4-Chloro-2H-chromene-3-carbaldehyde: a valid scaffold in organic synthesis and future molecules for healthcare and energy
Guru Prasad
Rameshbabu
and
Sabbasani
Rajasekhara Reddy
 *
*
Department of Chemistry, School of Advanced Sciences, Vellore Institute of Technology, Katpadi, Vellore, Tamil Nadu 632014, India. E-mail: sekharareddyiitm@gmail.com; sekharareddy@vit.ac.in; Tel: +919884968303
First published on 17th December 2025
Abstract
In the past decade, 4-chloro-2H-chromene-3-carbaldehyde scaffolds (CCCS) have emerged as versatile precursors in modern organic synthesis. This review critically analyzes recent advancements in the chemical reactivity and synthetic accessibility of CCCS. We evaluate their pivotal role in developing potent agents against inflammation, cancer, and microbial resistance, while also addressing applications in energy sectors. By identifying the most efficient derivatives within each series, this work provides a strategic framework for leveraging CCCS in future pharmaceutical and materials science research.
1. Introduction
In contemporary organic synthesis, 4-chloro-2H-chromene-3-carbaldehyde scaffolds (CCCS) function as crucial scaffolds for the synthesis of organic and bioactive molecules, owing to their benzopyran ring, β-chlorine, and carbonyl group. These flexible molecules facilitate the construction of diverse fused heterocyclic compounds, which are among the most structurally and functionally significant compounds.1–3 Their rigid, electronically tunable frameworks confer distinct steric and electronic properties, contributing to their widespread utility across pharmaceuticals,4 agrochemicals,5 and materials science.6 The prominence of these motifs in natural products and drugs underscores their biological relevance, with notable activity profiles in anticancer,7 anti-inflammatory,8 and neuroactive domains.9,10 Within this broad class of heterocyclic compounds, benzopyran molecules represent notable scaffolds and are extensively studied for their pharmacological and synthetic importance. Characterized by a benzene ring fused to a pyran moiety, benzopyrans occur naturally in flavonoids,11 coumarins,12,13 and isoflavones,14 which have been associated with antioxidant,15 cytotoxic,16 and enzyme-inhibitory activities.17 Their structural modularity permits extensive derivatization at multiple positions on the core, enabling structure–activity relationship (SAR) studies and analogue development.18,19Synthetic approaches for benzopyrans include substitution,20,21 cyclization22,23 and transition-metal-catalyzed oxidative addition,24 offering access to diverse substitution patterns and stereochemistry.25–27 In recent years, β-chlorovinylaldehydes have also emerged as versatile synthetic intermediates owing to their dual electrophilic nature and their ability to participate in diverse bond-forming reactions.
The conjugation between the aldehyde and β-chlorovinyl units enhances both reactivity and selectivity in condensation reactions such as the Claisen–Schmidt28 and Wittig reactions.29 Furthermore, the reactivity trend of the moiety enables substitution,30 metal-catalyzed cross-coupling,31 and annulation reactions,32 making them valuable building blocks for the synthesis of complex heterocyclic scaffolds and functionalized alkenes.33,34
Despite their distinct chemistries, both benzopyrans and β-chlorovinylaldehydes intersect in the context of heterocycle synthesis and drug-relevant small molecule development. Additionally, CCCS would be very much useful in building polycyclic heteroaromatics (PHAs) and polycyclic aromatic hydrocarbons (PAHs), which demonstrate exceptional electronic properties, including improved charge mobility and adjustable optical characteristics, making them vital in organic electronics such as OLEDs and OPVs.35–38 The presence of PHAs in natural products and their additional structural diversity enable applications in sensing, catalysis, and medicinal chemistry.39–41
This review aims to provide a comprehensive overview of recent advances in the synthesis, reactivity, and application of benzopyran frameworks, with particular emphasis on strategies involving β-chlorovinylaldehyde scaffolds, namely CCCS (Fig. 1).
2. Synthesis of CCCS
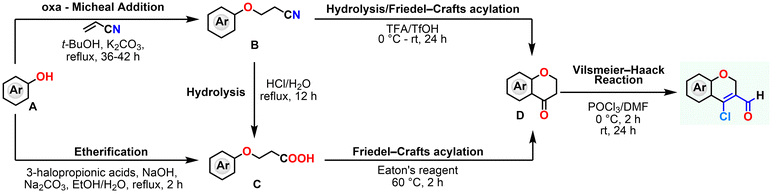
The synthesis of CCCS has been achieved through a multi-step pathway starting from readily available phenol derivatives (Scheme 1).42 In the initial step, phenol (A) undergoes nucleophilic substitution with acrylonitrile to furnish 3-phenoxypropanenitrile (B), which is then subjected to acid hydrolysis using hydrochloric acid to yield the corresponding carboxylic acid (C).43 Cyclization of this intermediate using Eaton's reagent facilitates intramolecular Friedel–Crafts acylation, affording chroman-4-one (D).42 Subsequently, the introduction of an aldehyde group at the 3-position is accomplished via a Vilsmeier–Haack formylation, yielding the CCCS.Several alternative synthetic strategies have also been reported. One approach utilizes sodium metal for the ether formation step,44 followed by cyclization employing a strong Brønsted acid such as triflic acid (TfOH) with trifluoroacetic acid (TFA),42 prior to Vilsmeier–Haack formylation. Another method involves direct conversion of phenols to 3-phenoxypropanoic acid (C) using 3-halopropionic acids,45 followed by subsequent cyclization and functionalization steps to yield the CCCS. These variations offer alternative routes depending on substrate tolerance, scalability, and reagent accessibility.
3. Applications
3.1 Synthetic applications
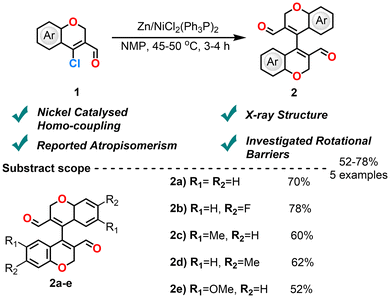
CCCS serve as synthetically valuable intermediates, combining the reactivity profiles of both 2H-chromenes and β-chlorovinylaldehydes. The chromene core enables diverse transformations such as oxidation,46,47 ring-opening,48 and metal-catalyzed couplings.49,50 Meanwhile, the β-chlorovinylaldehyde moiety facilitates nucleophilic vinylic substitution,51 aldol condensation,52 Wittig reaction53 and heterocycle formation.54 This dual functionality makes CCCS versatile scaffolds for the construction of structurally diverse and functionally rich compounds in organic synthesis.Balasubramanian et al. (2019) demonstrated the homo-coupling reactions of CCCS via a Zn/Ni catalytic system (Scheme 2).3 The in situ generation of a Ni(0) complex achieved (PPh3)2NiCl2 using (PPh3)2Cl2 with activated zinc, which enabled a more efficient dimerization than CuI, Ni(PPh3)2Cl2, and Pd(PPh3)4 catalytic systems. Notably, the product 4,4′-bichromene-3,3′-dicarbaldehyde exhibited atropisomerism due to restricted rotation and steric hindrance between the chromene units, as confirmed by single-crystal X-ray diffraction and detailed NMR analysis. The bichromene derivative was post-synthetically modified at the aldehyde to yield alcohol, acid, oxime, and nitrile analogues. All post-modified derivatives showed atropisomerism both in HPLC and NMR, except the nitrile group, which showed atropisomerism in HPLC but not in NMR, likely due to accidental equivalence in the 1H NMR. Additionally, the C-5 position of the chromene ring remains largely unexplored, limiting deeper insight into their chemical behaviour and potential.
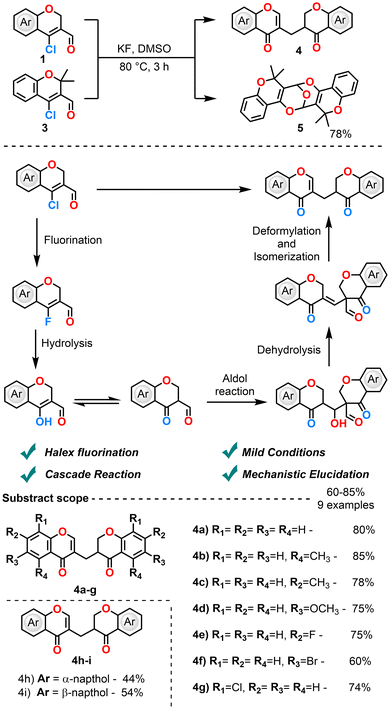
In 2019, the same group investigated the nucleophilic fluorination of CCCS employing KF in DMSO at 80 °C (Scheme 3).60 Instead of isolating the anticipated fluorinated products, the reaction predominantly furnished bischromene dimers. The pathway begins with a fluorine-substituted product, which hydrolyzes to yield 4-hydroxy chromene carbaldehyde. This further undergoes an intramolecular aldol reaction and dehydration, and then dimerizes via deformylation and isomerization to yield the bischromene dimers. Among the fluorinating agents screened, KF exhibited good reactivity and selectivity relative to TBAF and AgF. Benzochromene-based chloroaldehydes underwent fluorination, which was isolable after 45 minutes of reaction. The fluorinated product then underwent spontaneous dimerization via a cascade sequence. Further studies explained that the substrates bearing dimethyl substituents on the chromene ring facilitated the formation of structurally unique bicyclic benzopyran frameworks. Notably, chromene analogues lacking benzo-fusion did not yield isolable fluorinated species, highlighting a substrate-dependent limitation of the methodology. These observations underscore both the synthetic potential and the constraints of the protocol, particularly in the context of sterically encumbered systems, and suggest opportunities for further methodological refinement.
