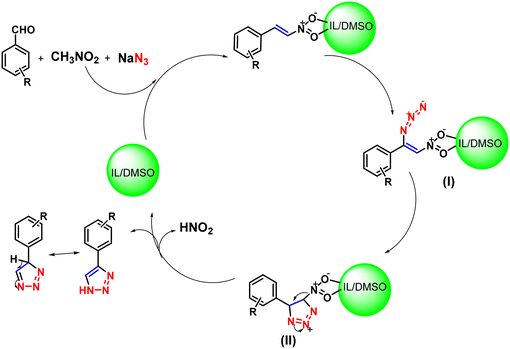 | ||
| Scheme 62 Mechanistic representation of 1,2,3-triazole synthesis using ([DDQM][TFSI]) in DMSO under microwave irradiation.138 | ||
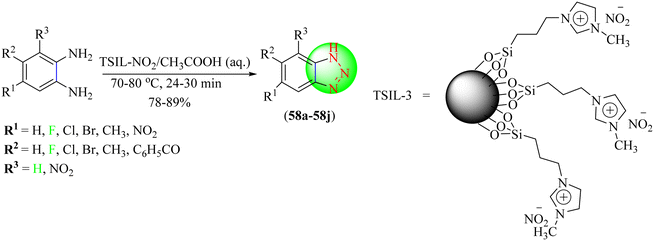
Valizadeh et al. reported that nanoparticles of organosilane-based nitrite IL immobilized upon silica, 1-butyl-3-methylimidazolium nitrite, and 1-(3-trimethoxysilylpropyl)-3-methylimidazolium nitrite were efficient reagents for the synthesis of benzotriazole derivatives (58a–58j) from 1,2-diaminobenzenes in solvent-free conditions. In this technique, these ILs serve as nitrosonium suppliers. The ILs were used to treat 1,2-diaminobenzene derivatives, yielding the corresponding diaminobenzenes in satisfactory to outstanding yields in less time (Scheme 63).139 Among the tested ILs, 1-(3-trimethoxysilylpropyl)-3-methylimidazolium nitrite was found to be most effective with 78%–89% yields of triazoles within 24–30 min. The organosilane-based nitrite IL (TSIL-3) provided very high yields in the shortest times among the three tested catalysts, i.e., TSIL-1, TSIL-2, and TSIL-3.
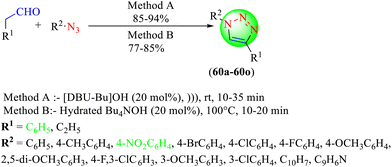
Singh and co-workers synthesized some 1,2,3-triazole hybrids (60a–60o) from aryl azides and aldehydes in the essential IL [DBU-Bu]OH by ultrasonication and using hydrated IL tetrabutylammonium hydroxide (Bu4NOH) through conventional heating (Scheme 65).141 The synthetic process was simple, provided a good yield of products, involved easy workup, operational simplicity, and required a shorter time for completion. Also, the IL could be recovered and recycled without the loss of any significant catalytic activity. The [DBU-Bu]OH (20 mol%) gave a high yield in the minimum time (10 min) without any solvent under ultrasonication. In contrast, hydrated Bu4NOH (20 mol%) provided an excellent yield without any solvent in a short time.
3. Advantages and disadvantages of ILs in organic synthesis
The superior physiochemical properties of ILs, such as low vapor pressure, non-volatility, non-flammability, excellent conductivity, and electrochemical and thermal stability, are responsible for their potential applications as catalysts and solvents in organic synthesis. These characteristics of ILs are also helpful for improving various aspects of chemical reactions, such as the catalyst recyclability, selectivity, and reactivity. Their use as benign solvents and green catalysts has garnered significant attention in recent organic synthesis. However, some gray sides of ILs as green solvents and catalysts may sometimes need our attention to carefully handle/use them as a part of reaction systems, such as that some ILs are harmful to human cells and aquatic creatures, which has prompted concerns about their safety in many applications. Also, sometimes the synthesis of valuable ILs requires multistep synthesis and expensive precursors, which makes their synthesis tedious and costly.4. Future perspective
The use of ILs as a reaction medium or catalysts and in other greener approaches, like stirring and heating technology, can help researchers to develop more environmentally friendly, sustainable, and efficient pathways to various chemical conversions, including 1,2,3-triazole synthesis. Some future perspectives in ILs are given below.(a) As the area develops, it would be useful to regularly monitor and reduce the environmental impact of the ILs of choice. Investigating the usage of renewable and biodegradable ILs may help to improve synthetic processes’ overall sustainability.
