Metal–organic framework (MOF) materials as polymerization catalysts: a review and recent advances
Timothy A.
Goetjen
 a,
Jian
Liu
a,
Jian
Liu
 a,
Yufang
Wu
a,
Jingyi
Sui
a,
Xuan
Zhang
a,
Yufang
Wu
a,
Jingyi
Sui
a,
Xuan
Zhang
 a,
Joseph T.
Hupp
a,
Joseph T.
Hupp
 a and
Omar K.
Farha
a and
Omar K.
Farha
 *ab
*ab
aInternational Institute of Nanotechnology and Department of Chemistry, Northwestern University, 2145 Sheridan Road, Evanston, Illinois 60208, USA. E-mail: o-farha@northwestern.edu
bDepartment of Chemical and Biological Engineering, Northwestern University, 2145 Sheridan Road, Evanston, Illinois 60208, USA
First published on 3rd August 2020
Abstract
Synthetic polymers are ubiquitous across both the industrial and consumer segments of the world economy. Catalysts enable rapid, efficient, selective, and even stereoselective, formation of desired polymers from any of a host of candidate monomers. While numerous molecular catalysts have been shown to be effective for these reactions, separation of the catalysts from reaction products is typically difficult – a potentially problematic complication that suggests instead the use of heterogeneous catalysts. Many of the most effective heterogeneous catalysts, however, comprise supported collections of reaction centres that are decidedly nonuniform in their composition, siting, and activity. Nonuniformity complicates atomic-scale evaluation of the basis for catalytic activity and thus impedes scientific hypothesis-driven understanding and development of superior catalysts. In view of the fundamental desirability of structural and chemical uniformity at the meso, nano, and even atomic scale, crystallographically well-defined, high-porosity metal–organic frameworks (MOFs) have attracted attention as model catalysts and/or catalyst-supports for a wide variety of chemical transformations. In the realm of synthetic polymers, catalyst-functionalized MOFs have been studied for reactions ranging from coordination-mediated polymerization of ethylene to visible-light initiated radical polymerizations. Nevertheless, many polymerization reactions remain to be explored – and, no doubt, will be explored, given the remarkable structural and compositional diversity of attainable MOFs. Noteworthy emerging studies include work directed toward more sophisticated catalytic schemes such as polymer templating using MOF pore architectures and tandem copolymerizations using MOF-supported reaction centres. Finally, it is appropriate to recognize that MOFs themselves are synthetic polymers – albeit, uncoventional ones.
I. Introduction
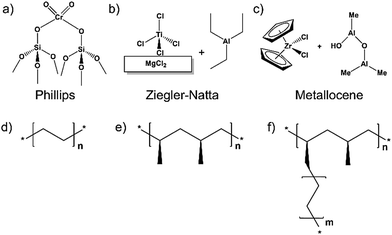
Since the creation of the first man-made polymer, nitrocellulose, almost two hundred years ago by Braconnot in 1833, researchers have been expanding the scope of polymer chemistry; meanwhile these plastics have taken on larger roles in society.1 Given their use for packaging and containment (polyethylene, PE and polypropylene, PP), synthetic rubber (polybutadiene or butadiene rubber, BR), water treatment (polyacrylamide, PAM), dental repair (polymethylmethacrylate, PMMA), textiles (nylon and other polyamides, etc.) and much more,2–7 polymers have become essential components of everyday life. Due to this prevalence, demand for efficiently produced polymers, characterized by desirable properties (for example, stereodefined polymers) is high. Among the most efficient catalysts are Phillips catalyst (Cr/SiO2),8 Ziegler–Natta catalysts (TiClx-based),9 and various metallocenes.10 These constitute the main catalysts used for the industrial-scale production of polyethylene, polypropylene, and specialty polymers, respectively (see Fig. 1 for structures).Great strides have been made in the development of homogeneous polymerization catalysts. However, industrial use is largely limited to heterogeneous catalysts, reflecting their typically higher stability under harsh reaction conditions and reflecting the comparative ease with which they can be separated from reaction products. Perhaps the most commonly known industrial-scale polymerization catalyst is the aforementioned, heterogeneous Cr-based Phillips catalyst; it accounts for ca. 50% of global PE production.8 However, due to the amorphous nature of the catalyst's SiO2 support, the precise chemical identity of the active species and the mechanism of its action are still debated.11 The absence of this kind of information, unfortunately, is inhibitory toward scientific-hypothesis-based advancement and development of Catalysis Science. Clearly desirable for fundamental studies are chemically and structurally well-defined supports and, where possible, equally well-defined catalyst active sites.
High-porosity, crystalline metal–organic frameworks (MOFs) capable of supporting catalysts are, in many respects, nearly ideal for fundamental studies of heterogeneous chemical catalysis, including catalysis of polymerization reactions. MOFs, comprised of metal ions or clusters (nodes) and multi-topic organic ligands (linkers), self-assemble to form periodically structured porous materials capable of presenting atomically identical arrays of catalysts or of grafting, binding, or growth sites for subsequently installed catalysts (Fig. 2).12 Due to the modularity of MOFs, techniques within inorganic and organic chemistry feed into their syntheses and allow for the use of a wide variety of building blocks with different properties and functionalities, making targeting specific properties facile.13 Taking advantage of their permanent porosity and inherent ease of functionalization, as well as other desirable properties, MOFs have been used for a host of applications including, but not limited to, gas storage and separations,14 drug delivery,15 sensing,16 water purification,17 and most important to this review, catalysis.18,19
 | ||
| Fig. 2 Schematic of the general formation of MOFs through assembly of inorganic (nodes) and organic (linkers) building units into a 3D structure. | ||
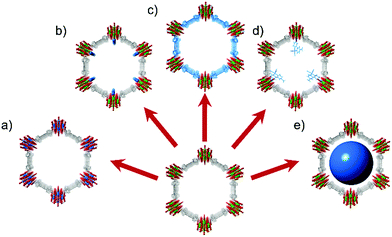
More specifically, exhibiting promising stability under desired reaction conditions, MOFs have been designed and tested for a broad range of heterogeneous catalytic reactions ranging from ozone decomposition20 to alcohol oxidation,21 and chemical warfare agent detoxification22 to olefin oligomerization.23 With an immense scope of targetable structures and an available host of potential post-synthetic modification techniques (Fig. 3),24–26 MOF-based heterogeneous catalysts have provided unique opportunities to study fundamentals of reaction mechanisms and substrate binding to well-defined catalytic sites.