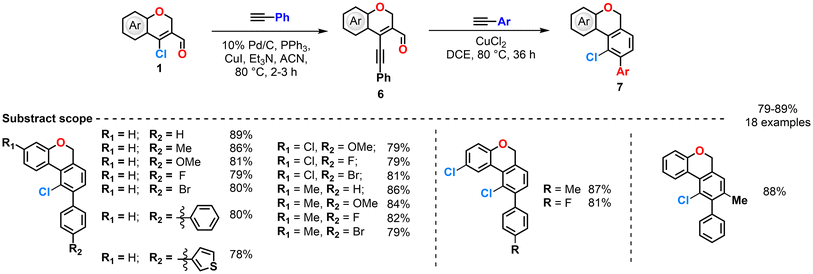
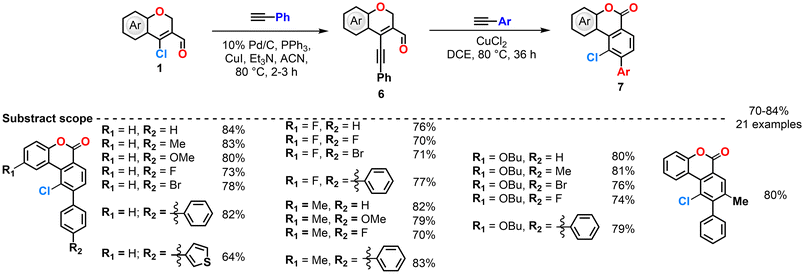
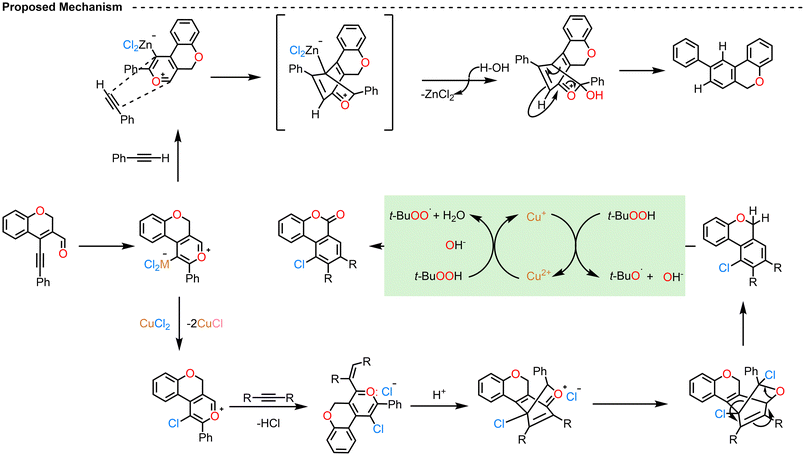
Reddy et al. (2017) reported a synthetic strategy for constructing polycyclic benzochromene and benzocoumarin derivatives (Scheme 4, 5, 6 and 7).68–70 The sequence begins with a Sonogashira C–C coupling between CCCS and arylacetylenes in the presence of Pd/C, PPh3, CuI, and triethylamine in refluxing acetonitrile, affording 4-(phenylethynyl)-2H-chromen-3-carbaldehyde intermediates. These intermediates subsequently react with arylacetylenes under different catalytic systems such as ZnCl2, CuCl2, and CuCl2/TBHP—to deliver distinct fused products. In the ZnCl2 system, π-complexation with the alkyne is followed by cyclization and retro-Diels–Alder elimination, producing 9-aryl-benzochromene analogues (Scheme 4).70 The group changed the catalyst to CuCl2, then the catalytic cycle involves π-complex formation, nucleophilic attack on the carbonyl carbon, and chlorination to generate a bicyclic intermediate, which then undergoes intramolecular chlorination and retro-Diels–Alder elimination to yield 10-chloro-9-aryl-benzochromenes. In this case, CuCl2 serves both as a catalyst and a chlorinating agent, providing higher yields compared with other chlorinating reagents such as NCS, Ca(OCl)2, FeCl3, AlCl3, SnCl4, LiCl, or PdCl2 (Scheme 5).69 Furthermore, upon modification of the catalytic system by the addition of TBHP, CuCl2 acted as both a catalyst and an oxidant, initiating a radical pathway. The initially formed 10-chloro-9-aryl-benzochromenes underwent hydrogen abstraction by t-BuO˙ radicals generated from TBHP, producing carbon-centered radicals that were subsequently oxidized to yield the final 10-chloro-9-aryl-benzocoumarins (Scheme 6).70 Possible mechanisms for these reactions are also provided (Scheme 7). Overall, this work demonstrates the versatility of chromenyl alkynes as key intermediates for constructing diverse fused polycyclic systems through metal- and oxidant-controlled annulation pathways.
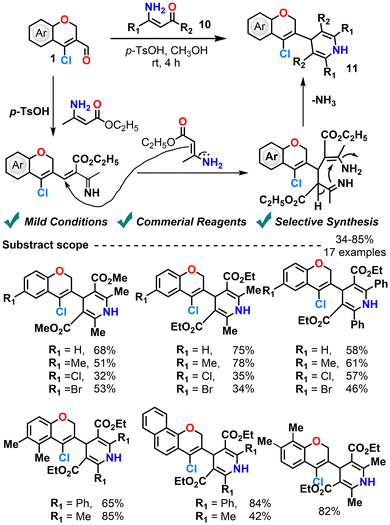
In another cyclization approach, Madhunala et al. (2019)71 utilized a Hantzsch-type multicomponent strategy for the one-pot synthesis of chromenyl-dihydropyridines from CCCS, ethyl acetoacetate, and ammonium hydroxide in methanol. Although the main product was the desired dihydropyridine, a competitive side reaction led to a minor formation of 5H-chromenopyridine. Attempts to suppress the side product using alternative ammonium salts such as NH4OH, NH4F, NH4Cl, NH4Br NH4NO3, NH4OAc, NH4(SO4)2 and (NH4)2CO3 and acid catalysts were only partially effective. However, replacing ethyl acetoacetate with ethyl 3-aminocrotonate and using p-toluenesulfonic acid (p-TsOH) as a catalyst afforded the desired product in up to 75% yield without any side reactions (Scheme 8).71 Condensation of the aldehyde by p-toluenesulfonic acid (p-TsOH) generates an iminium intermediate, which subsequently undergoes nucleophilic addition with ethyl 3-aminocrotonate to yield a β-enamino ester. Furthermore, intramolecular cyclization, followed by elimination of ammonia, leads to the formation of 1,4-dihydropyridine. Notably, chromene substrates bearing electron-donating groups afforded significantly higher yields, whereas those with electron-withdrawing substituents resulted in reduced efficiency.
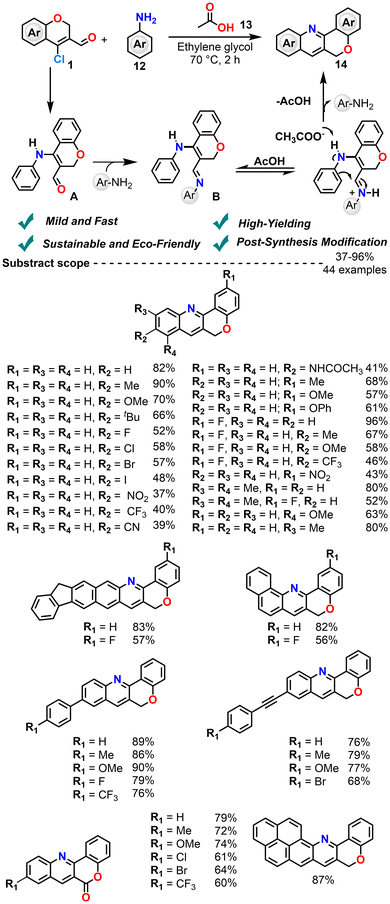
In 2025, Reddy et al. reported a sustainable and metal-free synthesis to access fused chromenoquinoline analogues (Scheme 9).72 The method involves a Doebner–Von Miller-type reaction of CCCS with substituted anilines, promoted by an acetic acid/ethylene glycol system. The mechanism proceeds via N-alkenylation (A), iminoenamine (B) formation, and intramolecular cyclization to afford the chromenoquinoline framework. This protocol is broadly applicable with 44 derivatives and yields up to 96%. The process is notably eco-friendly, with a high atom economy of 81.2% and a low E-factor of 0.231. Furthermore, they demonstrated the versatility of the products by performing late-stage functionalization. This was achieved through Suzuki–Miyaura coupling, Sonogashira coupling, and C(sp3)–H oxidation reactions.
