Illuminating CO2 reduction on frustrated Lewis pair surfaces: investigating the role of surface hydroxides and oxygen vacancies on nanocrystalline In2O3−x(OH)y†
Abstract
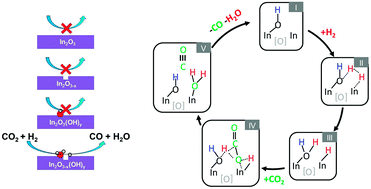
Designing catalytic nanostructures that can thermochemically or photochemically convert gaseous carbon dioxide into carbon based fuels is a significant challenge which requires a keen understanding of the chemistry of reactants, intermediates and products on surfaces. In this context, it has recently been reported that the reverse water gas shift reaction (RWGS), whereby carbon dioxide is reduced to carbon monoxide and water, CO2 + H2 → CO + H2O, can be catalysed by hydroxylated indium oxide nanocrystals, denoted In2O3−x(OH)y, more readily in the light than in the dark. The surface hydroxide groups and oxygen vacancies on In2O3−x(OH)y were both shown to assist this reaction. While this advance provides a first step toward the rational design and optimization of a single-component gas-phase CO2 reduction catalyst for solar fuels generation, the precise role of the hydroxide groups and oxygen vacancies in facilitating the reaction on In2O3−x(OH)y nanocrystals has not been resolved. In the work reported herein, for the first time we present in situ spectroscopic and kinetic observations, complemented by density functional theory analysis, that together provide mechanistic information into the surface reaction chemistry responsible for the thermochemical and photochemical RWGS reaction. Specifically, we demonstrate photochemical CO2 reduction at a rate of 150 μmol gcat−1 hour−1, which is four times better than the reduction rate in the dark, and propose a reaction mechanism whereby a surface active site of In2O3−x(OH)y, composed of a Lewis base hydroxide adjacent to a Lewis acid indium, together with an oxygen vacancy, assists the adsorption and heterolytic dissociation of H2 that enables the adsorption and reaction of CO2 to form CO and H2O as products. This mechanism, which has its analogue in molecular frustrated Lewis pair (FLP) chemistry and catalysis of CO2 and H2, is supported by preliminary kinetic investigations. The results of this study emphasize the importance of engineering the surfaces of nanostructures to facilitate gas-phase thermochemical and photochemical carbon dioxide reduction reactions to energy rich fuels at technologically significant rates.

 Please wait while we load your content...
Please wait while we load your content...