(b) The exploration of novel ILs with customized characteristics could improve the catalytic activity and selectivity in the production of 1,2,3-triazoles or other target molecules.
(c) Their potential benefits could prompt an interest among chemists to develop ILs that are more efficient, cost-effective, and completely ecosystem-friendly.
(d) A better understanding of the mechanisms involved in producing 1,2,3-triazoles in IL settings can provide valuable insights for further optimization and reaction control. Computational and kinetic research could help illuminate the numerous fine details of these systems.
(e) While many encouraging outcomes have been obtained in the laboratory already, efforts to scale up these technologies for industrial applications will be critical. Addressing scaling issues and assuring repeatability will be essential for these approaches’ effective inclusion in large-scale and commercial synthesis.
5. Conclusion
The synthesis of 1,2,3-triazoles using ILs has emerged as a versatile and promising method. Using ILs as a reaction medium or catalysts has enabled more environment friendly, efficient, and sustainable pathways to various 1,2,3-triazole derivatives. In particular, the interaction of ILs adds distinctive characteristics to the copper(I)-catalyzed azide–alkyne cycloaddition (CuAAC) process and enables the synthesis of a diverse range of functionalized 1,2,3-triazoles with applications in pharmaceutical chemistry, materials science, and beyond. Further, ILs have been used as a solvent, a catalytic system, and both a solvent and catalyst for the synthesis of 1,2,3-triazoles in different approaches. Many applications covered in this paper demonstrate the advantages of using ILs, such as increased reaction speeds, higher regioselectivity, and reduced environment hazards because of the reusability of the catalyst/reaction media. This review summarized various reports on the synthesis of 1,2,3-triazoles using ILs and also highlighted the results for the best catalytic systems or reaction media, which may help in the development of IL-based new catalytic systems or solvents. Overall, this review paper should support and hopefully motivate experimentalists/chemists to develop environmentally friendly, green, and sustainable approaches adhering the green chemistry principles instead of various classical chemical transformations currently used to synthesize 1,2,3-triazoles.Abbreviations
| [Bmim]BF4 | 1-Butyl-3-methylimidazolium tetrafluoroborate |
| Bu4NOAc | Tetrabutylammonium acetate |
| [Bmim]OH | 1-Butyl-3-methylimidazolium hydroxide |
| [Bmim]PF6 | Butyl-3-methylimidazolium hexafluorophosphate |
| CuAAC | Copper(I)-catalyzed azide–alkyne cycloaddition |
| CuSO4 | Copper sulfate |
| CuI | Copper iodide |
| CuCl2 | Cupric chloride |
| DMSO | Dimethyl sulfoxide |
| DCM | Dichloromethane |
| DBU | 1,8-Diazabicyclo[5.4. 0]undec-7-ene |
| DNA | Deoxyribonucleic acid |
| EAOC | Eliminative azide-olefinic cycloaddition |
| EHS | Environmental, health, and safety |
| [Hmim]TFA | 1-Methylimidazolium triflouroacetate |
| ILs | Ionic liquids |
| KOH | Potassium hydroxide |
| LCA | Life cycle assessment |
| MI | Microwave irradiation |
| NaN3 | Sodium azide |
| OAc | Acetate |
| PEG | Polyethylene glycol |
| PTSA | p-Toluenesulfonic acid |
| RNA | Ribonucleic acid |
Conflicts of interest
There are no conflicts to declare.Acknowledgements
Vijay Kumar (296/(CSIR-NET JUNE-2019)) acknowledges University Grant Commission, New Delhi, India for the Senior Research fellowship.References
- E. Kabir and M. Uzzaman, Results Chem., 2022, 4, 100606 CrossRef CAS
.