3.2 Medicinal application
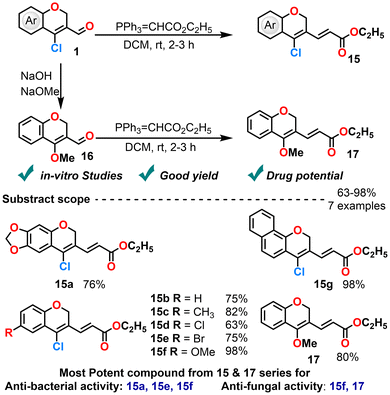
2H-Chromene and its derivatives constitute a privileged scaffold73 with diverse pharmacological activities, including anticancer,74 antimicrobial75 and antioxidant activities.76 Their biological relevance stems from structural features that enable strong interactions with various biomolecular targets.In a 2016 study, Subhashini et al. synthesized a series of (2E)-ethyl-3-(4-chloro-2H-chromen-3-yl)acrylates via a Wittig reaction starting from CCCS (Scheme 10).79 These compounds exhibited significant antimicrobial activity. Particularly, the derivative bearing a chloro-substituent at the 6-position of the chromene ring outperformed the standard antibiotic ampicillin against Bacillus subtilis in in vitro studies. Other derivatives, including those with methylenedioxy, methoxy and bromo substituents at position 6, also showed strong antibacterial activity against P. aeruginosa, S. aureus, and E. coli. Additionally, a methoxy substituent at the 6-position of the chromene ring moiety and a methoxy substituent at the 4-position demonstrated potent antifungal activity against Sclerotium rolfsii and Macrophomina phaseolina. These results underscore the critical role of halogen and electron-donating substituents on the chromene scaffold in modulating antimicrobial efficacy. However, the structure–activity relationship is very briefly discussed. Expanding the diversity of the compound library and conducting a more detailed analysis of substituent effects on antimicrobial efficacy could add significant value to the study. These areas also offer promising directions for future research to enhance the understanding and potential of these compounds in drug development.
In a related study (2019), Shivaraj et al. reported the synthesis of N-(4-chloro-2H-chromen-3-yl)methylenebenzohydrazides by condensing CCCS with various arylhydrazides (Scheme 11).80 Compounds such as 6-fluoro-chromen-3′,4′,5′-trimethoxybenzohydrazide and 6-bromo-chromen-isonicotinohydrazide exhibited broad-spectrum antibacterial activity against both Gram-positive and Gram-negative strains. Derivatives bearing electron-donating groups on the benzo-hydrazine moiety were particularly active. Moreover, a fluoro-substituted chromene analogue and a 4-substituted methoxy benzohydrazide derivative showed notable antifungal efficacy, especially against Candida albicans, further supporting the utility of halogen substitution in enhancing antimicrobial profiles. Additionally, the scope of this research can be extended to a thorough mechanistic investigation of how the compounds produce their antimicrobial activities. While some structure–activity insights are provided, a more comprehensive analysis could facilitate the optimal design and potency of the compounds.
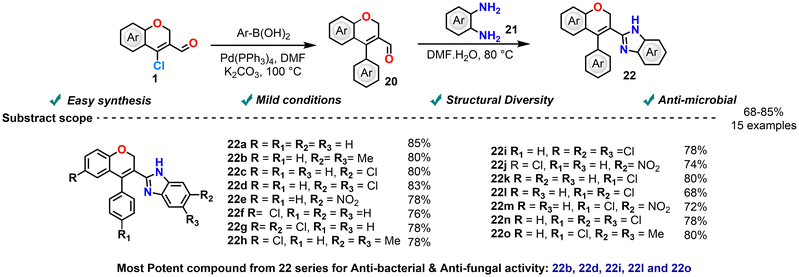
In 2021, the study conducted by Dasari et al. presented the synthesis and antimicrobial assessment of a novel series of 4-phenyl-2H-chromene-3-benzimidazole derivatives (Scheme 12).81 The synthetic route involved an initial Suzuki coupling between appropriate aryl boronic acids and 4-bromo-2H-chromenes, followed by condensation of the resulting intermediates with o-phenylenediamines to yield the final benzimidazole products. All synthesized compounds were evaluated for their in vitro antimicrobial activity. Antibacterial efficacy was tested against S. aureus, B. subtilis, E. coli, and P. aeruginosa using the agar well diffusion method, with ciprofloxacin serving as the reference standard. Antifungal activity was examined against A. niger and C. albicans via the disc diffusion method, with voriconazole as the standard. In particular, chlorine- and methyl-substituted analogues exhibited significant antibacterial and antifungal activity, in some cases surpassing that of the standard drugs. These promising results suggest that the 4-phenyl-2H-chromene-3-benzimidazole scaffolds hold potential for further development as a novel class of antimicrobial agents, meriting detailed exploration of their mechanism of action and pharmacological profiles.
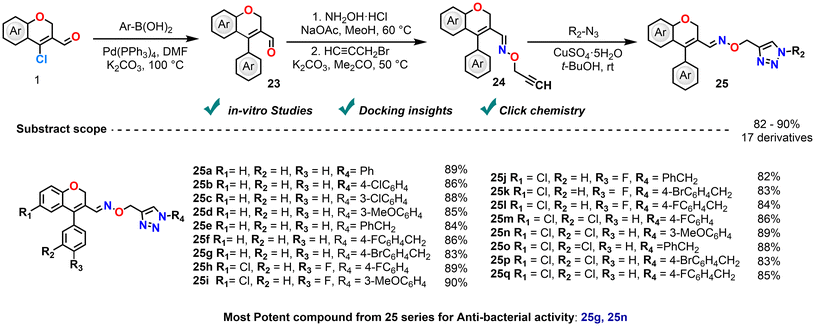
The same group synthesized a series of (E)-4-aryl-2H-chromene-3-carbaldehyde O-[(1-substituted-1H-1,2,3-triazol-4-yl)methyl]oximes to evaluate their antimicrobial efficacy. The synthetic route involved Suzuki coupling, followed by oxime formation, N-alkynation and Cu(I)-catalyzed 1,3-dipolar cycloaddition with organic azides (Scheme 13).82 Six compounds exhibited notable activity against E. coli, S. aureus, P. aeruginosa, and B. subtilis, with ciprofloxacin as the reference. Notably, chloro-substituted chromene analogues and bromo-benzyl-substituted triazole analogues displayed enhanced potency. Docking studies revealed strongly interacting derivatives with His377, supporting their relevance as lead antimicrobial candidates. Additionally, the scope of this research can be broadened to incorporate in vivo evaluation and thorough ADME profiling, which may aid in an improved understanding of a molecule's pharmacokinetic activities and overall therapeutic potential.
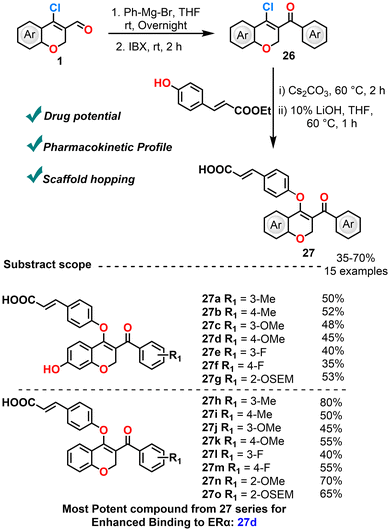
In 2021, Lu et al. synthesized a series of 2H-chromene-3-carbonyl derivatives starting from CCCS. Structural diversification was achieved via Grignard addition of phenyl magnesium bromide to form secondary alcohols, followed by oxidation and coupling with 4-hydroxycinnamic acid methyl esters (Scheme 14).85 Biological evaluation against MCF-7 breast cancer cells revealed that the presence of hydroxyl and methoxy substituents significantly enhanced binding to estrogen receptor α (ERα). The most potent compound—7-hydroxy-4′-methoxybenzoyl chromene acrylic acid—demonstrated marked antiestrogenic activity, reduced Ki67 expression, induced apoptosis, and outperformed fulvestrant, a drug which binds to estrogen receptor α, blocks its activity, and promotes its degradation, thereby shutting down estrogen-driven signaling in cancer cells. The compound exhibited strong pharmacological activity with an IC50 value of 0.81 µM. At 1 µM concentration, it achieved an inhibition of 99.78%, which is slightly higher than the reference drug fulvestrant (97.1%). These findings support the potential of chromene-based frameworks in targeting ERα-positive breast cancer cells, although the issue of drug resistance is acknowledged. Besides, there is scope for in-depth investigation of potential resistance mechanisms to the new compounds. This omission limits insight into strategies for overcoming resistance, such as combination therapies or structural modifications.
