Artificially engineered, bicontinuous anion-conducting/-repelling polymeric phases as a selective ion transport channel for rechargeable zinc–air battery separator membranes†
Abstract
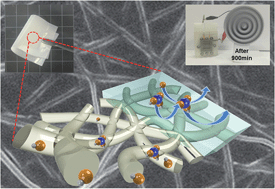
Zinc (Zn)–air batteries have recently attracted a great deal of attention as a promising energy storage system to fulfill our ever-increasing demand for higher energy density power sources. Despite commercial success of primary Zn–air batteries, performances of rechargeable Zn–air batteries are still far below practically satisfactory levels. Among critical challenges facing the electrochemical rechargeability, the crossover of zincate (Zn(OH)42−) ions from the Zn anode to the air cathode (via separator membranes) is a formidable bottleneck. Here, as a facile and scalable polymer architecture strategy to address this ion transport issue, we demonstrate a new class of polymer blend electrolyte membranes with artificially engineered, bicontinuous anion-conducting/-repelling phases (referred to as “PBE membranes”). As an anion-conducting continuous phase, an electrospun polyvinyl alcohol (PVA)/polyacrylic acid (PAA) nanofiber mat is fabricated. Into the PVA/PAA nanofiber mat, Nafion bearing pendant sulfonate groups is impregnated to form an anion-repelling continuous phase. Such bicontinuous phase-mediated structural uniqueness enables the PBE membrane to act as a selective ion transport channel, i.e., effectively suppresses Zn(OH)42− crossover (by a continuous Nafion phase offering the Donnan exclusion effect) with slightly impairing OH− conduction (predominantly through the PVA/PAA nanofiber mat), eventually improving the cycling stability (cycle time = over 2500 min for the PBE membrane vs. 900 min for a conventional polypropylene separator). The PBE membrane featuring the selective transport of OH− and Zn(OH)42− ions is anticipated to pave a new route that leads us closer toward rechargeable Zn–air batteries.

 Please wait while we load your content...
Please wait while we load your content...