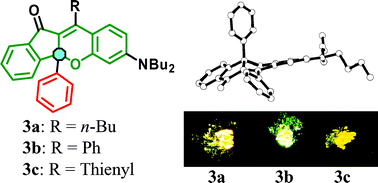
Novel solid-emissive indeno[1,2-b]benzo[4,5-e]pyran-11-one-type fluorophores 3a–3c having non-planar structures with sterically hindered substituents (R = butyl, phenyl, and thienyl) have been designed and conveniently synthesized. The fluorescence quantum yields of 3a–3c in 1,4-dioxane were 3a (Φ = 0.053) ≫
3b (Φ = 0.013) > 3c (Φ = 0.003). On the other hand, the solid-state fluorescence quantum yields of the fluorophores were 3a (Φ = 0.39) > 3b (Φ = 0.15) > 3c (Φ = 0.06). To elucidate the large differences in the quantum yields in solution and in the solid state and among the fluorophores 3a–3c, we performed time-resolved fluorescence spectroscopic measurements, semi-empirical molecular orbital calculations (AM1 and INDO/S), and X-ray crystallographic analyses of 3a–3c. The comparison of the values of the radiative and non-radiative rate constants determined by the time-resolved spectroscopic measurements in solution and in the crystalline state supported that non-radiative decay is reduced by restriction of the rotation of the phenyl and thienyl rings in the solid state. In addition, the X-ray crystal structures demonstrated that, in 3a and 3b, the non-planar structure with sterically hindered substituents prevents the fluorophores from forming short π–π contacts and produces strong solid-state fluorescence. On the other hand, in the crystal of 3c, the formation of continuous intermolecular CH⋯S bonding between neighboring fluorophores was found to increase short π–π contacts and reduce the fluorescence intensity.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...