In several instances, MOF-based heterogeneous catalysts comprise structurally well-defined analogues of molecular species, but ones that are simultaneously presented in high spatial density; in site-isolated, but fully reactant-accessible form; and in uniform chemical environments. Frameworks can be synthesized (or post-synthetically modified) to present pores of specific size and shape, as well as surrounding functional groups that may define or contribute to polymerization reaction selectivity.
With this in mind, MOFs represent an advance in physical and chemical tuneability of catalysts and supports over traditional catalyst materials, and further provide the opportunity for atomically precise characterization via single-crystal X-ray diffraction. Having a known structure, and the ability to make changes in the building blocks of the materials while retaining that structural framework, allow for the systematic study of MOF materials and fundamental understanding of reactions, such as polymerization. In addition, the highly tuneable porosity of MOFs and their confinement effect may lead to interesting findings on the polymerization reaction and the properties of the resulting polymers.
In the review that follows we offer an overview of the small but growing body of literature on MOF-based catalysis of polymerization reactions, highlighting studies that, in our view, represent especially useful or creative advances in this young sub-field. We conclude with a bit of speculation about what the near-term future may hold topically for research on MOF-enabled heterogeneous catalysis of polymerization reactions.
II. Polymerization catalysed by metal–organic frameworks
The discovery of the Phillips catalyst, coupled with the high reliance on polyethylene in society, served historically to focus research efforts developing and understanding of Cr-based catalysts, typically homogeneous, for polymerization of ethylene.27 For similar reasons, Ziegler–Natta catalysts featuring titanium and zirconium, have been a focus.28 While active for ethylene polymerization, Ziegler–Natta and metallocene catalysts, mainly based on first row transition metals (e.g., Ti, V, Cr, Co, and Ni), have also been used to great effect in the production of isotactic polypropylene or polymerization of conjugated dienes such as 1,3-butadiene.29,30 In addition to these coordination polymerization reactions involving gas-phase substrates, a much broader scope of polymerization reactions involving condensed-phase (solid or liquid) substrates exists, including radical-based mechanisms through techniques such as reversible addition–fragmentation chain transfer (RAFT) polymerization,31 photo-induced atom transfer radical polymerization (ATRP),32 and ring-opening polymerization (ROP).33 These mechanisms expand the substrate scope to include monomeric methacrylates, acrylamides, cyclic esters, and more, even lending themselves to co-polymerization.34 In these radical-based reactions, redox-active transition metals such as iron, copper, and titanium are used, as well as photoactive organic molecules. A host of literature regarding polymerization catalysis exists, with innumerable great advances, but to cover it all is a monumental task. This effort was recently taken up by Guironnet and co-workers in their comprehensive review of recent trends in polymerization catalysis.35 In the sections that follow on MOF-enabled catalysis of polymerization reactions we have stratified by type of substrate.Alkenes
Among the most ubiquitous polymers are polyalkenes. They find use in building and construction, car parts and electronic connections, toys, and even medical devices.4,36 Effectively tuning the properties of polymers is a synthetically nuanced endeavour, that entails targeting, for example, the formation of linear low-density polyethylene (LLDPE) over high-density polyethylene (HDPE), or isotactic polypropylene over atactic polypropylene (or vice versa). The potential promise of MOFs is to support true single-site catalysts whose compositions can be selected in a fashion that facilitates a high level of control over the molecular weight and the resulting polymer structure.Early studies by Wolczanski and Tanski in 2001 aimed to stabilize the Ziegler–Natta system within Ti-based coordination networks.37 They developed 1-dimensional (1D) analogues of previously synthesized 2D and 3D titanium aryldioxy networks, which they structurally characterized with single crystal X-ray diffraction prior to screening for polymerization activity with ethylene and propylene as substrates. Subsequently, they found that all the materials generated polydisperse linear polyethylene and atactic polypropylene, both polymers with undesirable properties. While not a promising initial study for their use as polymerization catalysts, this study broke ground for the idea of coordination network-based polymerization catalysts being structurally well-defined analogues of known molecular catalysts for application as heterogeneous catalysts.
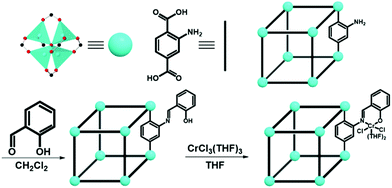
Seminal work by Li et al. from 2014 demonstrated the efficacy for ethylene polymerization of a chromium centre ligated by a phenoxy-imine, that in turn is pendant to a benzene-dicarboxylate linker. Thus, the pendant complex is essentially a MOF-heterogenized molecular catalyst.38 Starting with Zn-based isoreticular metal–organic framework-3 (IRMOF-3, [Zn4O(ATA)3]; ATA = 2-aminoterephthalate), subsequent post-synthetic modification with salicylaldehyde to induce a condensation reaction with ATA yielded a salicylidene moiety installed within the linkers of the MOF which then ligated Cr3+ (IRMOF-3-SI-Cr, see Fig. 4). With this Cr-based catalyst in hand, ethylene polymerization reactions were performed while screening different conditions, and in all cases the polyethylene produced had a relatively high polydispersity index (PDI). However, IRMOF-3-SI-Cr demonstrated substantial activity at optimal conditions (62 × 104 g mol−1 (Cr) h−1), paving the way for future studies into MOF-based ethylene polymerization catalysts which could then focus on narrowing the PDI.
Soon after, Mu and co-workers39 developed more phenoxy-imine catalysts, but rather than adding the phenoxy-imine post-synthetically, the framework was built from Zr4+ ions and a tritopic phenoxy-imine linker, producing a framework and a phenoxy-imine ligated catalyst concomitantly. This catalyst was found to be active, with the study nicely demonstrating that it is possible to use a candidate catalyst itself as a building block (node) for a porous network that is functional for the polymerization of ethylene.
