Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Metalloradical activation of α-formyldiazoacetates for the catalytic asymmetric radical cyclopropanation of alkenes†
Xue
Xu
b,
Yong
Wang
 a,
Xin
Cui
b,
Lukasz
Wojtas
b and
X. Peter
Zhang
a,
Xin
Cui
b,
Lukasz
Wojtas
b and
X. Peter
Zhang
 *ab
*ab
aDepartment of Chemistry, Merkert Chemistry Center, Boston College, Chestnut Hill, Massachusetts 02467, USA. E-mail: peter.zhang@bc.edu
bDepartment of Chemistry, University of South Florida, Tampa, FL 33620, USA
First published on 31st March 2017
Abstract
For the first time, α-formyldiazoacetates have been successfully applied for the asymmetric cyclopropanation of alkenes via Co(II)-based metalloradical catalysis. The cobalt(II) complex of the D2-symmetric chiral amidoporphyrin [Co(3,5-DitBu-ChenPhyrin)] is an effective metalloradical catalyst that can activate α-formyldiazoacetates to cyclopropanate both aromatic and aliphatic olefins with varied electronic properties, affording the synthetically useful 1,1-cyclopropaneformylesters in high yields with both high diastereo- and enantioselectivity.
Introduction
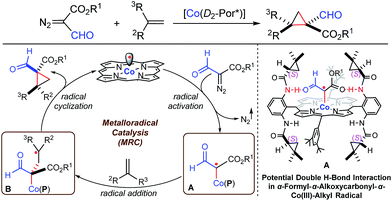
Radical reactions have been increasingly exploited as attractive tools in modern organic synthesis as they exhibit a number of unique features, including tolerance of functional groups.1 To address the existing challenges such as the control of enantioselectivity,2 metalloradical catalysis (MRC) has rendered a fundamentally new approach that enables the catalytic generation of metal-stabilized organic radicals as well as the selective control of their subsequent radical reactions.3,4 As stable metalloradicals, cobalt(II) complexes of the D2-symmetric chiral amidoporphyrins [Co(D2-Por*)] have emerged as a class of effective catalysts for asymmetric olefin cyclopropanation through a distinct radical process involving the catalytic generation of α-metalloalkyl radicals as the key intermediates (Scheme 1: A).5 It has been suggested that the unusual capability of [Co(D2-Por*)] in activating acceptor/acceptor-substituted diazo reagents as well as regulating the reactivity and selectivity of the radical processes is further enhanced by the postulated double hydrogen-bonding interactions between the amide N–H donors on the amidoporphyrin ligand and the two acceptors on the C-centered radical moiety (Scheme 1).5e,g,h Considering the demonstrated functional group tolerance of Co(II)-based metalloradical catalysis (Co(II)-MRC),5d,e,j–l we were attracted to the possibility of accessing a new type of α-metalloalkyl radical bearing both α-formyl and α-alkoxycarbonyl functionalities from the metalloradical activation of α-formyldiazoacetates (FDA).6 Despite the fact that free α-formylalkyl radicals are scarce and prone to H-atom abstraction because of the weak aldehydic C–H bonds,7 we reasoned that this type of α-metalloalkyl radical might be accessible on the basis of the combined effects of metal stabilization, double H-bonding interaction, and protection by the well-defined cavity of the ligand system (Scheme 1: A). Assuming that the α-formyl-α-alkoxycarbonyl-α-Co(III)-alkyl radicals (A) are capable of undergoing stereoselective radical addition with olefins, followed by the effective 3-exo-tet radical cyclization8 of the corresponding γ-Co(III)-alkyl radicals (B), we anticipated the potential development of a new catalytic process for the asymmetric synthesis of optically active cyclopropanes bearing both aldehyde and ester functionalities, which would be valuable for stereoselective organic synthesis (Scheme 1).9The catalytic asymmetric cyclopropanation of alkenes with diazo reagents represents the most general approach for the stereoselective synthesis of optically active cyclopropanes.10 While a number of diazo reagents have been successfully employed, there is no previous report of a catalytic system that is effective for asymmetric olefin cyclopropanation with α-formyldiazoacetates (FDAs). The development of this catalytic process apparently confronts the formidable challenges associated with the inherent low reactivity of the acceptor/acceptor-substituted diazo reagents and the incompatibility of the aldehyde functionality with existing catalytic systems.11 Recently, Fokin and coworkers developed a Rh2-catalyzed system for a highly asymmetric cyclopropanation with N-sulfonyl-1,2,3-triazoles for the production of cyclopropyl imines, which could be subsequently transformed into the corresponding formyl cyclopropane derivatives.12 While this offers a valuable alternative for the preparation of optically active formyl cyclopropanes, the direct synthesis via asymmetric cyclopropanation with α-formyl diazo reagents is an appealing process that remains to be developed. As a new application of Co(II)-MRC, we herein wish to report the first catalytic system based on [Co(D2-Por*)] that is highly effective in activating FDA for asymmetric cyclopropanation. This asymmetric radical process is generally applicable for a broad scope of alkenes, offering a direct method for the high-yielding synthesis of 1,1-cyclopropaneformylesters with excellent control of the diastereo- and enantioselectivity. The products can be readily transformed into other chiral 1,1-bifunctionalized cyclopropanes and chiral dihydrofurans.