- M. Ninomiya, D. R. Garud and M. Koketsu, Coord. Chem. Rev., 2011, 255, 2968–2990 CrossRef CAS
.
- M. Fesatidou, A. Petrou and G. Athina, Curr. Pharm. Des., 2020, 26, 867–904 CrossRef CAS PubMed
.
- C. T. Walsh, Tetrahedron Lett., 2015, 56, 3075–3081 CrossRef CAS
.
- R. Dua, S. Shrivastava, S. K. Sonwane and S. K. Srivastava, Adv. Biol. Res., 2011, 5, 120–144 CAS
.
- S. Mor, R. Mohil, S. Nagoria, A. Kumar, K. Lal, D. Kumar and V. Singh, J. Heterocycl. Chem., 2017, 54, 1327–1341 CrossRef CAS
.
- B. K. Devi, S. Naraparaju, C. Soujanya and S. D. Gupta, Curr. Green Chem., 2020, 7, 314–325 CrossRef CAS
.
- P. T. Anastas and T. C. Williamson, Green Chem., 1996, 1–17 CAS
.
- P. Anastas and N. Eghbali, Chem. Soc. Rev., 2010, 39, 301–312 RSC
.
- P. Pollet, E. A. Davey, E. E. Ureña-Benavides, C. A. Eckert and C. L. Liotta, Green Chem., 2014, 16, 1034–1055 RSC
.
- F. Pena-Pereira, A. Kloskowski and J. Namieśnik, Green Chem., 2015, 17, 3687–3705 RSC
.
- R. K. Henderson, C. Jiménez-González, D. J. C. Constable, S. R. Alston, G. G. A. Inglis, G. Fisher, J. Sherwood, S. P. Binks and A. D. Curzons, Green Chem., 2011, 13, 854–862 RSC
.
- B. Zhou, J. Yang, M. Li and Y. Gu, Green Chem., 2011, 13, 2204–2211 RSC
.
- C. Rus and B. Konig, Green Chem., 2012, 14, 2969–2982 RSC
.
- P. Phukan and D. Sarma, Curr. Org. Chem., 2021, 25, 1523–1538 CrossRef CAS
.
- Z. Lei, B. Chen, Y. M. Koo and D. R. MacFarlane, Chem. Rev., 2017, 117, 6633–6635 CrossRef PubMed
.
- E. Fabre and S. S. Murshed, J. Mater. Chem., 2021, 9, 15861–15879 RSC
.
- R. A. Sheldon, Green Chem., 2005, 7, 267–278 RSC
.
- R. L. Vekariya, J. Mol. Liq., 2017, 227, 44–60 CrossRef CAS
.
- S. K. Singh and A. W. Savoy, J. Mol. Liq., 2020, 297, 112038 CrossRef CAS
.
- N. V. Plechkova and K. R. Seddon, Chem. Soc. Rev., 2008, 37, 123–150 RSC
.
- K. Ghandi, Green Sustainable Chem., 2014, 4, 44–53 CrossRef CAS
.
- P. Dominguez de Maria, Angew. Chem., Int. Ed., 2008, 47, 6960–6968 CrossRef CAS
.
- D. Zhao, M. Wu, Y. Kou and E. Min, Catal. Today, 2002, 74, 157–189 CrossRef CAS
.
- F. Zhou, Y. Liang and W. Liu, Chem. Soc. Rev., 2009, 38, 2590–2599 RSC
.
- D. D. Patel and J. M. Lee, Chem. Rec., 2012, 12, 329–355 CrossRef CAS PubMed
.
- A. J. Greer, J. Jacquemin and C. Hardacre, Molecules, 2020, 25, 5207 CrossRef CAS
.