Extending the scope of catalyst types beyond the MOF node itself and beyond linker appendages, Klet et al. installed (coordinatively grafted) an organozirconium catalyst, zirconium benzyl (ZrBn) on the node of a well-defined framework, Hf-NU-1000 (Hf6(μ3-OH)4(μ3-O)4(OH)4(OH2)4(TBAPy)2, TBAPy = 1,3,6,8 tetrakis(p-benzoate)pyrene).40 In contrast to earlier work, the node-grafted version of the ZrBn catalyst was able to polymerize ethylene (linear ultrahigh molecular weight PE) without first being subjected to chemical activation. As shown in Fig. 5, the MOF supported ZrBn species also polymerized 1-hexene selectively to isotactic-poly(1-hexene) (>95%) with high activity (1.4 × 102 to 2.4 × 103 g polymer (mol cat)−1 h−1).
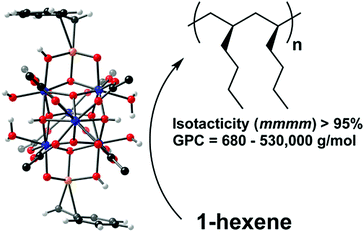 | ||
| Fig. 5 Illustration of reported 1-hexene polymerization by Hf-NU-1000-ZrBn. Atom colours: Hf (blue), Zr (orange), O (red), C (black), H (white). Reprinted with permission.40 Copyright 2015 American Chemical Society. | ||
In another novel catalyst development method, Lin et al. looked to the nodes of MOFs to serve directly as organometallic catalytic centres for olefin polymerization.41 They began by synthesizing MOF-808 (Zr-BTC; BTC = benzene tricarboxylate), a MOF consisting of a Zr6 node and tritopic benzene tricarboxylate linkers. Displacement of non-structural formate ligands first by hydroxos and then a pair of chlorides, plus charge-balancing proton, yielded the unit labelled ZrCl2-BTC in Fig. 6. Looking to conventional polymerization systems, Lin and co-workers then treated ZrCl2-BTC with an alkylaluminum reagent (MMAO-12; modified methylaluminoxane-12) which generated ZrMe-BTC. This activated MOF was shown to be competent for ethylene polymerization, yielding polyethylene of relatively low PDI and monomodal molecular weight distributions strongly indicative of a single-site or near single-site catalyst species.
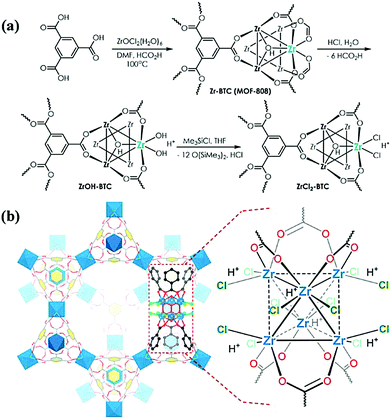 | ||
| Fig. 6 (a) ZrCl2-BTC synthetic scheme beginning with synthesis of Zr-BTC (MOF-808) and (b) proposed structure of ZrCl2-BTC. Reprinted with permission.41 Copyright 2017 American Chemical Society. | ||
Significant work on MOF-based polymerization catalysts comes from Dincă and co-workers, who have implemented the use of a cation exchange method for producing MOF nodes of compositions otherwise inaccessible de novo. They have investigated the exchange with a wide variety of transition metals (Ti, V, Cr, Fe, Co, and Ni, see Fig. 7) in Zn-based MOF MFU-4l, demonstrating the broad scope of this technique, while solely replacing tetrahedral Zn species, with the central octahedral Zn atom in the five-atom cluster node unaffected.42–45 In a study that looked at cation exchange with Ti3+/4+ and Cr2+/3+, Comito et al. demonstrated that these catalysts were highly active for ethylene polymerization yielding high molecular weight, high-density PE.42 Inspired by this, and given that vanadium catalysts are known to be sequence selective and stereoselective for light olefin polymerization, they performed the same cation exchange in MFU-4l with V4+, achieving yet another active ethylene polymerization catalyst, which more interestingly also exhibits strong selectively isotactic polypropylene production, and represents the only recent study to investigate propylene polymerization by a MOF catalyst.44 Most recently, Park et al. looked into the pre-treatment of the aforementioned Cr-based catalyst with an alkylaluminum species in order to alleviate some of the separation and processing issues between using solid catalysts and liquid co-catalyst solutions in batch reactions.45 Taking advantage of the site isolated nature of MOF nodes, they treated Cr(III)-MFU-4l with trimethylaluminum (AlMe3), and subsequently demonstrated that they achieved an order of magnitude higher activity as compared to the solvent based system. Given this significant improvement in activity through co-catalyst pre-treatment and running the catalytic reaction without solvent, Dincă and co-workers have paved the way for future studies on ethylene polymerization catalysts which will improve active site accessibility and influence further development of liquid-free polymerization systems.