Results and discussion
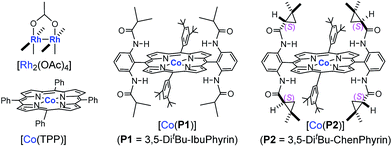
Initial experiments were carried out with styrene as the model substrate to examine the suitability of FDA for the catalytic radical cyclopropanation by Co(II)-MRC (Table 1). While [Rh2(OAc)4] was indeed incompatible (entry 1), [Co(TPP)] only produced trace amounts of the corresponding cyclopropane from ethyl α-formyldiazoacetate (EFDA) (entry 2). Remarkably, when the Co(II) complex of the D2h-symmetric achiral amidoporphyrin [Co(P1)]13 was used as the catalyst, the reaction proceeded successfully to form the desired (E)-1,1-cyclopropaneformylester in a 46% yield (entry 3). The dramatic difference in the catalytic activity between [Co(TPP)] and [Co(P1)] is in alignment with the hypothesized role of the double H-bonding interaction in activating EFDA and stabilizing the resulting intermediate A (Scheme 1). By switching to [Co(P2)],14 the reactivity was further enhanced with the observation of a significant level of enantioselectivity (entry 4). Of the solvents examined, toluene was proven to be the medium of choice (entries 4–8). Lowering the reaction temperature further increased the enantioselectivity, but decreased the yield (entries 8–10). The diastereoselectivity was greatly improved when the bulkier tert-butyl α-formyldiazoacetate (t-BFDA) was used, affording cyclopropane 1a in a 78% yield with 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 dr and 96% ee (entry 11). The product yield could be further improved to 84% by increasing the catalyst loading to 5 mol% while maintaining the high level of diastereo- and enantioselectivity (entry 12).
5 dr and 96% ee (entry 11). The product yield could be further improved to 84% by increasing the catalyst loading to 5 mol% while maintaining the high level of diastereo- and enantioselectivity (entry 12).
