Creating functional engineered variants of the single-module non-ribosomal peptide synthetase IndC by T domain exchange†‡
Abstract
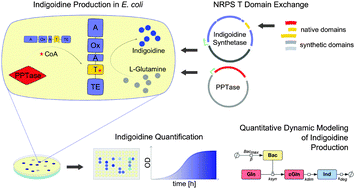
Non-ribosomal peptide synthetases (NRPSs) are enzymes that catalyze ribosome-independent production of small peptides, most of which are bioactive. NRPSs act as peptide assembly lines where individual, often interconnected modules each incorporate a specific amino acid into the nascent chain. The modules themselves consist of several domains that function in the activation, modification and condensation of the substrate. NRPSs are evidently modular, yet experimental proof of the ability to engineer desired permutations of domains and modules is still sought. Here, we use a synthetic-biology approach to create a small library of engineered NRPSs, in which the domain responsible for carrying the activated amino acid (T domain) is exchanged with natural or synthetic T domains. As a model system, we employ the single-module NRPS IndC from Photorhabdus luminescens that produces the blue pigment indigoidine. As chassis we use Escherichia coli. We demonstrate that heterologous T domain exchange is possible, even for T domains derived from different organisms. Interestingly, substitution of the native T domain with a synthetic one enhanced indigoidine production. Moreover, we show that selection of appropriate inter-domain linker regions is critical for functionality. Taken together, our results extend the engineering avenues for NRPSs, as they point out the possibility of combining domain sequences coming from different pathways, organisms or from conservation criteria. Moreover, our data suggest that NRPSs can be rationally engineered to control the level of production of the corresponding peptides. This could have important implications for industrial and medical applications.
- This article is part of the themed collection: 2014 Emerging Investigators

 Please wait while we load your content...
Please wait while we load your content...