An efficient synthetic method for organometallic radicals: structures and properties of gold(i)-(nitronyl nitroxide)-2-ide complexes†
Abstract
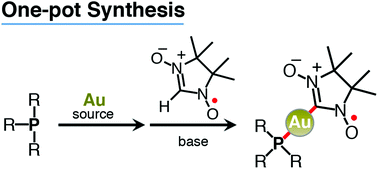
One-pot synthesis of (nitronyl nitroxide)-gold(I)-phosphine (NN-Au-P) complexes has been developed using chloro(tetrahydrothiophene)gold(I), phosphine ligands, nitronyl nitroxide radicals, and sodium hydroxide. The NN-Au-P complexes can be easily handled because they were quite stable under aerated conditions in both solution and crystalline states. They showed weak absorption bands with vibrational structures in the 450–650 nm region. The oxidation potentials assigned to the NN moieties of NN-Au-P complexes with aromatic phosphines were observed around −0.1 V vs. Fc/Fc+ (−0.11 V for NN-Au-1, −0.08 V for NN-Au-2, −0.13 V for NN-Au-5, and −0.07 V for NN-Au-6), somewhat lower than that of NN-Au-P complexes with aliphatic phosphines (−0.25 V for NN-Au-3 and −0.17 V for NN-Au-4).


 Please wait while we load your content...
Please wait while we load your content...