| Entry | Catalyst | R | Solvent | Temp. (°C) | Yieldb (%) | (E)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) (Z) (Z) |
eec (%) |
|---|---|---|---|---|---|---|---|
| a Carried out in one-portion under N2 with [olefin] = 0.20 M. b Isolated yields. c ee of major (E)-diastereomer determined by chiral HPLC. d Determined by 1H-NMR. e With 5 mol% of catalyst for 20 h. | |||||||
| 1 | [Rh2(OAc)4] | Et | DCM | 60 | 0 | — | — |
| 2 | [Co(TPP)] | Et | DCM | 60 | <10d | — | — |
| 3 | [Co(P1)] | Et | DCM | 60 | 46 | 84![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 16 16 |
— |
| 4 | [Co(P2)] | Et | DCM | 60 | 81 | 80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 20 |
81 |
| 5 | [Co(P2)] | Et | EtOAc | 60 | 81 | 80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 20 |
81 |
| 6 | [Co(P2)] | Et | PhCl | 60 | 73 | 82![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 18 18 |
83 |
| 7 | [Co(P2)] | Et | Hexanes | 60 | 73 | 80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 20 |
82 |
| 8 | [Co(P2)] | Et | Toluene | 60 | 86 | 85![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15 15 |
86 |
| 9 | [Co(P2)] | Et | Toluene | 40 | 73 | 86![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 14 14 |
90 |
| 10 | [Co(P2)] | Et | Toluene | RT | 60 | 87![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 13 13 |
93 |
| 11 | [Co(P2)] | t Bu | Toluene | 40 | 78 | 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 5 |
96 |
| 12 | [Co( P2 )] | t Bu | Toluene | 40 | 84 |
95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 5 |
96 |
|
|||||||
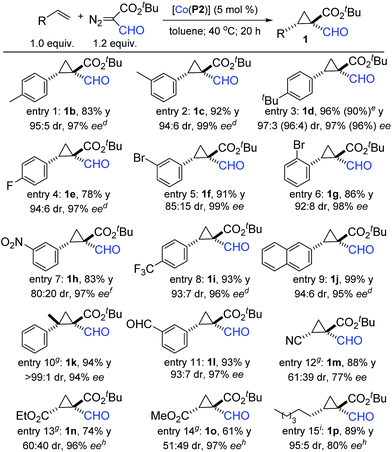
Under the optimized conditions, the scope of this Co(II)-based asymmetric radical cyclopropanation was investigated (Table 2). Like styrene, its derivatives bearing substituents with varied electronic and steric properties could be cyclopropanated by [Co(P2)] with t-BFDA. For example, p- and m-alkyl styrenes were cyclopropanated to formylcyclopropanes 1b–1d in high yields with excellent diastereo- and enantioselectivity (entries 1–3). Halogenated (entries 4–6) and electron-deficient (entries 7 and 8) styrene derivatives could also undergo high-yielding cyclopropanation, producing 1e–1i with high stereoselectivities. The configurations of the two contiguous chiral centers in 1h were established as [1R,2S] by X-ray crystal structural analysis (see ESI†). The cyclopropanation was also suitable for other aromatic olefins as exemplified with 2-naphthalene for near quantitative formation of cyclopropane 1j (entry 9). In addition, 1,1-disubstituted olefins such as α-methylstyrene could also be effectively employed, affording (E)-formylcyclopropane 1k in a 93% yield with remarkable control of both the diastereo- and enantioselectivity of the two newly-generated contiguous all-carbon quaternary stereogenic centers (entry 10). To demonstrate the functional group tolerance of the Co(II)-based radical cyclopropanation, m-formylstyrene could be effectively cyclopropanated to cyclopropane 1l in a high yield with high diastereo- and enantioselectivity (entry 11). Notably, the two unprotected formyl groups were well tolerated by the metalloradical system. It is also worth mentioning that the Co(II)-catalyzed cyclopropanation process could be scaled up ten-fold as demonstrated with the high-yielding synthesis of the cyclopropane 1d on a 1.0 mmol scale without affecting the excellent stereoselectivity (entry 3).
| a Carried out in one-portion under N2 with [olefin] = 0.20 M. b Isolated yields. c ee of major (E)-diastereomer determined by chiral HPLC. d ee determined upon derivatization. e Results in the parentheses were obtained for the reaction performed on a 1.0 mmol scale. f Absolute configuration determined by X-ray diffraction as [1R,2S]. g 5 equiv. olefin. h ee determined by chiral GC. i Neat condition. |
|---|
|
The Co(II)-based radical cyclopropanation was further highlighted for its exceptional reactivity toward electron-deficient olefins, which are typically problematic substrates for catalytic systems involving electrophilic metallocarbene intermediates. For example, [Co(P2)] could catalyze the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C cyclopropanation of acrylonitrile with t-BFDA to form 1,1,2-cyclopropaneformylesternitrile 1m in a high yield with good enantioselectivity (entry 12), leaving the cyano group untouched. In marked contrast, when treated with Rh2-based catalyst, acrylonitrile was previously shown to react with the C
C cyclopropanation of acrylonitrile with t-BFDA to form 1,1,2-cyclopropaneformylesternitrile 1m in a high yield with good enantioselectivity (entry 12), leaving the cyano group untouched. In marked contrast, when treated with Rh2-based catalyst, acrylonitrile was previously shown to react with the C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N bond of FDA to form oxazoles.15 Other electron-deficient olefins such as ethyl and methyl acrylates could also be cyclopropanated to form the 1,1,2-cyclopropaneformyldiesters 1n and 1o in good yields with 96% ee and 97% ee, respectively, although with diminished control of diastereoselectivity (entries 13 and 14). The presence of three electron-withdrawing groups in the cyclopropanes 1m–1o renders them highly electrophilic, making them valuable intermediates for synthetic applications.16 Furthermore, aliphatic olefins, another class of challenging substrate for asymmetric cyclopropanation, could also be cyclopropanated by [Co(P2)] as exemplified by the high-yielding reaction of 1-octene under neat condition, forming 1p with high stereoselectivity (entry 15).