- G. A. Tiago, I. A. Matias, A. P. Ribeiro and L. M. Martins, Molecules, 2020, 25, 5812 CrossRef CAS PubMed
.
- G. Kaur, H. Kumar and M. Singla, J. Mol. Liq., 2022, 351, 118556 CrossRef CAS
.
- I. M. Marrucho, L. C. Branco and L. P. N. Rebelo, Annu. Rev. Chem. Biomol. Eng., 2014, 5, 527–546 CrossRef CAS PubMed
.
- J. Lu, F. Yan and J. Texter, Prog. Polym. Sci., 2009, 34, 431–448 CrossRef CAS
.
- K. S. Egorova, E. G. Gordeev and V. P. Ananikov, Chem. Rev., 2017, 117, 7132–7189 CrossRef CAS PubMed
.
- D. Wei and A. Ivaska, Anal. Chim. Acta, 2008, 607, 126–135 CrossRef CAS PubMed
.
- J. F. Liu, G. B. Jiang and J. Å. Jönsson, TrAC, Trends Anal. Chem., 2005, 24, 20–27 CrossRef CAS
.
- R. Martínez-Palou, Mol. Diversity, 2010, 14, 3–25 CrossRef PubMed
.
- B. Floris, F. Sabuzi, P. Galloniand and V. Conte, Catalysts, 2017, 7, 261 CrossRef
.
- A. K. Pathak, C. Ameta, R. Ameta and P. B. Punjabi, J. Heterocycl. Chem., 2016, 53, 1697–1705 CrossRef CAS
.
- N. E. Leadbeater, H. M. Torenius and H. Tye, Comb. Chem. High Throughput Screening, 2004, 7, 511–528 CrossRef CAS
.
- A. G. Ying, X. Z. Chen, W. D. Ye, D. F. Zhang, L. Liu and J. H. Chen, Prog. Chem., 2008, 20, 1642–1650 CAS
.
- B. C. Ranu and R. Jana, J. Org. Chem., 2005, 70, 8621–8624 CrossRef CAS PubMed
.
- J. M. Lévêque and G. Cravotto, Chimia, 2006, 60, 313–313 CrossRef
.
- B. C. Ranu, R. Jana and S. S. Dey, Chem. Lett., 2004, 33, 274–275 CrossRef CAS
.
- N. Nehra, R. K. Tittal, V. D. Ghule and K. Lal, J. Mol. Struct., 2021, 1245, 131013 CrossRef CAS
.
- Naveen, R. K. Tittal, V. D. Ghule, P. Rani, K. Lal and A. Kumar, ChemistrySelect, 2020, 5, 6723–6729 CrossRef CAS
.
- S. Deswal, R. K. Tittal, P. Yadav, K. Lal, V. D. Ghule and N. Kumar, ChemistrySelect, 2019, 4, 759–764 CrossRef CAS
.
- M. Yadav, K. Lal, A. Kumar, A. Kumar and D. Kumar, J. Mol. Struct., 2022, 1261, 132867 CrossRef CAS
.
- M. K. Sharma, S. Parashar, M. Chahal, K. Lal, N. U. Pandya and H. Om, J. Mol. Struct., 2022, 1257, 132632 CrossRef CAS
.
- A. Kumar, K. Lal, V. Kumar, M. Murtaza, S. Jaglan, A. K. Paul, S. Yadav and K. Kumari, Bioorg. Chem., 2023, 133, 106388 CrossRef CAS PubMed
.
- N. Poonia, K. Lal and A. Kumar, Res. Chem. Intermed., 2021, 47, 1087–1103 CrossRef CAS
.
- K. Lal, L. Kumar, A. Kumar and A. Kumar, Curr. Top. Med. Chem., 2018, 18, 1506–1513 CrossRef CAS PubMed
.
- V. Kumar, K. Lal, A. Kumar, R. K. Tittal, M. B. Singh and P. Singh, Res. Chem. Intermed., 2023, 49, 917–937 CrossRef CAS
.
