Direct C–C coupling of bio-ethanol into 2,3-butanediol by photochemical and photocatalytic oxidation with hydrogen peroxide†
Abstract
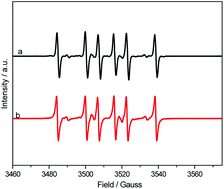
Theoretically, selective C–H manipulation in ethanol can result in a direct C–C coupling synthesis of 2,3-butanediol (2,3-BDO). However, this process is typically extremely difficult to achieve because of the high complexity of the involved chemical bonds. In this work, we determine that hydroxide radicals generated from the photolysis of H2O2 can selectively attack the α-hydrogen atom in ethanol aqueous solutions and crack the C–H bond to produce hydroxyethyl radicals, which subsequently undergo C–C coupling to form 2,3-BDO. This selective C–H breakage is determined by the reaction rate, which is primarily controlled by the local H2O2 concentration at a given irradiation intensity. At a moderate reaction rate of ethanol (37 mmol h−1), the 2,3-BDO selectivity reaching as high as 91% can be obtained. The introduction of a catalyst can further increase ethanol conversion and enhance the 2,3-BDO formation rate by controlling the reaction rate. This result provides an environment-friendly approach to convert bio-ethanol directly to 2,3-BDO and to manipulate a single bond selectively in complex bonding situations.

 Please wait while we load your content...
Please wait while we load your content...