N bond of FDA to form oxazoles.15 Other electron-deficient olefins such as ethyl and methyl acrylates could also be cyclopropanated to form the 1,1,2-cyclopropaneformyldiesters 1n and 1o in good yields with 96% ee and 97% ee, respectively, although with diminished control of diastereoselectivity (entries 13 and 14). The presence of three electron-withdrawing groups in the cyclopropanes 1m–1o renders them highly electrophilic, making them valuable intermediates for synthetic applications.16 Furthermore, aliphatic olefins, another class of challenging substrate for asymmetric cyclopropanation, could also be cyclopropanated by [Co(P2)] as exemplified by the high-yielding reaction of 1-octene under neat condition, forming 1p with high stereoselectivity (entry 15).
As an initial exploration of applications, the formyl unit of the resulting chiral 1,1-cyclopropaneformylesters could be readily converted into other functional groups, forming various cyclopropane derivatives while retaining high enantiopurity. For example, the formyl group in (E)-1a could be transformed into a trans-vinyl unit via the Horner–Wadsworth–Emmons reaction, affording (E)-1,1-cyclopropanevinylester 2 in a 78% yield with full retention of both the relative and absolute configurations (eqn (1)). When treated with Bestmann–Ohira reagent, the formyl group in (E)-1a could be smoothly converted to a terminal alkyne functionality, resulting in chiral (E)-1,1-cyclopropaneethynylester 3 in a 70% yield without any diminishment of the original stereochemistry (eqn (2)). This transformation provides an alternative way to direct asymmetric cyclopropanation with α-ethynyldiazoacetates for chiral 1,1-cyclopropaneethynylesters.17 It is noted that α-ethynyldiazoacetates containing terminal alkyne units seem synthetically inaccessible. While the [Co(P2)]-catalyzed cyclopropanation with FDA generally forms (E)-cyclopropanes, the (Z)-diastereoisomers could be conveniently accessed through the stereospecific epimerization previously reported.5e As demonstrated with (E)-1a, treatment with 5 equivalents of NaI at room temperature resulted in the formation of (Z)-1a as the major diastereomer with only partial loss of the original optical purity (eqn (3)). Interestingly, when (E)-1g was treated with 10 equivalents of NaI at an elevated temperature, a ring-expansion involving the formyl group occurred instead, affording 2,3-dihydrofuran 4 in a 74% yield (eqn (4)). In the absence of any external chiral induction, the enantiopurity appeared to be largely retained during the rearrangement.
 | (1) |
 | (2) |
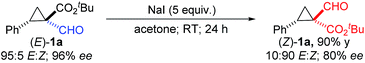 | (3) |
 | (4) |
Conclusions
In summary, we have demonstrated that the metalloradical catalyst [Co(P2)] can effectively activate α-formyldiazoacetates (FDAs) for a highly asymmetric olefin cyclopropanation, without affecting the otherwise reactive aldehyde functionality. This represents the first application of α-formyldiazo reagents for metal-catalyzed asymmetric cyclopropanation. The Co(II)-based radical cyclopropanation with FDA can be successfully applied to a broad scope of olefin substrates, permitting the direct synthesis of chiral 1,1-cyclopropaneformylesters in high yields with high diastereo- and enantioselectivity. Given that the resulting enantioenriched cyclopropanes contain two contiguous chiral centers in the ring structure, including one all-carbon quaternary stereogenic center bearing both aldehyde and ester functionalities, this new Co(II)-based asymmetric radical cyclopropanation process should find wide applications in stereoselective organic synthesis.Acknowledgements
We are grateful for financial support from the NIH (R01-GM102554).References
- (a) B. Quiclet-Sire and S. Z. Zard, Pure Appl. Chem., 2011, 83, 519–551 CAS; (b) M. H. Shaw, J. Twilton and D. W. C. MacMillan, J. Org. Chem., 2016, 81, 6898–6926 CrossRef CAS PubMed; (c) M. Yan, J. C. Lo, J. T. Edwards and P. S. Baran, J. Am. Chem. Soc., 2016, 138, 12692–12714 CrossRef CAS PubMed.