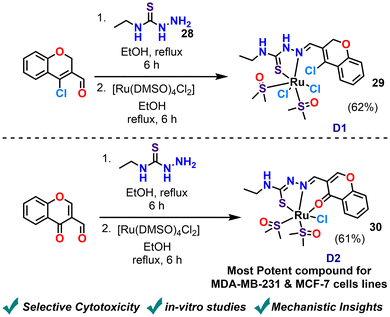
In 2022, Balakrishnan et al. explored the impact of ligand denticity in Ru(II)–DMSO complexes on their cytotoxicity against breast cancer cells. They synthesised two ruthenium complexes employing a Schiff base ligand derived from 2H-chromene or 4H-chromene-based carbaldehyde and N-ethylhydrazinecarbothioamide (Scheme 15).86 The complexes were evaluated for their anticancer potential. A key finding of the research is the superior in vitro anticancer activity of the complex with tridentate chromone TSC (D2) compared to the complex with bidentate chromene TSC (D1). The authors provide a clear rationale for this observed difference, attributing it to the enhanced thermodynamic stability of the tridentate system. Furthermore, hydrolysis studies reveal that D1 possesses a kinetically labile chloride ligand, suggesting greater potential for interaction with biological molecules. D2 also displays selective cytotoxicity against breast cancer cells while exhibiting lower toxicity towards normal cells, highlighting its potential as a more effective and safer anticancer agent. Notably, complex D2 displayed reasonable cytotoxicity against breast cancer cell lines, with IC50 values of 23.69 μM for MDA-MB-231 and 27.62 μM for MCF-7 cells.
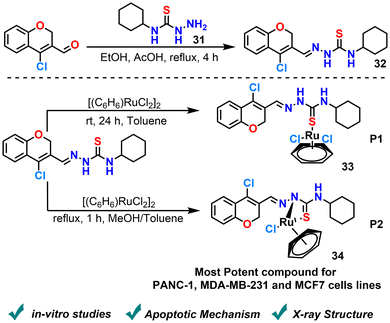
Haribabu et al. (2024) reported the synthesis of Ru(II)-benzene complexes utilizing a 2H-chromene-based thiosemicarbazone ligand (Scheme 16).87 Building upon the chromene scaffold, two coordination modes were explored, affording complexes P1 (monodentate) and P2 (bidentate). Both complexes were thoroughly characterized by spectroscopic and analytical techniques, confirming their structures and coordination geometries. Biological evaluation revealed strong binding affinities toward DNA and bovine serum albumin (BSA), suggesting high biomolecular interaction potential. Notably, the bidentate complex P2 exhibited superior anticancer (PANC-1, MDA-MB-231 and MCF7 cell lines) activity compared to P1 and outperformed cisplatin in cytotoxic assays. Mechanistic studies indicated that P2 induces reactive oxygen species (ROS)-mediated apoptosis, highlighting its promise as a next-generation metal-based chemotherapeutic agent. The study investigates a single thiosemicarbazone ligand with two coordination sites. Furthermore, there is scope for enhanced ligand design and in vivo evaluation, which can yield a thorough assessment of chromene- and thiosemicarbazone-based complexes for potential anticancer drugs.
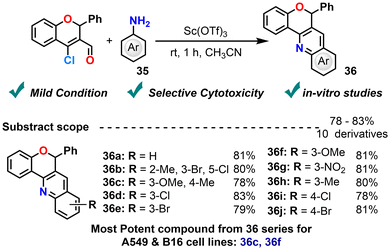
Rao et al. (2016) reported an efficient synthesis of 6-phenyl-6H-chromeno[4,3-b]quinoline derivatives via the reaction of CCCS with aniline, using scandium(III)trifluoromethanesulfonate [Sc(OTf)3] as a Lewis acid catalyst in acetonitrile at room temperature (Scheme 17).88 Although Sc(OTf)3 affords high yields, the reason for its effectiveness is not discussed. The appropriate reason that Sc(OTf)3 gives higher yields than BF3·OEt2, AlCl3, and Ln(OTf)3 is due to its superior water tolerance and stronger Lewis acidity. This enables understanding of the significance of this catalyst in a better way. Furthermore, the resulting chromenoquinoline compounds were evaluated for their anticancer potential against A549 (human lung carcinoma) and B16 (murine melanoma) cell lines. Among the tested compounds, the aniline derivative bearing methoxy and methyl groups at positions 3 and 4, respectively, shows a notably low IC50 of 0.19 μM against A549 cells, whereas the compound with the methoxy group at position 3 exhibits the most promising cytotoxic activity demonstrating good activity against both cell lines, with IC50 values of 3.20 μM (A549) and 1.90 μM (B-16), underscoring the relevance of electronic effects in modulating bioactivity. These findings highlight chromenoquinoline scaffolds as promising leads for future advancements in cancer chemotherapy. Building upon the current approach by exploring a broader range of aromatic amines and introducing variations in the chromene framework could further enrich the compound library, offering valuable opportunities to deepen the understanding of structure–activity relationships.
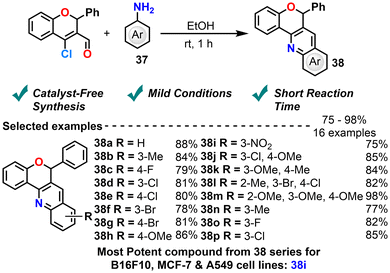
Kumar et al. (2017) developed a catalyst-free, one-pot synthesis of novel 6-phenyl-6H-chromeno[4,3-b]quinoline derivatives using 4-chloro-2-phenyl-2H-chromene-3-carbaldehyde and substituted aromatic anilines under ambient conditions (Scheme 18).89 This approach is operationally simple, proceeds at room temperature and affords good yields in short reaction times. The synthesized compounds were evaluated for cytotoxic activity against B16F10 (murine melanoma), MCF-7 (human breast cancer), and A549 (human lung carcinoma) cell lines. The biological evaluation is limited to the cytotoxicity of a single compound (i.e., m-substituted nitro aniline) against three cancer cell lines. It shows decent activity against B16F10 (IC50 14.8 µM) and MCF7 (IC50 21.32 µM) cells but poor activity against A549 cells (IC50 199.85 µM). Additionally, it is important to understand the reaction mechanism, conduct controlled experiments, and expand the synthesis scope for enhancing the library. Furthermore, there is an opportunity for improved ligand design and in vivo evaluation, which can provide a comprehensive assessment of 6-phenyl-6H-chromeno[4,3-b]quinoline derivatives as future anticancer agents.
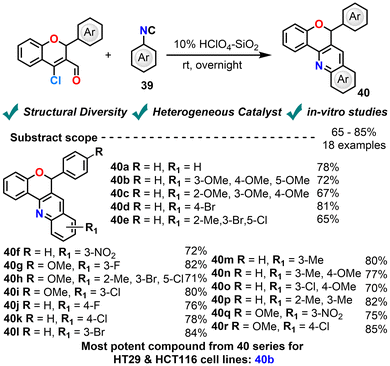
In a 2021 study, Sultana et al. synthesized a series of chromene–quinoline hybrids via the reaction of CCCS with substituted aryl isocyanides in methanol using silica-supported perchloric acid (HClO4-SiO2) as a catalyst via modified Friedländer annulations (Scheme 19).90 Varying the catalyst loading of HClO4-SiO2 allowed the assessment of its efficiency and provided insights into its potential for optimization. This method provided a straightforward and efficient route to access structurally diverse heterocyclic frameworks integrating two bioactive scaffolds—chromene and quinoline. The resulting compounds were screened for their cytotoxicity against a panel of cancer cell lines. Notably, the 3,4,5-trimethoxyphenyl isocyanide-derived analogue exhibited significant activity with a very low IC50 value of 2.61 ± 0.34 µM against the HT29 cell line and 8.1 ± 0.2 µM against HCT116. The study underscores the potential of these hybrid molecules in anticancer drug development, with the trimethoxy substitution pattern contributing to enhanced bioactivity.
