Cyclic azole-homologated peptides from Marine sponges
Abstract
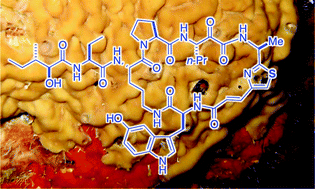
This review discusses the chemistry of cyclic azole-homologated peptides (AHPs) from the marine sponges, Theonella swinhoei, other Theonella species, Calyx spp. and Plakina jamaicensis. The origin, distribution of AHPs and molecular structure elucidations of AHPs are described followed by their biosynthesis, bioactivity, and synthetic efforts towards their total synthesis. Reports of partial and total synthesis of AHPs extend beyond peptide coupling reactions and include creative construction of the non-proteinogenic amino acid components, mainly the homologated heteroaromatic and α-keto-β-amino acids. A useful conclusion is drawn regarding AHPs: despite their rarity, exotic structures and the potent protease inhibitory properties of some members, their synthesis is under-developed and beckons solutions for outstanding problems towards their efficient assembly.


 Please wait while we load your content...
Please wait while we load your content...