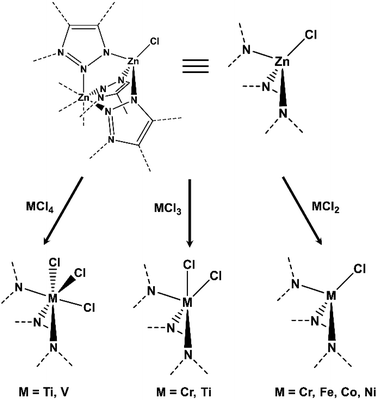 | ||
| Fig. 7 Scope of published cation exchange in MFU-4l used for polymerization catalysis.42–45 | ||
Dienes
In addition to polyalkenes, dienes play a key role in everyday life, as they are used extensively in the automotive industry and roofing due to their resistance to ozone, ultra-violet light, and heat.46 Given the additional complexity that a second double bond brings to polymerization reactions, the physical properties of diene-based polymers can be drastically changed through stereoselective control.47 Thus, dienes represent a proving ground for single-site MOF-based materials for selective polymerization reactions.Homogeneous catalytic systems based on neodymium have been shown to be efficient to perform stereospecific polymerizations of 1,3-dienes when combined with aluminum co-catalysts. Neodymium carboxylate-based MOFs were reported and used as heterogeneous catalysis for isoprene polymerization by Visseaux and co-workers for the first time.48 They reported that porous and non-porous neodymium-based MOFs, MIL-103(Nd) and MIL-81(Nd), have the ability to polymerize isoprene. Under certain conditions, the selectivity of cis-polyisoprene can reach up to 90.7%, with the catalytic activity and product selectivity found to be highly related to the porosity of materials. In their following report, these researchers synthesized another four neodymium-based MOFs using 2,6-naphthalenedicarboxylate (NDC) or formate (form) as linkers.49 Nd(2,6-NDC)(form) was found to be more active than the previous best sample of MIL-103(Nd) with comparable cis-selectivity. However, some MOF crystals were trapped inside the polyisoprene polymer; therefore Visseaux et al. chose to utilize this to their advantage in another study and prepare a luminescent polymer rubber.50 They demonstrated that MOF compounds containing two sets of different lanthanide elements (Nd3+, Eu3+/Tb3+) could be synthesized and used for luminescent polymer production, where physical mixtures of Nd(form)3 and Eu(form)3/Tb(form)3 successfully produced luminescent cis-1,4-polyisoprenes (Fig. 8), highlighting a novel synergistic use of a two MOF polymerization system.
 | ||
| Fig. 8 Polyisoprene rubbers show luminescence under the UV lamp. Reprinted with permission (open access).50 Copyright 2015 MDPI. | ||
Thus far, only one study by Dincă and co-workers has investigated a heterogeneous catalyst for 1,3-butadiene polymerization.43 Drawing from molecular catalyst literature, the authors chose a range of 3d transition metals (Ti, Cr, Fe, Co, and Ni) to study for 1,3-butadiene polymerization activity through the aforementioned cation exchange in MFU-4l. Co(II)-MFU-4l exhibited the highest stereoselectivity (>99%) at appreciable activity with negligible leaching. The stereoselectivity towards 1,4-cis-polymerization is desirable as the resulting elastomer polymer has better wear resistance and impact resilience. Further structural characterization of Co(II)-MFU-4l in tandem with molecular analogues by X-ray absorption spectroscopy (XAS) provided evidence of a single active site within the framework that is expected given the catalytic activity (Fig. 9).
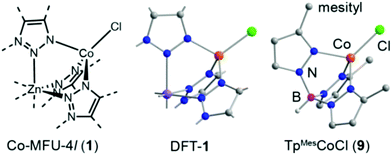 | ||
| Fig. 9 Proposed structure of Co-MFU-4l compared to DFT optimized and molecular analogue structures. Atom colours: Zn (purple), Co (orange), Cl (green), N (blue), C (grey), B (magenta). Reprinted with permission.43 Copyright 2017 American Chemical Society. | ||
While demonstrating the effectiveness and versatility of MOF catalysts, these diene-based studies currently only look at 1,3-butadiene or isoprene polymerization. Therefore, there exists an expansive experiment space for future studies into stereoselective polymerization of other more complex conjugated dienes with heterogeneous MOF catalysts, including those with structures that influence stereoselectivity through contained MOF pore architectures.
Acrylamides
Polyacrylamides see use in various industries including, but not limited to, the manufacturing of paper, oil and mineral extraction, and the treatment of water.7 Therefore, the efficient production of polyacrylamides with high molecular weight, to minimize adverse biological effects, is necessary. The typical radical-based mechanism of polyacrylamide production, reversible addition–fragmentation chain transfer (RAFT), demonstrates the first deviation of MOFs from the typical coordination polymerization reaction, adding an additional level of complexity to these systems. Thus, acrylamides represent another new avenue of research in MOF-based catalysts, where the advantages of MOFs can be utilized to achieve active-site uniformity and stabilization of enzyme-inspired structures.Simply by combining glycine and MIL-53(Fe), Gly/MIL-53(Fe) was synthesized and reported by Fu et al. for polymerization of N,N-dimethylacrylamide (DMA) via RAFT (Fig. 10).51 Structurally inspired by the enzyme peroxidase, Gly/MIL-53(Fe) functioned in a similar manner by degrading hydrogen peroxide to generate hydroxyl radicals, which serve as initiators for RAFT polymerization, thereby controlling polymerization. More interestingly, Gly/MIL-53(Fe) demonstrated an extended substrate scope including hydrophilic N,N-dimethylacrylamide (DMA), N-hydroxyethyl acrylamide (NHEA), and 2-(dimethylamino)ethyl methacrylate (DMAEMA), as well as hydrophobic methyl acrylate (MA). Prepared by a simple strategy, peroxidase-like Gly/MIL-53(Fe) exhibited good stability and high catalytic activity for polymerization of various monomers, displaying the great potential of designing MOFs with structures or as composites inspired by enzymes for controlled polymerization reactions.
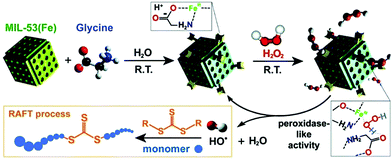 | ||
| Fig. 10 Proposed mechanisms for the formation of Gly/MIL-53(Fe) and the initiation of RAFT polymerization by hydroxyl radicals. Reproduced with permission.51 Copyright 2018 The Royal Society of Chemistry. | ||
In addition, Reyhani and co-workers introduced an Fe(II) MOF-Fenton-RAFT polymerization system to produce PDMA and poly(2-hydroxyethyl acrylate) (PHEA) in the presence of air without adding any enzyme or reducing agents to aid in deoxygenation.52 They provide good guidance for the development of deoxygenation technology in subsequent radical polymerization reactions.