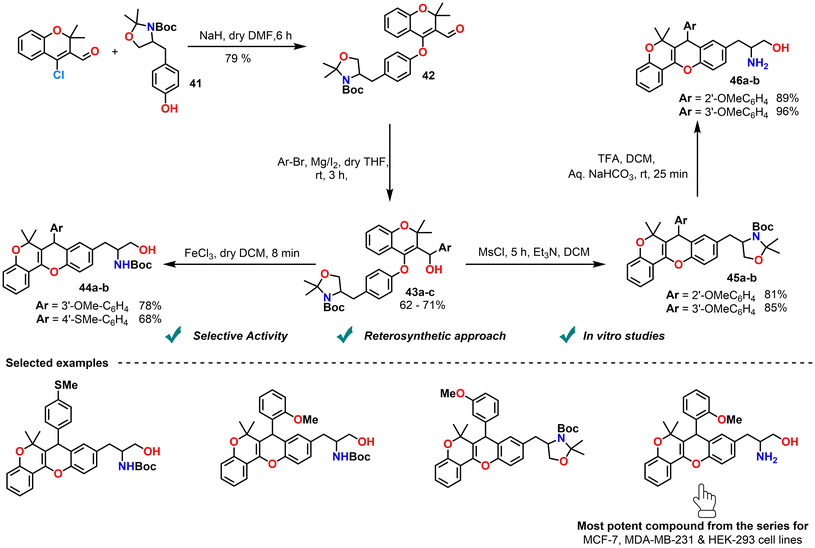
In 2018, Panda et al. reported the synthesis of chromene–xanthene hybrids via a strategy incorporating L-tyrosine as a hydroxylated/thiohydroxylated arene intermediate. This derivative reacted with CCCS, followed by Grignard addition and Friedel–Crafts type cyclization, yielding arenoxy carbinols that underwent cyclization. When FeCl3 was used, deprotection occurred and the oxazolidine ring opened because FeCl3, as a strong Lewis acid, coordinates with the heteroatom, polarizes the C–N/O bonds, and triggers cleavage. In contrast, MsCl acts only as a sulfonylating reagent that functionalizes the heteroatom without destabilizing the ring, so it remained intact; however, subsequent treatment with TFA in DCM followed by aqueous NaHCO3 promoted ring opening to afford the desired chromene–xanthene hybrids (Scheme 20).91 The use of L-tyrosine introduced polar functionalities, enhancing both solubility and bioactivity. Biological evaluation against breast cancer cell lines revealed potent cytotoxicity of the 2-methoxyphenyl-substituted analogue, with IC50 values of 2.6 ± 0.667 µM against MCF-7, 2.5 ± 0.181 µM against MDA-MB-231 and >25 µM against HEK-293, significantly lower than those of most other compounds. This analogue induced G1 phase arrest and apoptosis in MDA-MB-231 cells, a triple-negative breast cancer model. The study highlighted the value of chromene–xanthene hybridization and amino acid incorporation as a strategy to improve pharmacological properties. These findings support further development of chromene-based hybrids as anticancer agents, with future SAR and in vivo studies to optimize therapeutic potential.
Structural modifications, such as incorporating hydroxyl or methoxy groups, have been found to enhance blood–brain barrier permeability and improve therapeutic efficacy. These properties position 2H-chromene derivatives as promising candidates for further development in neurodegenerative disorder treatment.
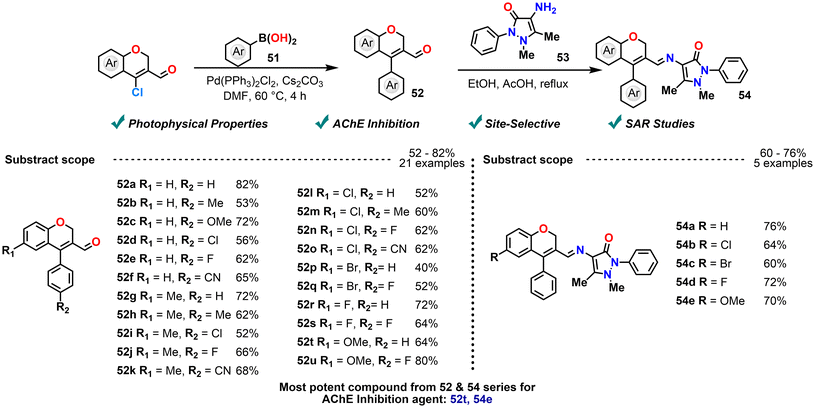
In 2023, Reddy et al. synthesized benzisoxazole-linked chromenes via site-selective C–N coupling between CCCS and fluoro-substituted benzisoxazole derivatives through an aza-Michael addition/elimination.94 In a subsequent 2024 study, they developed a sustainable route to benzisoxazole–chromene acyl hydrazones through condensation with benzo hydrazides and 6-fluorobenzo[d]isoxazole-piperidine. The synthesized compounds were then evaluated for acetylcholinesterase inhibition by in silico studies, and the results showed good activity towards AChE inhibition (Scheme 21).95 The in silico studies that are molecular docking, DFT calculations, and molecular dynamics simulations revealed strong binding affinities of the hydrazone derivatives toward acetylcholinesterase (AChE). In particular, the presence of a 4-hydroxy and 4-nitro moiety on the hydrazone core significantly enhanced binding interactions, with selectivity towards AChE over BChE. The results highlight the promise of these molecules as candidates for neurodegenerative disease therapy based on docking investigations. Further investigation into in vitro enzyme inhibition assays and in vivo validation is necessary to substantiate the efficacy of the AChE inhibition assertions.
The study was extended by carrying out a Pd(II)-catalyzed site-selective cross-coupling approach to synthesize fluorescent aryl-formyl chromenes and their iminoantipyrine analogues, targeting acetylcholinesterase (AChE) inhibition. The method employed Suzuki–Miyaura coupling to efficiently generate 4-aryl-3-formyl-2H-chromenes, followed by imine formation with 4-amino-dimethylphenyl-pyrazolone (Scheme 22).96 Notably, the synthesized compounds showed green-yellow fluorescence. Among the synthesized compounds, the 6-methoxy-substituted chromene derivative exhibited notable AChE inhibitory activity, as supported by molecular docking and DFT studies. These results highlight their potential as lead compounds for neurodegenerative disease therapy. These findings highlight the potential of these compounds for neurodegenerative disease treatment. Additional investigation into in vitro enzyme inhibition assays and in vivo validation is required to confirm the claims of AChE inhibition efficacy.
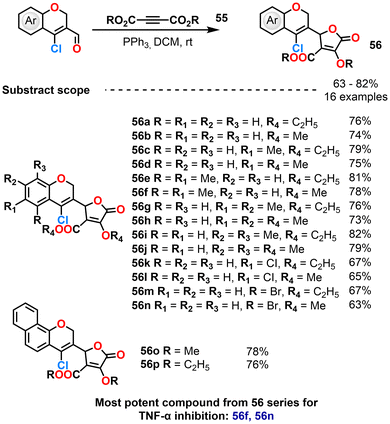
Kurma et al. (2020) evaluated the anti-inflammatory potential of chromenyl-dihydrofuran-carboxylates, which were efficiently synthesized via a [3 + 2] cycloaddition of 2H-chromene-3-carbaldehydes with electron-deficient alkynes using PPh3 as the catalyst (Scheme 23).98 The synthesized compounds were evaluated for their inhibitory effects on tumour necrosis factor-alpha (TNF-α), a central mediator of inflammation. Derivatives featuring 5,6-dimethoxy substitution on the chromene ring coupled with a methoxy group on the furanone moiety with an IC50 value of 0.142 ± 0.001 μM, as well as those bearing a 5-bromo substituent on the chromene ring with an ethoxy group on the furanone ring with an IC50 value of 3.261 ± 0.23 μM, demonstrated the most potent TNF-α inhibition with minimal cytotoxicity. The presence of the chloro substituent on the chromene scaffold was found to significantly enhance anti-inflammatory efficacy. In vivo assays further validated the therapeutic potential of these compounds, showing marked reductions in TNF-α and interleukin-6 (IL-6) levels, supporting their application in treating inflammation-associated disorders.
3.3 Miscellaneous application
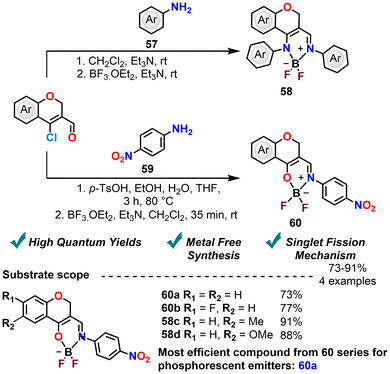
2H-Chromenes have garnered significant attention in fluorescence and phosphorescence studies owing to their strong emission properties, making them valuable candidates for use as fluorescent probes99 and dyes in imaging and sensing applications. Their tunable optical characteristics and high photostability also position them as promising building blocks in the development of advanced optoelectronic and photonic materials. Moreover, chromene derivatives have been employed in the total synthesis of natural products and bioactive compounds, leveraging their structural versatility and reactivity. Beyond medicinal chemistry and organic synthesis, their incorporation into functional materials—such as polymeric sensors,100 organic light-emitting diodes (OLEDs),101 and responsive molecular systems—continues to expand the scope of their applications.Balasubramanian et al. (2012) synthesized β-iminoenamine–BF2 BODIPY like complexes using the CCCS and various anilines,1 aiming to explore their aggregation-induced emission (AIE) characteristics and to examine how aliphatic ring substitution influences non-radiative deactivation pathways. BODIPYs (boron-dipyrromethenes) are fluorescent dyes composed of a dipyrromethene core chelated to a boron difluoride (BF2) unit, whose rigid, planar π-conjugated framework imparts high photostability, sharp absorption profiles, and strong fluorescence. Their results demonstrated that solid-state emission quantum yields were significantly higher than those observed in solution at room temperature. Subsequently, in 2014, they reported the metal-free synthesis of β-hydroxyvinylimines via the condensation of β-chlorovinylaldehydes with 4-nitroaniline, catalyzed by p-toluenesulfonic acid (p-TsOH). The obtained intermediates were subsequently transformed into boron-containing oxa-BODIPY like derivatives via treatment with BF3·OEt2. Oxa-BODIPYs are structurally analogous to conventional BODIPYs, except that one of the core dipyrromethene nitrogen atoms is replaced by an oxygen atom. This heteroatom substitution induces distinct electronic perturbations within the π-conjugated framework, typically resulting in hypsochromic shifts in both absorption and fluorescence emission spectra relative to their parent BODIPY like analogues (Scheme 24).2 The resulting materials exhibited room-temperature phosphorescence via singlet fission, both in solution and within poly(methyl methacrylate) (PMMA) matrices. Non-substituted CCCS have the highest quantum yield. Single-crystal X-ray diffraction revealed that π–π stacking interactions and excimer formation facilitated two-photon generation. Notably, the quantum yield (QY) was maintained at low concentrations but declined at higher concentrations due to self-quenching effects.
