Sub-optimal phenotypes of double-knockout mutants of Escherichia coli depend on the order of gene deletions†
Abstract
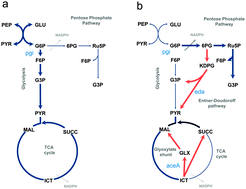
Metabolic networks are characterized by multiple redundant reactions that do not have a clear biological function. The redundancies in the metabolic networks are implicated in adaptation to random mutations and survival under different environmental conditions. Reactions that are not active under wild-type growth conditions, but get transiently activated after a mutation event such as gene deletion are known as latent reactions. Characterization of multiple-gene knockout mutants can identify the physiological roles of latent reactions. In this study, we characterized double-gene deletion mutants of E. coli with the aim of investigating the sub-optimal physiology of the mutants and the possible roles of latent reactions. Specifically, we investigated the effects of the deletion of the glyoxylate-shunt gene aceA (encoding a latent reaction enzyme, isocitrate lyase) on the growth characteristics of the mutant E. coli Δpgi. The deletion of aceA reduced the growth rate of E. coli Δpgi, indicating that the activation of the glyoxylate shunt plays an important role in adaptation of the mutant E. coli Δpgi when no other latent reactions are concurrently inactivated. We also investigated the effect of the order of the gene deletions on the growth rates and substrate uptake rates of the double-gene deletion mutants. The results indicate that the order in which genes are deleted determines the phenotype of the mutants during the sub-optimal growth phase. To elucidate the mechanism behind the difference between the observed phenotypes, we carried out transcriptomic analysis and constraint-based modeling of the mutants. Transcriptomic analysis showed differential expression of the gene aceK (encoding the protein isocitrate dehydrogenase kinase) involved in controlling the isocitrate flux through the TCA cycle and the glyoxylate shunt. Higher acetate production in the E. coli ΔaceA1 Δpgi2 mutant was consistent with the increased aceK expression, which limits the TCA cycle flux and causes acetate production via overflow metabolism.
- This article is part of the themed collection: Integrative Approaches for Signalling and Metabolic Networks

 Please wait while we load your content...
Please wait while we load your content...