Isolation, structure and reactivity of a scandium boryl oxycarbene complex†
Abstract
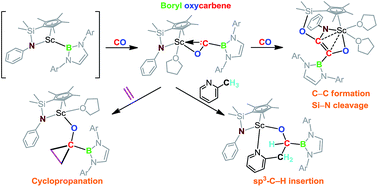
The reaction of a half-sandwich scandium boryl complex 1 with CO (1 atm) afforded a novel boryl oxycarbene complex 2. The structure of 2 was characterized by 1H, 13C and 11B NMR, X-ray diffraction, and DFT analysis. Further reaction of 2 with CO (1 atm) yielded a phenylamido- and boryl-substituted enediolate complex 3 through C–C bond formation between CO and the carbene unit in 2 and cleavage and rearrangement of the Si–N bond in the silylene-linked Cp–amido ligand. Upon heating, insertion of the carbene atom into a methine C–H bond in the boryl ligand of 2 took place to give an alkoxide complex 4. The reactions of 2 with pyridine and 2-methylpyridine led to insertion of the carbene atom into an ortho-C–H bond of pyridine and into a methyl C–H bond of 2-methylpyridine, respectively. The reaction of 2 with ethylene yielded a borylcyclopropyloxy complex 7 through cycloaddition of the carbene atom to ethylene.

 Please wait while we load your content...
Please wait while we load your content...