Methacrylates
Known for their common use in the dental industry, polymethacrylates are desired in the form of lightweight materials with high impact strength.5 With the development of MOF-based catalysts for polymerization of methacrylates, the scope of polymerization reactions catalysed by MOFs expands further to include atom transfer radical polymerization (ATRP) reactions, common methods of polymethacrylate synthesis. Subsequently, methacrylates present the additional challenge to MOF-based catalysts of including a light-mediated process. However, MOFs rise to this challenge with their modularity, allowing for the use of photoactive organic molecules or redox active metal ions as linkers or nodes.Utilizing photoactive nodes is an effective approach to form visible-light-responsive MOFs. Nguyen et al. used spatially isolated titanium clusters as the MOF nodes in the synthesis of MOF-901 and MOF-902.53,54 With the linker elongating from one phenyl ring to two phenyl rings, MOF-902 has an isoreticular structure to MOF-901, however with an elongated conjugation system (Fig. 11). The researchers reported red-shifted visible light adsorption, compared to MOF-901, due to the increased conjugation present in the structural backbone of MOF-902. Both MOF-901 and MOF-902 exhibited high catalytic activity for photopolymerization of methyl methacrylate (MMA), outperforming commercial P-25 TiO2 in terms of PMMA molecular weight, with MOF-902 investigated using various monomers other than MMA.
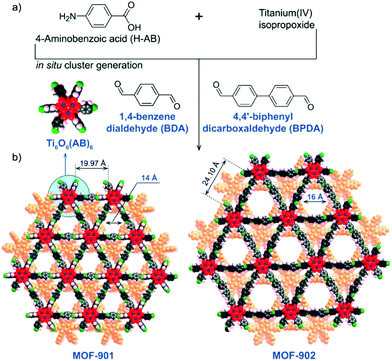 | ||
| Fig. 11 (a) Synthesis procedure and (b) crystal structures of MOF-901 and MOF-902. Atom colours: Ti (blue), C (black), O (red), N (green), H (pink), and second layer (orange). Reprinted with permission.54 Copyright 2017 American Chemical Society. | ||
Another node-based photoactive MOF was employed by Schmidt and co-workers for controllable photopolymerization of vinylpyridines and methacrylates under visible light.55 The researchers found that the nucleophilic N-containing monomers binding with Cu(II) ions of the node could enable the MOF to harvest visible light rather than ultraviolet (UV) light, and that intermittent illumination showed enhanced catalysis under irradiation. This MOF system demonstrated that multiple MOF systems can be visual light responsive, and in addition have temporal polymerization control through intermittent light exposure.
Rather than using a photoactive node in MOFs, incorporation of the chromophore components into ligands, such as anthracene, is another way to synthesize photoactive MOFs. A few examples include Zr-based NNU-28,56 In-based NNU-32,57 and Zn-based NNU-35.58 These MOFs mediated visible-light-induced atom transfer and radical formation in the photopolymerization of i-butyl methacrylate (i-BMA), n-butyl methacrylate (n-BMA) and MMA.
Seeking a new technique beyond visible light-mediated polymerization, MOF-907(Fe), a nha net MOF based on Fe3O(–CO2)6 clusters and 4,4′,4′′-benzene-1,3,5-triyl-tris(benzoic acid) (H3BTB) and NDC linkers, was synthesized by Nguyen et al. for use as a catalyst in the microwave-assisted radical polymerization of MMA.59 MOF-907(Fe) showed excellent catalytic activity toward microwave-assisted MMA polymerization, with high PMMA yield of 98%, high molecular weight of 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 680 g mol−1 and low polydispersity of 1.23 obtained within 30 min. This study emphasizes the tuneability of MOF building units for targeted applications, including the unique microwave-assisted polymerization method.
680 g mol−1 and low polydispersity of 1.23 obtained within 30 min. This study emphasizes the tuneability of MOF building units for targeted applications, including the unique microwave-assisted polymerization method.
In an effort to learn from nature, Jiang and co-workers fabricated a DhHP-6(Fe)@ZIF-8 composite by embedding a peroxidase-like iron porphyrin moiety, Deuterohemin-β-Ala-His-Thr-Val-Glu-Lys (DhHP-6), onto the surface of zeolitic imidazolate framework-8 (ZIF-8).60 The resulting composite showed great catalytic ability for controlled ATRP of poly(ethylene glycol) methyl ether methacrylate (PEGMA500). Compared to that obtained using free DhHP-6, PEGMA500 with higher monomer conversion (76.1%) and molecular weight (45![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 900 g mol−1) was produced via ATRP catalysed by DhHP-6(Fe)@ZIF-8. With almost no iron residues in the product (less than 3% of DhHP-6 released within 100 hours), this enzyme-inspired MOF composite provides a good model for developing efficient enzyme-inspired heterogeneous catalysts for polymerization.
900 g mol−1) was produced via ATRP catalysed by DhHP-6(Fe)@ZIF-8. With almost no iron residues in the product (less than 3% of DhHP-6 released within 100 hours), this enzyme-inspired MOF composite provides a good model for developing efficient enzyme-inspired heterogeneous catalysts for polymerization.
Cyclic esters
As environmental concerns grow for developing sustainable pathways to polymers, research into the efficient synthesis of degradable and recyclable polymers has expanded.6 Due to their extent of natural occurrence and bio renewable sourcing, lactones have gathered increased interest for this use. However, this adds yet another method of polymerization into the mix, as lactones are cyclic esters that require ring opening prior to polymerizing. Ring-opening polymerization (ROP) catalysts have seen the use of amorphous supports for adapting homogeneous systems, however MOFs bring the added bonus of structural uniformity and crystallinity. This leads to the development of well-defined systems with controlled reactivity that are amenable to mechanistic investigation.Wu et al. showed that a titanium alkoxide based-MOF could be synthesized having the formula of [Ti2L3(LH)2]∞ (where LH2 = 1,4-butanediol).61 They found that this MOF is soluble in organic solvents and pointed out that a discrete molecular titanium alkoxide containing chelating and/or bridging 1,4-butoxide ligands is the initiator in ROP of ε-caprolactone, L/rac-lactide, and other cyclic esters, which was further confirmed by identical catalytic performance observed when using a molecular analogue of the MOF. Verpoort and co-workers developed a series of stable MOFs for ROP of L-lactide, which are MDABCO (M = Co, Ni, Cu, and Zn; DABCO = 1,4-diazabicyclo[2.2.2]octane).62 They found the introduction of lactic acid results in a higher activity polymerization system, which comes from different working mechanisms with and without lactic acid addition. The use of lactic acid led to the formation of intermediate product zinc L-lactate to enhance the yield of isotactic polylactides (PLAs). In addition, this intermediate product came from the limited chemical stability of ZnDABCO under polymerization conditions with lactic acid. Compared to related homogeneous examples, ZnDABCO is demonstrated to afford highly isotactic polylactide, which would be ascribed to the unique structural properties of MOFs.
