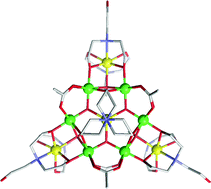
Two new potential single molecule magnets [Mn10O3(O2CCH3)6(tea)(teaH)3(teaH2)3][NO3]2·3H2O (1) and [Mn18(N3)12O12(tea)2(teaH)4(OMe)(MeOH)]·2MeOH·H2O·Et2O (2) have been synthesised using manganese(II) nitrate, the tripodal ligand triethanolamine (teaH3) and co-ligands such as acetate for 1 and azide for 2. Both compounds are mixed-valent with 1 consisting of 4 Mn(II) and 6 Mn(III) ions and 2 consisting of 3 Mn(II) and 15 Mn(III) ions. Complex 1 has a near planar Mn7(II/III) disc-like core with 3 Mn(II) ions positioned on the periphery of the disc, forming a star-like geometry. Complex 2 contains a hexa-capped truncated tetrahedral core. Variable temperature DC magnetic susceptibility data collected in 2, 1, 0.1 and 0.01 T fields, and in the 2–300 K temperature range, for 1 and 2, indicate a large ground spin-state for both compounds. Using a new approach to calculating the DC Mvs. H isotherms (or isofield Mvs. H/T curves) by means of the thermal population of Zeeman levels generated from a group of closely spaced S states (within 9 cm−1 of ground), the ground S of 1 is 10. The ZFS term, D, is close to zero. For 2, the magnetisation isotherms, and low temperature susceptibilities, point to a ground state spin of 21/2 with many closely spaced spin states (probably from 17/2 to 35/2) close in energy, the lowest Zeeman level energies of which change as the field increases. AC magnetic susceptibility data show frequency-dependent out-of-phase signals indicating both clusters are possibly single molecule magnets. Extrapolation of the in-phase component, χ'MT, to 0 K suggests spin ground state of S = 10 for 1 and 21/2 for 2, in agreement with the DC data.

 Please wait while we load your content...
Please wait while we load your content...