- For selected examples of recent approaches to control enantioselectivity of radical reactions, see: (a) Q. M. Kainz, C. D. Matier, A. Bartoszewicz, S. L. Zultanski, J. C. Peters and G. C. Fu, Science, 2016, 351, 681–684 CrossRef CAS PubMed; (b) J. L. Jeffrey, J. A. Terrett and D. W. C. MacMillan, Science, 2015, 349, 1532–1536 CrossRef CAS PubMed; (c) J. M. Hoyt, V. A. Schmidt, A. M. Tondreau and P. J. Chirik, Science, 2015, 349, 960–963 CrossRef CAS PubMed; (d) J. N. Du, K. L. Skubi, D. M. Schultz and T. P. Yoon, Science, 2014, 344, 392–396 CrossRef CAS PubMed; (e) N. Funken, F. Mühlhaus and A. Gansäuer, Angew. Chem., Int. Ed., 2016, 55, 12030–12034 CrossRef CAS PubMed.
- For reviews and highlights on Co(II)-MRC, see: (a) A. Studer and D. P. Curran, Angew. Chem., Int. Ed. Engl., 2016, 55, 58–102 CrossRef CAS PubMed; (b) H. Pellissier and H. Clavier, Chem. Rev., 2014, 114, 2775–2823 CrossRef CAS PubMed; (c) A. I. Olivos Suarez, V. Lyaskovskyy, J. N. Reek, J. I. van der Vlugt and B. de Bruin, Angew. Chem., Int. Ed. Engl., 2013, 52, 12510–12529 CrossRef PubMed; (d) L. Degennaro, P. Trinchera and R. Luisi, Chem. Rev., 2014, 114, 7881–7929 CrossRef CAS PubMed; (e) H. Lu and X. P. Zhang, Chem. Soc. Rev., 2011, 40, 1899–1909 RSC; (f) M. P. Doyle, Angew. Chem., Int. Ed. Engl., 2009, 48, 850–852 CrossRef CAS PubMed.
- For reviews and examples of Ti(III)-based MRC, see: (a) A. Gansauer, S. Hildebrandt, E. Vogelsang and R. A. Flowers Ii, Dalton Trans., 2016, 45, 448–452 RSC; (b) W. A. Nugent and T. V. RajanBabu, J. Am. Chem. Soc., 1988, 110, 8561–8562 CrossRef CAS; (c) A. Gansäuer, B. Rinker, M. Pierobon, S. Grimme, M. Gerenkamp and C. Mück-Lichtenfeld, Angew. Chem., Int. Ed., 2003, 42, 3687–3690 CrossRef PubMed; (d) A. Gansäuer, A. Fleckhaus, M. A. Lafont, A. Okkel, K. Kotsis, A. Anoop and F. Neese, J. Am. Chem. Soc., 2009, 131, 16989–16999 CrossRef PubMed; (e) A. Gansäuer, S. Hildebrandt, A. Michelmann, T. Dahmen, D. von Laufenberg, C. Kube, G. D. Fianu and R. A. Flowers, Angew. Chem., Int. Ed., 2015, 54, 7003–7006 CrossRef PubMed.