III. Conclusions and outlook
As catalytic materials for polymerization reactions, metal–organic frameworks have shown significant promise despite being new to the sub-field. From coordinative ethylene polymerization to microwave-assisted radical polymerization of MMA, MOF catalysts have been implemented for a wide range of monomers and methods of polymerization. Taking advantage of their inherent porosity and synthetic capabilities to form structurally well-defined single-site catalysts, researchers have developed these MOF-based catalysts to great effect in polymerization reactions. From supporting organometallic moieties as heterogeneous analogues of molecular catalysts to employing photo responsive catalysts for light-mediated polymerization reactions, MOFs are paving the way for systematic studies of fundamental polymerization reactions and development of next-generation materials. These MOF polymerization catalyst materials have yet to reach commercialization and implementation in industrial processes, likely due to their youth within this sub-field, however, that is not to say that MOF materials have not in general reached commercialization.63 Considering recent commercial success with MOF materials, the future looks bright for their eventual implementation as they compete with current industrial catalysts in activity and selectivity.Looking forward, the design space and potential for future research is broad, owing to the tuneability and versatility of metal–organic framework catalysts, and suggested by the wide scope of polymerization reactions already demonstrated. One of the areas just starting to be explored is the templating of polymers using the MOF pore architecture. Rivera-Torrente et al. have touched on this topic; they showed that a structurally similar pair of MOFs, Cr-MIL-100 and Cr-MIL-101, can be activated for ethylene polymerization, but with distinctively different morphological outcomes: fibres for Cr-MIL-100 and beads for Cr-MIL-101.64 Notably, the active sites in the two MOFs are identical; differences are present only several angstroms removed from the active sites. The researchers were able to show that the formation of beads is tied directly to the ability of the growing polymer to physically fracture the framework-defined pores of Cr-MIL-101, but not those of Cr-MIL-100. This remarkable, but unexpected, morphological differentiation suggests the topic is ripe for systematic investigation – perhaps culminating in precise framework design rules for engendering desired morphological or chemical outcomes. As polymer physical properties are highly tuneable, having a geometrically constrained catalyst that controls them and can produce the desired morphology is ideal for developing polymers for specific applications. We envision an exciting future here, as the surface has only just been scratched in terms of demonstrating the ability of frameworks to template or otherwise define polymer morphology and other physical properties. With this in mind, future studies should include the variation of physical properties of the MOF catalyst and identification of the subsequent effects on polymer properties.
Another emerging area is the synthesis of copolymers. Owing to their versatility, one can envision MOF catalysts containing multiple distinct catalytic sites for catalysing multiple types of polymerization reactions. There have been some initial efforts into MOF catalysed copolymerization of olefins, rod-coil conjugated grafts, and poly(hydroxyalkanoates) (PHAs).42,65,66 While the olefin (ethylene/propylene) and rod-coil conjugation graft (poly(3-hexyl thiophene), P3HT and PMMA) copolymerizations both utilize single technique polymerizations at one active site, they demonstrate the first endeavors towards copolymerization within MOFs and provide promise for more complex tandem copolymerization systems.42,65 That being said, an interesting terpolymerization system was developed by Padmanaban et al. for the synthesis of industrially relevant PHAs utilizing CO2, propylene oxide (PO), and β-butyrolactone (BBL) as monomers.66 With this initial study, MOFs have shown promise for the production of complex specialty polymers, requiring coordination and ring opening polymerization reactions for the synthesis of a copolymer from a three monomer system.
As structural understanding of MOFs continues to increase and synthetic techniques become more sophisticated, we will be able to better target ideal MOF structures and identify active species. Ideally this will drive a deeper mechanistic understanding of polymerization reactions with comparison to traditional polymerization catalyst mechanisms in an effort to identify how the MOF catalyst influences potential mechanistic changes. Some initial experimental mechanistic studies have been conducted;42,43,52 however, most efforts remain at the proposed plausible mechanism stage,55–58,62 with one study comparing catalyst selectivity to DFT calculations.40 Therefore, this represents a need for the future of this field to make strides into identifying mechanisms with MOF catalysts, with comparison to traditional heterogeneous catalyst mechanisms, and using that knowledge to feed into the development of next-generation laboratory and industrial catalysts, and perhaps even MOF-based industrial catalysts. With this in mind, and the inspiring initial efforts into polymer morphology templating and copolymerization reactions, MOF-based catalysts have a bright future for contributing to the field of heterogeneous polymerization catalysis in unique ways.
Conflicts of interest
There are no conflicts to declare.Acknowledgements
This work was supported as part of the Inorganometallic Catalyst Design Center, an EFRC funded by the DOE, Office of Science, Basic Energy Sciences (DE-SC0012702).Notes and references
- L. A. Utracki, Polym. Eng. Sci., 1995, 35, 2–17 CrossRef CAS.
- A. L. Andrady and M. A. Neal, Philos. Trans. R. Soc., B, 2009, 364, 1977–1984 CrossRef CAS PubMed.
- P. Galli and G. Vecellio, Prog. Polym. Sci., 2001, 26, 1287–1336 CrossRef CAS.
- A. Vaughan, D. S. Davis and J. R. Hagadorn, Polymer Science: A Comprehensive Reference, 2012, 657–672, DOI:10.1016/b978-0-444-53349-4.00080-7.
- M. Hassan, M. Asghar, S. U. Din and M. S. Zafar, Mater. Biomed. Eng., 2019, 273–308, DOI:10.1016/b978-0-12-816874-5.00008-6.
