Understanding selective molecular recognition in integrated carbon nanotube–polymer sensors by simulating physical analyte binding on carbon nanotube–polymer scaffolds†
Abstract
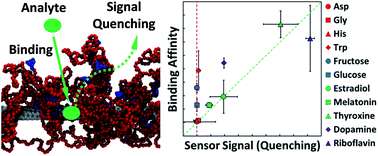
Macromolecular scaffolds made of polymer-wrapped single-walled carbon nanotubes (SWCNTs) have been explored recently (Zhang et al., Nature Nanotechnology, 2013) as a new class of molecular-recognition motifs. However, selective analyte recognition is still challenging and lacks the underlying fundamental understanding needed for its practical implementation in biological sensors. In this report, we combine coarse-grained molecular dynamics (CGMD) simulations, physical adsorption/binding theories, and photoluminescence (PL) experiments to provide molecular insight into the selectivity of such sensors towards a large set of biologically important analytes. We find that the physical binding affinities of the analytes on a bare SWCNT partially correlate with their distribution coefficients in a bulk water/octanol system, suggesting that the analyte hydrophobicity plays a key role in determining the binding affinities of the analytes considered, along with the various specific interactions between the analytes and the polymer anchor groups. Two distinct categories of analytes are identified to demonstrate a complex picture for the correlation between optical sensor signals and the simulated binding affinities. Specifically, a good correlation was found between the sensor signals and the physical binding affinities of the three hormones (estradiol, melatonin, and thyroxine), the neurotransmitter (dopamine), and the vitamin (riboflavin) to the SWCNT–polymer scaffold. The four amino acids (aspartate, glycine, histidine, and tryptophan) and the two monosaccharides (fructose and glucose) considered were identified as blank analytes which are unable to induce sensor signals. The results indicate great success of our physical adsorption-based model in explaining the ranking in sensor selectivities. The combined framework presented here can be used to screen and select polymers that can potentially be used for creating synthetic molecular recognition motifs.

 Please wait while we load your content...
Please wait while we load your content...