- For detailed studies on the involvement of α-Co(III)-alkyl radical intermediates (also known as Co(III)-carbene radicals) for catalytic cyclopropanation, see: (a) H. J. Lu, W. I. Dzik, X. Xu, L. Wojtas, B. de Bruin and X. P. Zhang, J. Am. Chem. Soc., 2011, 133, 8518–8521 CrossRef CAS PubMed; (b) J. L. Belof, C. R. Cioce, X. Xu, X. P. Zhang, B. Space and H. L. Woodcock, Organometallics, 2011, 30, 2739–2746 CrossRef CAS PubMed; (c) W. I. Dzik, X. Xu, X. P. Zhang, J. N. H. Reek and B. de Bruin, J. Am. Chem. Soc., 2010, 132, 10891–10902 CrossRef CAS PubMed; For selected examples of asymmetric radical cyclopropanation and related processes via Co(II)-MRC, see: (d) Y. Wang, X. Wen, X. Cui, L. Wojtas and X. P. Zhang, J. Am. Chem. Soc., 2017, 139, 1049–1052 CrossRef CAS PubMed; (e) X. Xu, S. F. Zhu, X. Cui, L. Wojtas and X. P. Zhang, Angew. Chem., Int. Ed., 2013, 52, 11857–11861 CrossRef CAS PubMed; (f) X. Xu, H. J. Lu, J. V. Ruppel, X. Cui, S. L. de Mesa, L. Wojtas and X. P. Zhang, J. Am. Chem. Soc., 2011, 133, 15292–15295 CrossRef CAS PubMed; (g) S. F. Zhu, X. Xu, J. A. Perman and X. P. Zhang, J. Am. Chem. Soc., 2010, 132, 12796–12799 CrossRef CAS PubMed; (h) S. F. Zhu, J. A. Perman and X. P. Zhang, Angew. Chem., Int. Ed., 2008, 47, 8460–8463 CrossRef CAS PubMed; (i) S. F. Zhu, J. V. Ruppel, H. J. Lu, L. Wojtas and X. P. Zhang, J. Am. Chem. Soc., 2008, 130, 5042–5043 CrossRef CAS PubMed; (j) Y. Chen, J. V. Ruppel and X. P. Zhang, J. Am. Chem. Soc., 2007, 129, 12074–12075 CrossRef CAS PubMed; (k) X. Cui, X. Xu, L. Wojtas, M. M. Kim and X. P. Zhang, J. Am. Chem. Soc., 2012, 134, 19981–19984 CrossRef CAS PubMed; (l) X. Cui, X. Xu, H. J. Lu, S. F. Zhu, L. Wojtas and X. P. Zhang, J. Am. Chem. Soc., 2011, 133, 3304–3307 CrossRef CAS PubMed.
- FDAs are a class of commonly used α-formyldiazo reagent due to their easy access and relative stability: (a) O. Sezer and O. Anac, Helv. Chim. Acta, 1994, 77, 2323–2334 CrossRef CAS; (b) O. Sezer, K. Dabak, O. Anac and A. Akar, Helv. Chim. Acta, 1997, 80, 960–965 CrossRef CAS.
- G. G. Melikyan, in Organic Reactions, John Wiley & Sons, Inc., 1997, vol. 49, pp. 427–675 Search PubMed.
- For select examples of 3-exo-tet radical cyclization, see: (a) A. M. del Hoyo and M. García Suero, Eur. J. Org. Chem., 2017 DOI:10.1002/ejoc.201601604; (b) A. M. del Hoyo, A. G. Herraiz and M. G. Suero, Angew. Chem., Int. Ed., 2017, 56, 1610–1613 CrossRef CAS PubMed; (c) T. Ohkita, Y. Tsuchiya and H. Togo, Tetrahedron, 2008, 64, 7247–7251 CrossRef CAS; (d) D. P. Curran and A. E. Gabarda, Tetrahedron, 1999, 55, 3327–3336 CrossRef CAS.