- Q. Song, J. Zhao, G. Zhang, F. Peruch and S. Carlotti, Polym. J., 2019, 52, 3–11 CrossRef.
- M. Sun, M.-X. Qiao, J. Wang and L.-F. Zhai, ACS Sustainable Chem. Eng., 2017, 5, 7832–7839 CrossRef CAS.
- M. P. McDaniel, A Review of the Phillips Supported Chromium Catalyst and Its Commercial Use for Ethylene Polymerization, Elsevier Inc, 1 edn, 2010 Search PubMed.
- T. E. Nowlin, R. I. Mink and Y. V. Kissin, in Handbook of Transition Metal Polymerization Catalysts, John Wiley & Sons, Ltd, 2010, ch. 6, pp. 131–155 DOI:10.1002/9780470504437.
- W. Kaminsky, Rend. Fis. Acc. Lincei, 2017, 28, 87–95 CrossRef.
- E. Groppo, C. Prestipino, F. Cesano, F. Bonino, S. Bordiga, C. Lamberti, P. C. Thüne, J. W. Niemantsverdriet and A. Zecchina, J. Catal., 2005, 230, 98–108 CrossRef CAS.
- H. C. Zhou, J. R. Long and O. M. Yaghi, Chem. Rev., 2012, 112, 673–674 CrossRef CAS PubMed.
- A. J. Howarth, Y. Liu, P. Li, Z. Li, T. C. Wang, J. T. Hupp and O. K. Farha, Nat. Rev. Mater., 2016, 1, 1–15 Search PubMed.
- H. Li, K. Wang, Y. Sun, C. T. Lollar, J. Li and H. C. Zhou, Mater. Today, 2018, 21, 108–121 CrossRef CAS.
- I. Abánades Lázaro and R. S. Forgan, Coord. Chem. Rev., 2019, 380, 230–259 CrossRef.
- Y. Zhang, S. Yuan, G. Day, X. Wang, X. Yang and H. C. Zhou, Coord. Chem. Rev., 2018, 354, 28–45 CrossRef CAS.
- R. J. Drout, L. Robison, Z. Chen, T. Islamoglu and O. K. Farha, Trends Chem., 2019, 1, 304–317 CrossRef.
- A. Dhakshinamoorthy, Z. Li and H. Garcia, Chem. Soc. Rev., 2018, 47, 8134–8172 RSC.
- M. C. Wasson, C. T. Buru, Z. Chen, T. Islamoglu and O. K. Farha, Appl. Catal., A, 2019, 586, 117214 CrossRef CAS.
- H. Wang, P. Rassu, X. Wang, H. Li, X. Wang, X. Wang, X. Feng, A. Yin, P. Li, X. Jin, S. L. Chen, X. Ma and B. Wang, Angew. Chem., Int. Ed., 2018, 57, 16416–16420 CrossRef CAS PubMed.
- X. Wang, X. Zhang, P. Li, K. I. Otake, Y. Cui, J. Lyu, M. D. Krzyaniak, Y. Zhang, Z. Li, J. Liu, C. T. Buru, T. Islamoglu, M. R. Wasielewski, Z. Li and O. K. Farha, J. Am. Chem. Soc., 2020, 141, 8306–8314 Search PubMed.
- Y. Liu, A. J. Howarth, N. A. Vermeulen, S. Y. Moon, J. T. Hupp and O. K. Farha, Coord. Chem. Rev., 2017, 346, 101–111 CrossRef CAS.
- T. A. Goetjen, X. Zhang, J. Liu, J. T. Hupp and O. K. Farha, ACS Sustainable Chem. Eng., 2019, 7, 2553–2557 CrossRef CAS.
- S. Yuan, Y. P. Chen, J. Qin, W. Lu, X. Wang, Q. Zhang, M. Bosch, T. F. Liu, X. Lian and H. C. Zhou, Angew. Chem., Int. Ed., 2015, 54, 14696–14700 CrossRef CAS PubMed.
- T. Islamoglu, S. Goswami, Z. Li, A. J. Howarth, O. K. Farha and J. T. Hupp, Acc. Chem. Res., 2017, 50, 805–813 CrossRef CAS PubMed.
- S. Liang, X. L. Wu, J. Xiong, M. H. Zong and W. Y. Lou, Coord. Chem. Rev., 2020, 406, 213149 CrossRef CAS.
- C. Bariashir, C. Huang, G. A. Solan and W. H. Sun, Coord. Chem. Rev., 2019, 385, 208–229 CrossRef CAS.
- H. G. Alt and A. Köppl, Chem. Rev., 2000, 100, 1205–1221. CrossRef CAS PubMed.
- A. Zambelli, I. Sessa, F. Grisi, R. Fusco and P. Accomazzi, Macromol. Rapid Commun., 2001, 22, 297–310 CrossRef CAS.
- B. Cornils, W. A. Herrmann, M. Beller and R. Paciello, Applied homogeneous catalysis with organometallic compounds: A comprehensive handbook in three volumes, 3 edn, 2012 Search PubMed.
- E. V. Chernikova and E. V. Sivtsov, J. Polym. Sci., Part B: Polym. Phys., 2017, 59, 117–146 CAS.
- S. Dadashi-Silab, M. Atilla Tasdelen and Y. Yagci, J. Polym. Sci., Part A: Polym. Chem., 2014, 52, 2878–2888 CrossRef CAS.
- O. Nuyken and S. D. Pask, Polymers, 2013, 5, 361–403 CrossRef.
- S. Klaus, M. W. Lehenmeier, C. E. Anderson and B. Rieger, Coord. Chem. Rev., 2011, 255, 1460–1479 CrossRef CAS.
- D. J. Walsh, M. G. Hyatt, S. A. Miller and D. Guironnet, ACS Catal., 2019, 9, 11153–11188 CrossRef CAS.
- H. Makio, H. Terao, A. Iwashita and T. Fujita, Chem. Rev., 2011, 111, 2363–2449 CrossRef CAS PubMed.
- J. M. Tanski and P. T. Wolczanski, Inorg. Chem., 2001, 40, 2026–2033 CrossRef CAS PubMed.
