One-pot approach to synthesize hyperbranched poly(thiol–ether amine) (hPtEA) through sequential “thiol–ene” and “epoxy–amine” click reactions†
Abstract
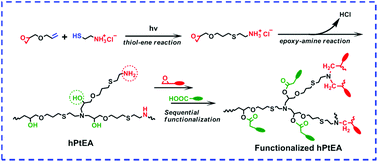
We here demonstrated a novel strategy of a one-pot approach to synthesize hyperbranched poly(thiol–ether amine) (hPtEA) through sequential “thiol–ene” and “epoxy–amine” click reactions, both of which were well traced using in situ1H-NMR spectra. The obtained hPtEA, with a high branching degree (DB) of 0.83 and a molecular weight (Mn) of 8.6 × 103, was composed of a large amount of hydroxyl groups in the backbone and amino groups in the periphery. Moreover, hPtEA could be further functionalized orthogonally due to the difference in the chemistry between the hydroxyl and amino groups. So fluorescent anthracene (AN) and carboxyl (SA) groups were introduced to the periphery and backbone of hPtEA, respectively, and the amphiphilic and zwitterionic hPtEA (SA-hPtEA-AN) was obtained. The resulting SA-hPtEA-AN could self-assemble into microspheres in aqueous solution with uniform sizes of 750 nm in diameter, and was further cross-linked through photo-dimerization of the AN groups. The microgel of SA-hPtEA-AN is fluorescent and responsive to environmental stimuli such as temperature and pH, and can be used in the encapsulation and controlled release of guest molecules. In addition, the controlled release of guest molecules from the microgel of SA-hPtEA-AN can be monitored by its fluorescence change.

 Please wait while we load your content...
Please wait while we load your content...