- (a) J. W. Bode and S. S. Sohn, J. Am. Chem. Soc., 2007, 129, 13798–13799 CrossRef CAS PubMed; (b) G. Q. Li, L. X. Dai and S. L. You, Org. Lett., 2009, 11, 1623–1625 CrossRef CAS PubMed; (c) H. Lv, J. M. Mo, X. Q. Fang and Y. R. Chi, Org. Lett., 2011, 13, 5366–5369 CrossRef CAS PubMed; (d) S. S. Sohn and J. W. Bode, Angew. Chem., Int. Ed., 2006, 45, 6021–6024 CrossRef CAS PubMed; (e) X. Y. Tang, Y. Wei and M. Shi, Org. Lett., 2010, 12, 5120–5123 CrossRef CAS PubMed; (f) S. Kalidindi, W. B. Jeong, A. Schall, R. Bandichhor, B. Nosse and O. Reiser, Angew. Chem., Int. Ed., 2007, 46, 6361–6363 CrossRef CAS PubMed; (g) B. M. Trost, Y. Hu and D. B. Horne, J. Am. Chem. Soc., 2007, 129, 11781–11790 CrossRef CAS PubMed; (h) J. D. White, W. H. C. Martin, C. Lincoln and J. Yang, Org. Lett., 2007, 9, 3481–3483 CrossRef CAS PubMed.
- (a) D. Intrieri, D. M. Carminati and E. Gallo, Dalton Trans., 2016, 45, 15746–15761 RSC; (b) G. Bartoli, G. Bencivenni and R. Dalpozzo, Synthesis, 2014, 46, 979–1029 CrossRef; (c) S. Zhu, X. Cui and X. P. Zhang, Eur. J. Inorg. Chem., 2012, 2012, 430–434 CrossRef CAS; (d) H. M. L. Davies and E. G. Antoulinakis, in Organic Reactions, John Wiley & Sons, Inc., 2001, vol. 57, p. 1 Search PubMed; (e) H. Lebel, J.-F. Marcoux, C. Molinaro and A. B. Charette, Chem. Rev., 2003, 103, 977–1050 CrossRef CAS PubMed; (f) M. P. Doyle and D. C. Forbes, Chem. Rev., 1998, 98, 911–935 CrossRef CAS PubMed.
- E. Wenkert, T. P. Ananthanarayan, V. F. Ferreira, M. G. Hoffmann and H. Kim, J. Org. Chem., 1990, 55, 4975–4976 CrossRef CAS.
- (a) N. Grimster, L. Zhang and V. V. Fokin, J. Am. Chem. Soc., 2010, 132, 2510–2511 CrossRef CAS PubMed; (b) S. Chuprakov, S. W. Kwok, L. Zhang, L. Lercher and V. V. Fokin, J. Am. Chem. Soc., 2009, 131, 18034–18035 CrossRef CAS PubMed.
- J. V. Ruppel, J. E. Jones, C. A. Huff, R. M. Kamble, Y. Chen and X. P. Zhang, Org. Lett., 2008, 10, 1995–1998 CrossRef CAS PubMed.
- Y. Chen, K. B. Fields and X. P. Zhang, J. Am. Chem. Soc., 2004, 126, 14718–14719 CrossRef CAS PubMed.
- (a) R. D. Connell, M. Tebbe, A. R. Gangloff, P. Helquist and B. Akermark, Tetrahedron, 1993, 49, 5445–5459 CrossRef CAS; (b) R. D. Connell, M. Tebbe, P. Helquist and B. Akermark, Tetrahedron Lett., 1991, 32, 17–20 CrossRef CAS; (c) J. Linder, T. P. Garner, H. E. L. Williams, M. S. Searle and C. J. Moody, J. Am. Chem. Soc., 2011, 133, 1044–1051 CrossRef CAS PubMed.
- (a) F. Gnad and O. Reiser, Chem. Rev., 2003, 103, 1603–1623 CrossRef CAS PubMed; (b) C. Cativiela and M. D. Diaz-de-Villegas, Tetrahedron: Asymmetry, 2000, 11, 645–732 CrossRef CAS; (c) S. Danishefsky, Acc. Chem. Res., 1979, 12, 66–72 CrossRef CAS; (d) H. N. C. Wong, M. Y. Hon, C. W. Tse, Y. C. Yip, J. Tanko and T. Hudlicky, Chem. Rev., 1989, 89, 165–198 CrossRef CAS.
- H. M. L. Davies and T. A. Boebel, Tetrahedron Lett., 2000, 41, 8189–8192 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available. CCDC 1532256. For ESI and crystallographic data in CIF or other electronic format see DOI: 10.1039/c7sc00658f |
| This journal is © The Royal Society of Chemistry 2017 |