- Naveen, R. K. Tittal, V. D. Ghule, P. Yadav, K. Lal and A. Kumar, Steroids, 2020, 161, 108675 CrossRef CAS PubMed
.
- A. Kumar, Y. Rohila, V. Kumar and K. Lal, Curr. Top. Med. Chem., 2023, 23, 833–847 CrossRef CAS
.
- S. G. Agalave, S. R. Maujan and V. S. Pore, Chem. – Asian J., 2011, 6, 2696–2718 CrossRef CAS PubMed
.
- S. Kumar, S. L. Khokra and A. Yadav, Future J. Pharm. Sci., 2021, 7, 106 CrossRef PubMed
.
- N. Agouram, E. M. El Hadrami and A. Bentama, Molecules, 2021, 26, 2937 CrossRef CAS
.
- C. Ornelas, J. R. Aranzaes, E. Cloutet, S. Alves and D. Astruc, Angew. Chem., 2007, 119, 890–895 CrossRef
.
- J. Camponovo, J. Ruiz, E. Cloutet and D. Astruc, Chem. – Eur. J., 2009, 15, 2990–3002 CrossRef CAS PubMed
.
- A. R. Katritzky, Y. Song and R. Sakhuja,
et al.
, J. Polym. Sci., Part A: Polym. Chem., 2009, 47, 3748–3756 CrossRef CAS
.
- C. J. Hawker and J. M. Frechet, J. Am. Chem. Soc., 1990, 112, 7638–7647 CrossRef CAS
.
- P. Wu, M. Malkoch, J. N. Hunt, R. Vestberg, E. Kaltgrad, M. G. Finn, V. V. Fokin, K. B. Sharpless and C. J. Hawker, Chem. Commun., 2005, 46, 5775–5777 RSC
.
- P. Wu, A. K. Feldman, A. K. Nugent, C. J. Hawker, A. Scheel, B. Voit, J. Pyun, J. M. J. Fréchet, K. B. Sharpless and V. V. Fokin, Angew. Chem., 2004, 116, 4018–4022 CrossRef
.
- S. Kantheti, R. Narayan and K. V. S. N. Raju, RSC Adv., 2015, 5, 3687–3708 RSC
.
- W. H. Binder and R. Sachsenhofer, Macromol. Rapid Commun., 2008, 29, 952–981 CrossRef CAS
.
- W. Li, L. Yan, H. Zhou and W. You, Chem. Mater., 2015, 27, 6470–6476 CrossRef CAS
.
- Y. S. Yen, J. L. Hsu, J. S. Ni and J. T. Lin, Dyes Pigm., 2021, 188, 109220 CrossRef CAS
.
- L. Lan, G. Zhang, Y. Dong, L. Ying, F. Huang and Y. Cao, Polymer, 2015, 67, 40–46 CrossRef CAS
.
- N. P. Debia, M. T. Saraiva, B. S. Martins, R. Beal, P. F. Gonçalves, F. S. Rodembusch, D. Alves and D. S. Lüdtke, J. Org. Chem., 2018, 83, 1348–1357 CrossRef CAS PubMed
.
- P. Rani, K. Lal, R. Shrivastava and V. D. Ghule, J. Mol. Struct., 2020, 1203, 127437 CrossRef CAS
.
- P. Rani, K. Lal, K. S. Negi, R. Shrivastava and V. D. Ghule, Polycyclic Aromat. Compd., 2023, 43, 2376–2388 CrossRef CAS
.
- G. Singh, N. George, R. Singh, G. Singh, G. Kaur, H. Singh and J. Singh, Appl. Organomet. Chem., 2023, 37, e6897 CrossRef CAS
.
- R. Ujan, N. Arshad, F. Perveen, P. A. Channar, B. Lal, M. Hussain, Z. Hussain, S. Saeed and S. A. Shehzadi, Chem. Pap., 2021, 75, 6377–6388 CrossRef CAS
.
