Theoretical study about Mo2C(101)-catalyzed hydrodeoxygenation of butyric acid to butane for biomass conversion†
Abstract
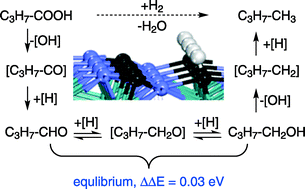
In order to understand the conversion mechanism of fatty acids to long-chain alkanes using molybdenum carbide as a catalyst, the full potential energy surface of the hydrogenation of butyric acid to butane on the H-pre-covered hexagonal Mo2C(101) surface has been systematically computed on the basis of density functional theory including dispersion (PBE-D3) and zero-point energy corrections. The first step of the reaction is butyric acid dissociation into surface OH and acyl [R-COOH + H = R-CO + OH + H], followed by the formation of butanal from acyl hydrogenation [R-CO + OH + H = R-CHO + OH]. The second step of the reaction is butanal hydrogenation into butanol via the surface butoxyl intermediate [R-CHO + H + OH = R-CH2O + OH; R-CH2O + H + OH = R-CH2OH + OH]. The third step is butanol dissociation into surface OH and butyl [R-CH2OH + H = R-CH2 + OH + H], which is further hydrogenated into butane [R-CH2 + H + OH = R-CH3 + OH]. The formation of butanal and butanol as intermediates in equilibrium as well as butane as the final product is in full agreement with the experiment of stearic acid hydrodeoxygenation into octadecane. For the conversion, butanol dissociation [R-CH2OH + H = R-CH2 + OH + H] has the highest barrier and represents the rate-determining step. It is noted that the metallic Mo2C(001) surface can bind surface H2O, OH and O much more strongly than the Mo2C(101) surface with exposed Mo and C atoms (C/Mo = 1). Since our results are obtained only from the most stable Mo2C(101) surface, they cannot correlate with the whole experimentally observed reactivity and selectivity of different Mo2C phases with different surface terminations. Nevertheless, our results provide the basis for exploring the intrinsic nature of Mo2C catalysts in deoxygenation of oxygenates involved in the refining of biomass-derived oils.

 Please wait while we load your content...
Please wait while we load your content...