Bismuth-based cyclic synthesis of 3,5-di-tert-butyl-4-hydroxybenzoic acid via the oxyarylcarboxy dianion, (O2CC6H2tBu2O)2−†
Abstract
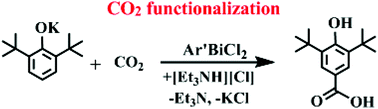
3,5-Di-tert-butyl-4-hydroxybenzoic acid can be made under mild conditions in a cyclic process from carbon dioxide and 3,5-di-tert-butyl-4-phenol using bismuth-based C–H bond activation and CO2 insertion chemistry starting with the Bi3+ complex, Ar′BiCl2, of the NCN pincer ligand, Ar′ = 2,6-(Me2NCH2)2C6H3. Complexes of the recently discovered oxyaryl dianion, (C6H2tBu2-3,5-O-4)2−, and the oxyarylcarboxy dianion, [O2C(C6H2tBu2-3,5-O-4)]2−, are intermediates in the process. Further studies of the oxyarylcarboxy dianion in Ar′Bi[O2C(C6H2tBu2-3,5-O-4)-κ2O,O′], show that it undergoes decarboxylation upon reaction with I2 and it reacts with trimethylsilyl chloride to produce the trimethylsilyl ether of the trimethylsilyl ester of 3,5-di-tert-butyl-4-hydroxybenzoic acid and the Ar′BiCl2 starting material.

 Please wait while we load your content...
Please wait while we load your content...