- T. A. Bakka, M. B. Strøm, J. H. Andersen and O. R. Gautun, Bioorg. Med. Chem., 2017, 25, 5380–5395 CrossRef CAS PubMed
.
- N. M. Grob, D. Häussinger, X. Deupi, R. Schibli, M. Behe and T. L. Mindt, J. Med. Chem., 2020, 63, 4484–4495 CrossRef CAS PubMed
.
-
T. Zheng, S. H. Rouhanifard, A. S. Jalloh and P. Wu, Click triazoles for bioconjugation, Springer, 2012, vol. 28, pp. 163–183 Search PubMed
.
-
R. Huisgen and A. Padwa, 1,3-Dipolar Cycloaddition Chemistry, Wiley, New York, 1984, vol. 1, pp. 55–92 Search PubMed
.
- C. W. Tornøe, C. Christensen and M. Meldal, J. Org. Chem., 2002, 67, 3057–3064 CrossRef PubMed
.
- V. V. Rostovtsev, L. G. Green, V. V. Fokin and K. B. Sharpless, Angew. Chem., 2002, 114, 2708–2711 CrossRef
.
- J. Lu, E. Q. Ma, Y. H. Liu, Y. M. Li, L. P. Mo and Z. H. Zhang, RSC Adv., 2015, 5, 59167–59185 RSC
.
- M. M. Masood, Polycyclic Aromat. Compd., 2023, 1–32, DOI:10.1080/10406638.2023.2247119
.
- S. Kumar, B. Lal and R. K. Tittal, Green Chem., 2024, 26, 1725–1769 RSC
.
- A. A. Ali, M. Konwar, M. Chetiaand and D. Sarma, Tetrahedron Lett., 2016, 57, 5661–5665 CrossRef CAS
.
- M. Javaherian, F. Kazemi and M. Ghaemi, Chin. Chem. Lett., 2014, 25, 1643–1647 CrossRef CAS
.
- D. Raut, K. Wankhede and V. Vaidya,
et al.
, Catal. Commun., 2009, 10, 1240–1243 CrossRef CAS
.
- J. Yan and L. Wang, Synthesis, 2010, 447–452 CAS
.
- Y. B. Zhao, Z. Y. Yan and Y. M. Liang, Tetrahedron Lett., 2006, 47, 1545–1549 CrossRef CAS
.
- P. Zhong and S. R. Guo, Chin. J. Chem., 2004, 22, 1183–1186 CrossRef CAS
.
- A. Garg, D. Borah and P. Trivedi,
et al.
, ACS Omega, 2020, 5, 29830–29837 CrossRef CAS PubMed
.
- S. V. Akolkar, A. A. Nagargoje, V. S. Krishna, D. Sriram, J. N. Sangshetti, M. Damale and B. B. Shingate, RSC Adv., 2019, 9, 22080–22091 RSC
.
- M. N. S. Rad, S. Behrouz, H. Saremi and J. Mohammadtaghi-Nezhad, J. Mol. Liq., 2020, 299, 112245 CrossRef CAS
.
- N. Singh, C. R. Chim., 2015, 18, 1257–1263 CrossRef CAS
.
- S. Narsimha, K. Sbattula, Y. N. Reddy and V. R. Nagavelli, Chem. Heterocycl. Compd., 2018, 54, 1161–1167 CrossRef CAS
.
- J. D. Patil, S. A. Patil and D. M. Pore, RSC Adv., 2015, 5, 21396–21404 RSC
.
- H. R. Babu, M. Ravinder and S. Narsimha, Indian J. Heterocycl. Chem., 2019, 29, 389–395 CAS
.
- M. Dabiri, S. K. Movahed and D. I. MaGee, Res. Chem. Intermed., 2015, 41, 3335–3347 CrossRef CAS
.
- S. Seeka, S. Narsimha, K. Battula, A. H. Shaikh, S. J. Tangeda and V. R. Nagavelli, Eur. J. Chem., 2015, 6, 482–487 CrossRef CAS
.