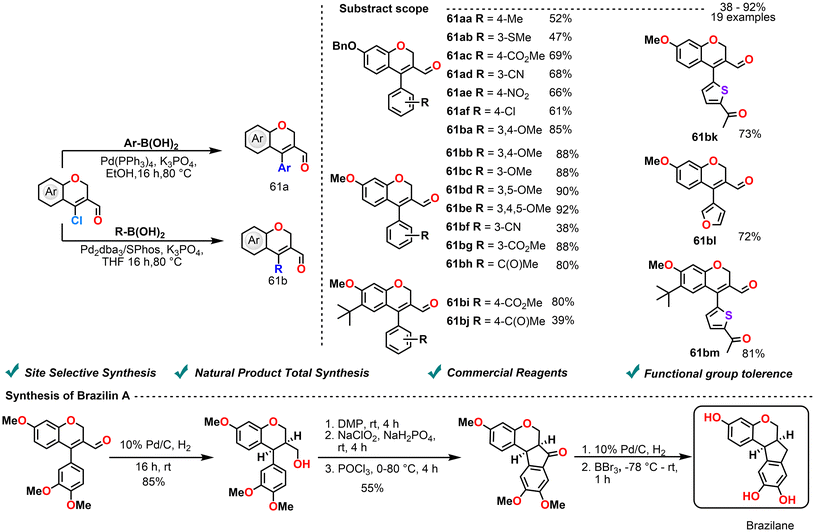
In 2018, Kim et al. reported the synthesis of 4-(hetero)aryl-2H-chromene-3-carbaldehydes as intermediates for the preparation of natural products such as homoisoflavonoids and brazilane. Utilizing a Suzuki–Miyaura cross-coupling reaction, a variety of arylboronic acids were efficiently coupled under standard palladium-catalyzed conditions, affording β-aryl-α,β-unsaturated aldehydes in high yields. This method demonstrated broad substrate tolerance, accommodating electron-rich, electron-deficient, and heteroaryl boronic acids. Subsequent efforts focused on aldehyde cyclization, which involved a two-step Kraus–Pinnick oxidation, followed by treatment with POCl3 to yield the corresponding indanone. Catalytic hydrogenation of ketone and selective deprotection with BBr3 ultimately led to the synthesis of brazilin A. This study outlines an efficient synthetic route to complex natural products via sequential coupling, oxidation, and reduction steps (Scheme 25).102
4. Conclusion
In this review, we present the structurally diversified 4-chloro-2H-chromene-3-carbaldehyde (CCCS) and how it emerged as a highly adaptable scaffold that enables a broad spectrum of synthetic transformations through the combined reactivity of its aldehyde and β-chlorovinyl groups. Condensations such as aldol and Wittig provide α,β-unsaturated systems, while Pd-, Ni- and Cu-catalysed couplings or nucleophilic substitutions at the vinylic chloride furnish aryl, heteroaryl and alkynyl derivatives. Dimerization under transition metal catalysis affords bichromene frameworks with atropisomerism, and annulations of alkynes deliver benzochromenes and benzocoumarins. Multicomponent Hantzsch condensations further extend the chemistry to chromenyl-dihydropyridines with excellent selectivity.Additionally, CCCS have enabled several advanced transformations. Aza-Michael reactions generate benzisoxazole-linked chromenes, while Cu(I)-catalysed click reactions furnish triazolyl oximes with modular functionality. Grignard additions at the aldehyde centre yield secondary alcohols readily oxidised or esterified to chromene-acrylic acids. Condensations with o-phenylenediamines give access to benzimidazole frameworks, and Schiff base formation with thiosemicarbazones provides ligands for Ru(II) coordination, affording stable metal complexes. Modified Friedländer annulations employing isocyanides have delivered chromenoquinoline hybrids under heterogeneous catalysis. Furthermore, sustainable variants such as metal-free Doebner–Von Miller cyclizations offer fused chromenoquinolines in high yields. These methodologies clearly demonstrate that CCCS are highly notable and versatile building blocks, capable of bridging classical condensation chemistry with modern catalytic, multicomponent and green strategies.
CCCS-derived scaffolds have attracted significant attention in medicinal chemistry owing to their broad pharmacological profile. A variety of derivatives exhibit antibacterial, antifungal and anticancer activities, with several compounds displaying micromolar to sub-micromolar potency. Structural features such as methoxy and halogen substitution often enhance activity, while annulated heterocycles derived from CCCS show promising selectivity towards tumour cell lines. Certain analogues also demonstrate anti-inflammatory and neuroprotective effects, suggesting potential utility in the treatment of chronic degenerative disorders. Schiff base formation followed by coordination with transition metals, particularly Ru(II), has yielded metal complexes with marked cytotoxicity, highlighting an important extension into organometallic therapeutics. In addition, benzimidazole- and triazole-linked hybrids derived from CCCS have shown notable efficacy against resistant bacterial strains, reinforcing the medicinal utility of this scaffold. The most potent members of each series are highlighted in the appropriate schemes and these show the potential for developing new pharmaceuticals.
Beyond therapeutics, CCCS derivatives also contribute to the development of advanced functional materials. Their conjugated chromene backbone supports aggregation-induced emission, as well as room-temperature phosphorescence and fluorescence. These promising photophysical properties enable potential applications in energy-related materials, including optoelectronics, semiconductors, solar technologies, chemical sensing, luminescent probes, and OLEDs.
Looking ahead, these frameworks hold significant promise, with opportunities for advancement in in vivo studies, enhanced reaction selectivity, and the establishment of precise structure–activity relationships across diverse therapeutic areas. CCCS are well-positioned to act as both a vital foundation and a unifying bridge between synthetic chemistry, drug discovery, and materials research. Future research will benefit from prioritizing environmentally sustainable and targeted strategies for regioselective, stereospecific, and stereoselective synthesis. Coupled with more refined biological evaluations, these efforts are expected to unlock the full potential of CCCS and drive innovation in next-generation pharmaceuticals.
Conflicts of interest
The authors declare no conflict of interest.Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this review.Acknowledgements
S. R. Reddy is grateful to Anusandhan National Research Foundation (ANRF) India (Grant No. CRG/2023/008520) and SPARC/2019-2020/P1905/SL for providing financial support for carrying out the research work. Furthermore, we thank the Vellore Institute of Technology (VIT), Vellore, India, for providing the seed grant SG20230119 and VIN/2023-24/037 (VIN-award by VIT, Vellore) for providing financial assistance.References
- K. Perumal, J. A. Garg, O. Blacque, R. Saiganesh, S. Kabilan, K. K. Balasubramanian and K. Venkatesan, Chem. – Asian J., 2012, 7, 2670–2677 CrossRef CAS PubMed.
- M. Koch, K. Perumal, O. Blacque, J. A. Garg, R. Saiganesh, S. Kabilan, K. K. Balasubramanian and K. Venkatesan, Angew. Chem., Int. Ed., 2014, 53, 6378–6382 CrossRef CAS.
- S. Muthuramalingam, J. A. Garg, R. Karthick, T. Fox, O. Blacque, K. Venkatesan, S. Ramanathan, S. Kabilan and K. K. Balasubramanian, Org. Chem. Front., 2019, 6, 134–139 RSC.
- E. Kabir and M. Uzzaman, Results Chem., 2022, 4, 100606 CrossRef CAS.
- P. Chandra, ChemistrySelect, 2021, 6, 10274–10322 CrossRef CAS.
- L. Mercs and M. Albrecht, Chem. Soc. Rev., 2010, 39, 1903 RSC.
- M. H. A. Al-Jumaili, A. A. Hamad, H. E. Hashem, A. D. Hussein, M. J. Muhaidi, M. A. Ahmed, A. H. A. Albanaa, F. Siddique and E. A. Bakr, J. Mol. Struct., 2023, 1271, 133970 CrossRef CAS.
- S. Sharma, D. Kumar, G. Singh, V. Monga and B. Kumar, Eur. J. Med. Chem., 2020, 200, 112438 CrossRef CAS PubMed.
- W. Luo, Y. Liu, H. Qin, Z. Zhao, S. Wang, W. He, S. Tang and J. Peng, Eur. J. Med. Chem., 2024, 279, 116838 CrossRef CAS.
- N. Puranik and M. Song, Neurol. Int., 2025, 17, 26 CrossRef CAS PubMed.
- A. N. Panche, A. D. Diwan and S. R. Chandra, J. Nutr. Sci., 2016, 5, e47 CrossRef CAS.
