Synthetic studies on the bioactive tetramic acid JBIR-22 using a late stage Diels–Alder reaction†
Abstract
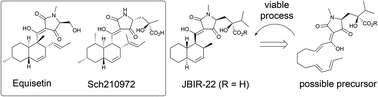
A late stage Diels–Alder reaction is used to prepare a mixture of JBIR-22, a natural product from the Equisetin family of tetramic acids, and one of its diastereomers. This is achieved in just 8 steps from pyruvate. The success of the late stage DA approach is discussed in the context of the biosynthesis of JBIR-22 (and perhaps related natural products).

 Please wait while we load your content...
Please wait while we load your content...