- A. Marra, A. Vecchi, C. Chiappe, B. Melai and A. Dondoni, J. Org. Chem., 2008, 73, 2458–2461 CrossRef CAS PubMed
.
- I. V. Seregin, L. V. Batog and N. N. Makhova, Mendeleev Commun., 2002, 12, 83–84 CrossRef
.
- E. R. Sucharitha, T. M. Krishna, R. Manchal, G. Ramesh and S. Narsimha, Bioorg. Med. Chem. Lett., 2021, 47, 128201 CrossRef
.
- H. Singh, J. Sindhu, J. M. Khurana, C. Sharma and K. R. Aneja, Eur. J. Med. Chem., 2014, 77, 145–154 CrossRef CAS PubMed
.
- O. I. Artyushin, D. V. Vorob'eva, T. P. Vasil'eva, S. N. Osipov, G. V. Röschenthaler and I. L. Odinets, Heteroat. Chem., 2008, 19, 293–300 CrossRef CAS
.
- A. Vecchi, B. Melai, A. Marra, C. Chiappe and A. Dondoni, J. Org. Chem., 2008, 73, 6437–6440 CrossRef CAS PubMed
.
- A. A. Saikia, R. N. Rao, S. Das, S. Jena, S. Rej, B. Maitin and K. Chanda, Tetrahedron Lett., 2020, 61, 152273 CrossRef CAS
.
- N. Sharma, M. Gupta, B. Chowhan and A. Frontera, J. Mol. Struct., 2021, 1224, 129029 CrossRef CAS
.
- M. Daraie and M. M. Heravi, Int. J. Biol. Macromol., 2019, 140, 939–948 CrossRef CAS PubMed
.
- S. A. Sonawane and D. M. Pore, Catal. Lett., 2022, 152, 3317–3331 CrossRef CAS
.
- P. G. Kargar, G. Bagherzade and H. Eshghi, RSC Adv., 2020, 10, 32927–32937 RSC
.
- H. G. Hosseini, E. Doustkhah, M. V. Kirillova, S. Rostamnia, G. Mahmoudi and A. M. Kirillov, Appl. Catal., 2017, 548, 96–102 CrossRef CAS
.
- M. Thakur, P. Choudhury, A. Kejriwal, A. Mukherjee and K. Biswas, Inorg. Chim. Acta, 2023, 549, 121405 CrossRef CAS
.
- E. Afzali, Z. Mirjafary, A. Akbarzadeh and H. Saeidian, Inorg. Chem. Commun., 2021, 132, 108858 CrossRef CAS
.
- A. Pawar, S. Gajare, A. Jagdale, S. Patil, W. Chandane, G. Rashinkar and S. Patil, Catal. Lett., 2022, 152, 1854–1868 CrossRef CAS
.
- A. H. Leilan, M. Babazadeh, M. Hekmati and E. Ghasemi, Inorg. Chem. Commun., 2022, 141, 109566 CrossRef
.
- K. Fehér, E. Nagy and P. Szabó,
et al.
, Appl. Organomet. Chem., 2018, 32, e4343 CrossRef
.
- A. Pourjavadi, M. Tajbakhsh, M. Farhang and S. H. Hosseini, New J. Chem., 2015, 39, 4591–4600 RSC
.
- B. Mohan, H. Kang and K. H. Park, Inorg. Chem. Commun., 2013, 35, 239–241 CrossRef CAS
.
- X. G. Liu, X. L. Zhao, Y. Zhang and J. R. Gao, Lett. Org. Chem., 2016, 13, 224–230 CrossRef CAS
.
- Y. Wang, J. Liu and C. Xia, Adv. Synth. Catal., 2011, 353, 1534–1542 CrossRef CAS
.
- M. Tavassoli, A. Landarani-Isfahani, M. Moghadam, S. Tangestaninejad, V. Mirkhani and I. Mohammadpoor-Baltork, ACS Sustainable Chem. Eng., 2016, 4, 1454–1462 CrossRef CAS
.
