Enantio- and diastereoselective palladium catalysed arylative and vinylative allene carbocyclisation cascades†
Abstract
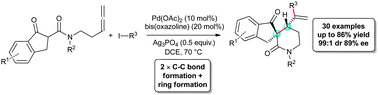
The enantioselective synthesis of heavily decorated spirolactams has been accomplished via an arylative or vinylative
* Corresponding authors
a
Department of Chemistry, Chemistry Research Laboratory, University of Oxford, 12 Mansfield Road, Oxford, OX1 3TA, UK
E-mail:
darren.dixon@chem.ox.ac.uk
b Department of Inorganic and Physical Chemistry, Ghent University, Krijgslaan 281-S3, 9000 Gent, Belgium
c Department of Chemistry, University of Antwerp, Groenenborgerlaan 171, 2020 Antwerp, Belgium
The enantioselective synthesis of heavily decorated spirolactams has been accomplished via an arylative or vinylative

 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?
M. Li, A. Hawkins, D. M. Barber, P. Bultinck, W. Herrebout and D. J. Dixon, Chem. Commun., 2013, 49, 5265 DOI: 10.1039/C3CC42079E
To request permission to reproduce material from this article, please go to the Copyright Clearance Center request page.
If you are an author contributing to an RSC publication, you do not need to request permission provided correct acknowledgement is given.
If you are the author of this article, you do not need to request permission to reproduce figures and diagrams provided correct acknowledgement is given. If you want to reproduce the whole article in a third-party publication (excluding your thesis/dissertation for which permission is not required) please go to the Copyright Clearance Center request page.
Read more about how to correctly acknowledge RSC content.
 Fetching data from CrossRef.
Fetching data from CrossRef.
This may take some time to load.
Loading related content
