Ultrahigh yield of hydrogen peroxide on graphite felt cathode modified with electrochemically exfoliated graphene
Abstract
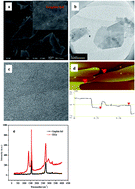
The development of an efficient oxygen reduction reaction (ORR) cathode for hydrogen peroxide production represents an important challenge in the field of electrochemical processes and is highly demanded for chemical industries and environmental remediation applications. In this work, a novel graphite felt cathode modified with electrochemically exfoliated graphene (EEGr) and carbon black was developed, and it presented a very high H2O2 generation rate of 7.7 mg h−1 cm−2 with a relatively low energy consumption (9.7 kW h kg−1). Characterized by SEM, TEM, AFM, Raman, XRD, and XPS, the synthesized EEGr was proven to be a 3–4 layers thin sheet with low defects. Important cathode manufacture parameters, including the ratio and loading of EEGr, were optimized, and the dependence of H2O2 generation with the pH and cathode potential as well as the performance stability were investigated. At an optimized cathode potential of −0.9 V and pH 7, the modified cathode kept a stable performance for H2O2 generation during 10 cycles, which was 2 times that of the cathode without EEGr. Further explored by CV, rotating disk electrode (RDE), and contact angles analysis, the presence of EEGr was found to accelerate the electron transfer rate and benefit the oxygen surface reaction, but not change the two-electrons ORR number, which contributed to the enhanced performance for H2O2 production, and a possible mechanism is suggested. Finally, such a graphene-modified cathode demonstrated effectiveness for the degradation of four kinds of representative pollutants (orange II, methylene blue, phenol, and sulfadiazine) by an electro-Fenton process, proving it has great potential practical application for organic wastewater treatment.


 Please wait while we load your content...
Please wait while we load your content...