- R. Pavela, F. Maggi and G. Benelli, Nat. Prod. Res., 2021, 35, 1566–1571 CrossRef CAS.
- J. Sharifi-Rad, N. Cruz-Martins, P. López-Jornet, E. P.-F. Lopez, N. Harun, B. Yeskaliyeva, A. Beyatli, O. Sytar, S. Shaheen, F. Sharopov, Y. Taheri, A. O. Docea, D. Calina and W. C. Cho, Oxid. Med. Cell. Longevity, 2021, 2021, 6492346 CrossRef PubMed.
- J.-F. Wang, S.-S. Liu, Z.-Q. Song, T.-C. Xu, C.-S. Liu, Y.-G. Hou, R. Huang and S.-H. Wu, Molecules, 2020, 25, 5112 CrossRef CAS.
- P. Sivaguru, R. Sandhiya, M. Adhiyaman and A. Lalitha, Tetrahedron Lett., 2016, 57, 2496–2501 CrossRef CAS.
- N. M. Sabry, H. M. Mohamed, E. S. A. E. H. Khattab, S. S. Motlaq and A. M. El-Agrody, Eur. J. Med. Chem., 2011, 46, 765–772 CrossRef CAS.
- L. Dinparast, S. Hemmati, A. A. Alizadeh, G. Zengin, H. S. Kafil, M. B. Bahadori and S. Dastmalchi, J. Mol. Struct., 2020, 1203, 127426 CrossRef CAS.
- M. Puppala, X. Zhao, D. Casemore, B. Zhou, G. Aridoss, S. Narayanapillai and C. Xing, Bioorg. Med. Chem., 2016, 24, 1292–1297 CrossRef CAS PubMed.
- S. Ghomashi, R. Ghomashi, M. S. Damavandi, Z. Fakhar, S. Y. Mousavi, A. Salari-Jazi, S. Gharaghani and A. R. Massah, Sci. Rep., 2024, 14, 12878 CrossRef CAS PubMed.
- R. Pratap and V. J. Ram, Chem. Rev., 2014, 114, 10476–10526 CrossRef CAS.
- S. Maity, A. Roy, S. Duari, S. Biswas, A. M. Elsharif and S. Biswas, ACS Omega, 2023, 8, 46614–46627 CrossRef CAS PubMed.
- N. Majumdar, K. A. Korthals and W. D. Wulff, J. Am. Chem. Soc., 2012, 134, 1357–1362 CrossRef CAS.
- C.-C. Chen, M.-Y. Wu, H.-Y. Chen and M.-J. Wu, J. Org. Chem., 2017, 82, 6071–6081 Search PubMed.
- R. K. Maurya, A. Dey, V. Kumara and M. Khatravath, Asian J. Org. Chem., 2024, e202400259 Search PubMed.
- Q. Yang, Y. Wang, S. Luo and J. Wang, Angew. Chem., Int. Ed., 2019, 58, 5343–5347 CrossRef CAS PubMed.
- K. U. Sadek, R. A. H. Mekheimer, M. Abd-Elmonem, A. Abdel-Hameed and M. H. Elnagdi, Tetrahedron: Asymmetry, 2017, 28, 1462–1485 CrossRef CAS.
- H. C. Shen, Tetrahedron, 2009, 65, 3931–3952 CrossRef CAS.
- D. Ispán, B. Varga, S. Balogh, B. Zsirka, Á. Gömöry and R. Skoda-Földes, ChemistrySelect, 2021, 6, 5705–5710 CrossRef.
- A. Alizadeh and A. Rostampoor, ChemistrySelect, 2022, 7, e202200299 Search PubMed.
- Y. Dong, Y. Tu, K. Wang, C. Xu, Y. Yuan and J. Wang, Angew. Chem., Int. Ed., 2020, 59, 7168–7172 CrossRef CAS.
- K. Samanta, G. K. Kar and A. K. Sarkar, Tetrahedron Lett., 2008, 49, 1461–1464 Search PubMed.
- M. Gil-Ordóñez, A. Maestro and J. M. Andrés, J. Org. Chem., 2023, 88, 6890–6900 CrossRef.
- L. Goswami, K. Neog, K. Sharma and P. Gogoi, Org. Biomol. Chem., 2017, 15, 6470–6473 RSC.
- P. Saini, Jyoti, P. K. Sharma and V. Singh, Eur. J. Org. Chem., 2024, e202401058 Search PubMed.
- J. Wagner, D. Kumar, M. A. Kochman, T. Gryber, M. Grzelak, A. Kubas, P. Data and M. Lindner, ACS Appl. Mater. Interfaces, 2023, 15, 37728–37740 Search PubMed.
- Y. Yang, Y. Wu, Z. Bin, C. Zhang, G. Tan and J. You, J. Am. Chem. Soc., 2024, 146, 1224–1243 Search PubMed.
- S. M. Elbert, M. Reinschmidt, K. Baumgärtner, F. Rominger and M. Mastalerz, Eur. J. Org. Chem., 2018, 532–536 CrossRef CAS.
- S. S. Ichake, B. K. Villuri, S. R. Reddy, V. Kavala and C.-F. Yao, Org. Lett., 2019, 21, 2256–2260 CrossRef CAS.
- V. Yadav, J. Reang, V. Sharma, J. Majeed, P. C. Sharma, K. Sharma, N. Giri, A. Kumar and R. K. Tonk, Chem. Biol. Drug Des., 2022, 100, 389–418 CrossRef CAS.
- D. Tiglani, Salahuddin, A. Mazumder, M. S. Yar, R. Kumar and M. J. Ahsan, Polycyclic Aromat. Compd., 2022, 42, 5044–5066 CrossRef CAS.
- P. Yadav and K. Shah, Bioorg. Chem., 2021, 109, 104639 CrossRef CAS PubMed.
- Y.-L. Zhong, D. T. Boruta, D. R. Gauthier and D. Askin, Tetrahedron Lett., 2011, 52, 4824–4826 CrossRef CAS.
- I. Carson, K. J. MacRuary, E. D. Doidge, R. J. Ellis, R. A. Grant, R. J. Gordon, J. B. Love, C. A. Morrison, G. S. Nichol, P. A. Tasker and A. M. Wilson, Inorg. Chem., 2015, 54, 8685–8692 CrossRef CAS.
- A. F. Begum, K. K. Balasubramanian and B. Shanmugasundaram, Tetrahedron Lett., 2021, 82, 153372 CrossRef CAS.
- B. Kesteleyn, K. Amssoms, W. Schepens, G. Hache, W. Verschueren, W. Van De Vreken, K. Rombauts, G. Meurs, P. Sterkens, B. Stoops, L. Baert, N. Austin, J. Wegner, C. Masungi, I. Dierynck, S. Lundgren, D. Jönsson, K. Parkes, G. Kalayanov, H. Wallberg, Å. Rosenquist, B. Samuelsson, K. Van Emelen and J. W. Thuring, Bioorg. Med. Chem. Lett., 2013, 23, 310–317 CrossRef CAS.
- G. Shen, Z. Li, L. Zhao, Y. Zhang, Y. Zhang, Z. Li, X. Huang and X. Lv, Chem. – Eur. J., 2024, 30, e202401011 CrossRef CAS.
- S. Keskin and M. Balci, Org. Lett., 2015, 17, 964–967 CrossRef CAS PubMed.
- A. Migani, P. L. Gentili, F. Negri, M. Olivucci, A. Romani, G. Favaro and R. S. Becker, J. Phys. Chem. A, 2005, 109, 8684–8692 CrossRef CAS.
- J. M. Gil-Negrete, J. P. Sestelo and L. A. Sarandeses, Org. Lett., 2016, 18, 4316–4319 Search PubMed.
- A. Kondoh and M. Terada, Chem. – Eur. J., 2022, 28, e202201198 CrossRef CAS PubMed.
- A. L. LaFrate, J. R. Gunther, K. E. Carlson and J. A. Katzenellenbogen, Bioorg. Med. Chem., 2008, 16, 10075–10084 Search PubMed.
- H. Y. Kim, J. Li and K. Oh, Angew. Chem., 2013, 125, 3824–3828 CrossRef.
- A. Alizadeh and A. Rostampoor, ChemistrySelect, 2022, 7, e202200299 CrossRef CAS.
- A. S. Komendantova, A. N. Fakhrutdinov, L. G. Menchikov, A. Yu. Sukhorukov, I. V. Zavarzin and Y. A. Volkova, Eur. J. Org. Chem., 2019, 527–536 CrossRef CAS.
- S. Mandal, S. Mandal, S. K. Ghosh, A. Ghosh, R. Saha, S. Banerjee and B. Saha, Synth. Commun., 2016, 46, 1327–1342 CrossRef CAS.
- G. D. Yadav and D. P. Wagh, ChemistrySelect, 2020, 5, 9059–9085 CrossRef CAS.
- C. Liu, H. Zhang, W. Shi and A. Lei, Chem. Rev., 2011, 111, 1780–1824 CrossRef CAS.
- B. Luo, W. Dong, Q. Ma, H. Yang and W. Tang, Org. Lett., 2024, 26, 8736–8740 Search PubMed.