- M. Tavassoli, A. Landarani-Isfahani, M. Moghadam, S. Tangestaninejad, V. Mirkhani and I. Mohammadpoor-Baltork, Appl. Catal., A, 2015, 503, 186–195 CrossRef CAS
.
- M. Dabiri, S. I. Alavioon and S. K. Movahed, J. Iran. Chem. Soc., 2018, 15, 2463–2474 CrossRef
.
- B. Li, R. Hu, A. Qin and B. Z. Tang, Polym. Chem., 2020, 11, 2006–2014 RSC
.
- C. N. Dige, J. D. Patil and D. M. Pore, Catal. Lett., 2017, 147, 301–309 CrossRef
.
- P. Phukan, A. Kulshrestha, A. Kumar, S. Chakraborti, P. Chattopadhyay and D. Sarma, J. Chem. Sci., 2021, 133, 131 CrossRef CAS
.
- A. Z. Ahmady, F. Heidarizadeh and M. Keshavarz, Synth. Commun., 2013, 43, 2100–2109 CrossRef CAS
.
- S. Koguchi and K. Nakamura, Synlett, 2013, 24, 2305–2309 CrossRef CAS
.
- M. Keshavarz, B. Karami, A. Z. Ahmady, A. Ghaedi and H. Vafaei, C. R. Chim., 2014, 17, 570–576 CrossRef CAS
.
- N. Ketsomboon, R. Saeeng, K. Srisook and U. Sirion, Tetrahedron Lett., 2021, 80, 153325 CrossRef CAS
.
- M. Saquib, S. Ahamad, M. F. Khan, M. I. Khan and M. K. Hussain, Front. Pharmacol., 2023, 14, 1168566 CrossRef CAS PubMed
.
- L. Maiuolo, B. Russo, V. Algieri, M. Nardi, M. L. Di Gioia, M. A. Tallarida and A. De Nino, Tetrahedron Lett., 2019, 60, 672–674 CrossRef CAS
.
- H. Singh, J. Sindhu and J. M. Khurana, J. Iran. Chem. Soc., 2013, 10, 883–888 CrossRef CAS
.
- H. Valizadeh, M. Amiri and E. Khalili, Mol. Diversity, 2012, 16, 319–323 CrossRef CAS PubMed
.
- K. Sarkar, A. Bose and T. Pathak, Eur. J. Org. Chem., 2022, e202200189 CrossRef CAS
.
- A. De Nino, V. Algieri, M. A. Tallarida, P. Costanzo, M. Pedrón, T. Tejero, P. Merino and L. Maiuolo, Eur. J. Org. Chem., 2019, 5725–5731 CrossRef CAS
.
- Y. Jiang, K. Wu, X. Tan, D. Zhang, W. Dong, W. Li, G. Xu and W. Zhang, J. Chem. Res., 2017, 41, 631–635 CrossRef CAS
.
- A. Kayet and T. Pathak, Tetrahedron Lett., 2018, 59, 3341–3344 CrossRef CAS
.
- A. De Nino, P. Merino and V. Algieri,
et al.
, Catalysts, 2018, 8, 364 CrossRef
.
- B. Dutta, A. Garg, P. Phukan, A. Kulshrestha, A. Kumar and D. Sarma, New J. Chem., 2021, 45, 12792–12797 RSC
.
- B. Dutta, P. K. Hazarika, P. Saikia, S. Konwer, L. Borthakur and D. Sarma, New J. Chem., 2023, 47, 11389–11393 RSC
.
- H. Valizadeh, H. Gholipour and M. Mahmoodian, Synth. Commun., 2013, 43, 2801–2808 CrossRef CAS
.
- A. H. Banday and V. J. Hruby, Curr. Organocatal., 2021, 8, 223–227 CrossRef CAS
.
- H. Singh, G. Khanna and B. Nand,
et al.
, Monatsh. Chem., 2016, 147, 1215–1219 CrossRef CAS
.
| This journal is © The Royal Society of Chemistry 2024 |