- B. Liu, S. Jie, Z. Bu and B. G. Li, J. Mol. Catal. A: Chem., 2014, 387, 63–68 CrossRef CAS.
- H. Li, B. Xu, J. He, X. Liu, W. Gao and Y. Mu, Chem. Commun., 2015, 51, 16703–16706 RSC.
- R. C. Klet, S. Tussupbayev, J. Borycz, J. R. Gallagher, M. M. Stalzer, J. T. Miller, L. Gagliardi, J. T. Hupp, T. J. Marks, C. J. Cramer, M. Delferro and O. K. Farha, J. Am. Chem. Soc., 2015, 137, 15680–15683 CrossRef CAS PubMed.
- P. Ji, J. B. Solomon, Z. Lin, A. Johnson, R. F. Jordan and W. Lin, J. Am. Chem. Soc., 2017, 139, 11325–11328 CrossRef CAS PubMed.
- R. J. Comito, K. J. Fritzsching, B. J. Sundell, K. Schmidt-Rohr and M. Dincă, J. Am. Chem. Soc., 2016, 138, 10232–10237 CrossRef CAS PubMed.
- R. J. Dubey, R. J. Comito, Z. Wu, G. Zhang, A. J. Rieth, C. H. Hendon, J. T. Miller and M. Dincă, J. Am. Chem. Soc., 2017, 139, 12664–12669 CrossRef CAS PubMed.
- R. J. Comito, Z. Wu, G. Zhang, J. A. Lawrence, M. D. Korzyński, J. A. Kehl, J. T. Miller and M. Dincă, Angew. Chem., Int. Ed., 2018, 57, 8135–8139 CrossRef CAS PubMed.
- H. D. Park, R. J. Comito, Z. Wu, G. Zhang, N. Ricke, C. Sun, T. Van Voorhis, J. T. Miller, Y. Román-Leshkov and M. Dincă, ACS Catal., 2020, 10, 3864–3870 CrossRef CAS.
- P. S. Ravishankar, Rubber Chem. Technol., 2012, 85, 327–349 CrossRef CAS.
- E. M. Lauretti and B. Miani, Rubber World, 1994, 210, 34 CAS.
- M. J. Vitorino, T. Devic, M. Tromp, G. Férey and M. Visseaux, Macromol. Chem. Phys., 2009, 210, 1923–1932 CrossRef CAS.
- I. Rodrigues, I. Mihalcea, C. Volkringer, T. Loiseau and M. Visseaux, Inorg. Chem., 2012, 51, 483–490 CrossRef CAS PubMed.
- S. Russell, T. Loiseau, C. Volkringer and M. Visseaux, Inorganics, 2015, 3, 467–481 CrossRef CAS.
- Q. Fu, H. Ranji-Burachaloo, M. Liu, T. G. McKenzie, S. Tan, A. Reyhani, M. D. Nothling, D. E. Dunstan and G. G. Qiao, Polym. Chem., 2018, 9, 4448–4454 RSC.
- A. Reyhani, H. Ranji-Burachaloo, T. G. McKenzie, Q. Fu and G. G. Qiao, Macromolecules, 2019, 52, 3278–3287 CrossRef CAS.
- H. L. Nguyen, F. Gándara, H. Furukawa, T. L. H. Doan, K. E. Cordova and O. M. Yaghi, J. Am. Chem. Soc., 2016, 138, 4330–4333 CrossRef CAS PubMed.
- H. L. Nguyen, T. T. Vu, D. Le, T. L. H. Doan, V. Q. Nguyen and N. T. S. Phan, ACS Catal., 2017, 7, 338–342 CrossRef CAS.
- H. C. Lee, M. Fantin, M. Antonietti, K. Matyjaszewski and B. V. K. J. Schmidt, Chem. Mater., 2017, 29, 9445–9455 CrossRef CAS.
- H. Xing, D. Chen, X. Li, Y. Liu, C. Wang and Z. Su, RSC Adv., 2016, 6, 66444–66450 RSC.
- X. Li, D. Chen, Y. Liu, Z. Yu, Q. Xia, H. Xing and W. Sun, CrystEngComm, 2016, 18, 3696–3702 RSC.
- Y. Liu, D. Chen, X. Li, Z. Yu, Q. Xia, D. Liang and H. Xing, Green Chem., 2016, 18, 1475–1481 RSC.
- H. L. Nguyen, T. T. Vu, D. K. Nguyen, C. A. Trickett, T. L. H. Doan, C. S. Diercks, V. Q. Nguyen and K. E. Cordova, Commun. Chem., 2018, 1, 1–7 CrossRef CAS.
- W. Jiang, X. Wang, J. Chen, Y. Liu, H. Han, Y. Ding, Q. Li and J. Tang, ACS Appl. Mater. Interfaces, 2017, 9, 26948–26957 CrossRef CAS PubMed.
- C. J. Chuck, M. G. Davidson, M. D. Jones, G. Kociok-Kohn, M. D. Lunn and S. Wu, Inorg. Chem., 2006, 45, 6595–6597 CrossRef CAS PubMed.
- Z. Luo, S. Chaemchuen, K. Zhou, A. A. Gonzalez and F. Verpoort, Appl. Catal., A, 2017, 546, 15–21 CrossRef CAS.
- Nat. Chem., 2016, 8, 987 Search PubMed.
- M. Rivera-Torrente, P. D. Pletcher, M. K. Jongkind, N. Nikolopoulos and B. M. Weckhuysen, ACS Catal., 2019, 9, 3059–3069 CrossRef CAS.
- H. M. Tran, L.-T. T. Nguyen, T. H. Nguyen, H. L. Nguyen, N. T. S. Phan, G. Zhang, T. Yokozawa, H. L. Tran, P. T. Mai and H. T. Nguyen, Eur. Polym. J., 2019, 116, 190–200 CrossRef CAS.
- S. Padmanaban, S. Dharmalingam and S. Yoon, Catalysts, 2018, 8, 1–10 CrossRef.
| This journal is © The Royal Society of Chemistry 2020 |