- S. Zhang, X. Liu and T. Wang, Adv. Synth. Catal., 2011, 353, 1463–1466 CrossRef CAS.
- M. Somasundaram, J. A. Garg, S. Naidu, V. Ramkumar, R. Saiganesh, S. Kabilan and K. K. Balasubramanian, Eur. J. Org. Chem., 2019, 6269–6277 CrossRef CAS.
- Z. Zhou, Z.-X. Wang, Y.-C. Zhou, W. Xiao, Q. Ouyang, W. Du and Y.-C. Chen, Nat. Chem., 2017, 9, 590–594 CrossRef CAS PubMed.
- A. Thakur and J. Louie, Acc. Chem. Res., 2015, 48, 2354–2365 Search PubMed.
- D. G. Hall, T. Rybak and T. Verdelet, Acc. Chem. Res., 2016, 49, 2489–2500 CrossRef CAS PubMed.
- J. Fidelius, K. Schwedtmann, S. Schellhammer, J. Haberstroh, S. Schulz, R. Huang, M. C. Klotzsche, A. Bauzá, A. Frontera, S. Reineke and J. J. Weigand, Chem, 2024, 10, 644–659 CAS.
- G. Delaittre, N. K. Guimard and C. Barner-Kowollik, Acc. Chem. Res., 2015, 48, 1296–1307 CrossRef CAS.
- K. M. Schelkle, M. Bender, S. Beck, K. F. Jeltsch, S. Stolz, J. Zimmermann, R. T. Weitz, A. Pucci, K. Müllen, M. Hamburger and U. H. F. Bunz, Macromolecules, 2016, 49, 1518–1522 CrossRef CAS.
- P. Karthikeyan, A. M. Rani, R. Saiganesh, K. K. Balasubramanian and S. Kabilan, Tetrahedron, 2009, 65, 811–821 CrossRef CAS.
- B. Muralidhar and S. R. Reddy, ChemistrySelect, 2017, 2, 2539–2543 Search PubMed.
- B. Muralidhar and S. R. Reddy, Bull. Chem. Soc. Jpn., 2018, 91, 65–70 CrossRef CAS.
- B. Muralidhar, G. G. Victoria, K. S. Kumar and S. R. Reddy, Asian J. Org. Chem., 2022, 11, e202200044 CrossRef CAS.
- S. Madhunala, S. H. Kurma, V. R. Etikala and C. R. Bhimapaka, Synth. Commun., 2019, 49, 2538–2545 Search PubMed.
- P. Panjacharam and S. R. Reddy, Asian J. Org. Chem., 2025, e70106 CrossRef CAS.
- A. Ghatak, A. Pramanik and M. Das, Tetrahedron, 2022, 106–107, 132628 Search PubMed.
- B. S. Panda, B. Samanta, S. S. S. S. S. Ambadipudi, S. Nayak, V. L. Nayak, S. Ramakrishna, S. Mohapatra, P. M. Behera and L. Samanta, ChemistrySelect, 2024, 9, e202400115 Search PubMed.
- N. P. Mishra, S. Mohapatra, C. R. Sahoo, B. P. Raiguru, S. Nayak, S. Jena and R. N. Padhy, J. Mol. Struct., 2021, 1246, 131183 CrossRef CAS.
- V. T. Angelova, N. G. Vassilev, B. Nikolova-Mladenova, J. Vitas, R. Malbaša, G. Momekov, M. Djukic and L. Saso, Med. Chem. Res., 2016, 25, 2082–2092 Search PubMed.
- P. I. C. Godinho, P. Pérez-Ramos, Y. Gabasa, E. Cornielle, S. M. Soto, R. G. Soengas and A. M. S. Silva, Antibiotics, 2025, 14, 218 Search PubMed.
- V. Raj and J. Lee, Front. Chem., 2020, 8, 623 Search PubMed.
- N. J. P. Subhashini, M. Ravi, D. Cherupally, B. C. Raju, E. V. Reddy and H. Bee, Russ. J. Gen. Chem., 2016, 86, 2900–2905 CrossRef CAS.
- B. B. Shivaraj, K. S. Hariprasad, B. B. Rathod, R. S. Prakasham and B. C. Raju, Indian J. Chem., Sect. B: Org. Chem. Incl. Med. Chem., 2019, 58, 497–503 Search PubMed.
- R. Dasari, G. Thalari, J. R. Yerrabelly and P. R. Chitneni, Asian J. Chem., 2021, 33, 1723–1728 CAS.
- R. Dasari, G. Thalari, J. R. Yerrabelly and P. R. Chittneni, Russ. J. Org. Chem., 2021, 57, 1519–1532 Search PubMed.
- S. Rahmani-Nezhad, M. Safavi, M. Pordeli, S. K. Ardestani, L. Khosravani, Y. Pourshojaei, M. Mahdavi, S. Emami, A. Foroumadi and A. Shafiee, Eur. J. Med. Chem., 2014, 86, 562–569 Search PubMed.
- G. Gudipudi, S. R. Sagurthi, S. Perugu, G. Achaiah and G. L. D. Krupadanam, RSC Adv., 2014, 4, 56489–56501 RSC.
- L. Tao, S. Ji, D. Szalóki, T. Kovács, A. Mándi, S. Antus, X. Ding, T. Kurtán and H. Zhang, Acta Pharmacol. Sin., 2021, 42, 36–44 CrossRef CAS PubMed; X. Lu, Y. Teng, X. Lin, M. Xiao, C. Liu, X. Chi, Y. Zhang, G. Luo and H. Xiang, Bioorg. Chem., 2021, 109, 104714 CrossRef.
- N. Balakrishnan, J. Haribabu, M. Dharmasivam, S. Swaminathan and R. Karvembu, Appl. Organomet. Chem., 2022, 36, e6750 Search PubMed.
- J. Haribabu, R. Arulkumar, D. Mahendiran, K. Jeyalakshmi, S. Swaminathan, P. Venuvanalingam, N. Bhuvanesh, J. F. Santibanez and R. Karvembu, Inorg. Chim. Acta, 2024, 565, 121973 CrossRef CAS.
- Y. J. Rao, E. P. Reddy, G. Thirupathi, E. Y. Goud, M. Sowjanya and Y. Hemasri, Russ. J. Gen. Chem., 2016, 86, 1730–1734 CrossRef CAS.
- A. S. Kumar, R. A. Kumar, V. Satyanarayana, E. P. Reddy, B. J. M. Reddy, D. N. Kumar, A. Khurana, G. Chandraiah and J. S. Yadav, Nat. Prod. Commun., 2017, 12, 1934578X1701200732 Search PubMed.
- R. Sultana and R. R. Tippanna, Lab Chip, 2021, 18, 265–272 CAS.
- M. S. L. Kumar, J. Singh, S. K. Manna, S. Maji, R. Konwar and G. Panda, Bioorg. Med. Chem. Lett., 2018, 28, 778–782 CrossRef.
- Q.-W. Tan, L.-Y. He, Z.-W. He, W.-H. Liu, S.-S. Zhang, L. Lin, H.-L. Yang and L.-P. Guan, Med. Chem. Res., 2021, 30, 1427–1437 Search PubMed.
- D. Malafaia, A. Oliveira, P. A. Fernandes, M. J. Ramos, H. M. T. Albuquerque and A. M. S. Silva, Int. J. Mol. Sci., 2021, 22, 4145 Search PubMed.
- G. V. Govada and S. R Reddy, Chem. Biodivers., 2023, 20, e202300573 Search PubMed.
- G. V. Govada, H. Bhatt, P. Panjacharam, S. Pal, S. Kumar, C. Lin and S. R. Reddy, ChemistrySelect, 2024, 9, e202401348 CrossRef CAS.
- G. V. Govada, S. Pal, P. Panjacharam, H. S. Bhatt, S. Kumar, C. Lin, S. Wang and S. R. Reddy, Chem. Biodiversity, 2024, 21, e202400719 CrossRef CAS PubMed.
- Z.-Y. Fu, Q.-H. Jin, Y.-L. Qu and L.-P. Guan, Bioorg. Med. Chem. Lett., 2019, 29, 1909–1912 CrossRef CAS PubMed.
- S. H. Kurma, S. Karri, M. Kuncha, R. Sistla and C. R. Bhimapaka, Bioorg. Med. Chem. Lett., 2020, 30, 127341 CrossRef CAS PubMed.
- Y.-P. Zhang, Q.-H. Xue, Y.-S. Yang, X.-Y. Liu, C.-M. Ma, J.-X. Ru and H.-C. Guo, Inorg. Chim. Acta, 2018, 479, 128–134 Search PubMed.
- J. W. Lee, S. Bae and W. H. Jo, J. Mater. Chem. A, 2014, 2, 14146–14153 RSC.
- Y. Sun, S. Xu, H. Hang, J. Xi, H. Dong, B. Jiao, G. Zhou and X. Yang, Chem. Sci., 2024, 15, 8506–8513 RSC.
- J. Kim and I. Kim, Org. Biomol. Chem., 2018, 16, 89–100 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |