Synergistic inhibitory effects of naproxen in combination with magnolol on TPA-induced skin inflammation in mice
Abstract
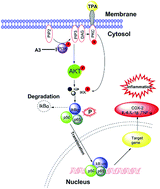
The combination of naproxen with herbs having a range of pharmacological effects is a potential alternative to improve their bioactivity and limit their adverse effects. Inspired by magnolol, an active constituent found in the bark of Magnolia officinalis, we therefore evaluated the efficacy of naproxen (N) plus magnolol (M) in 12-O-tetradecanoylphorbol-13-acetate (TPA)-induced skin inflammation. During application, treatment with N and M significantly reduced skin inflammation, and the combination of N and M resulted in a synergistic effect. Further mechanistic investigations clearly demonstrated that A3 (N : M = 1 : 3, mol mol−1) markedly suppressed TPA-induced pro-inflammatory cytokines and COX-2 expressions, inhibited NF-κB activity, downregulated IκBα and IκB kinase (IKK) activities, and inhibited PI3K/Akt and PI3K/PKC signaling pathways. These results indicated that a combination of N and M effectively inhibited TPA-induced skin inflammation via blocking PI3K/Akt/IKK and PI3K/PKC/IKK signaling pathways.

 Please wait while we load your content...
Please wait while we load your content...