Novel metallo-therapeutics of the NSAID naproxen. Interaction with intracellular components that leads the cells to apoptosis†
Abstract
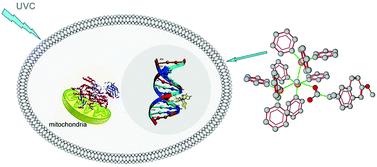
Two new mixed ligand–silver(I) complexes of the anti-inflammatory drug naproxen (naprH) and triphenylphosphine (tpp) or tri(p-tolyl)phosphine (tptp) of formulae {[Ag(tpp)3(napr)](H2O)} (1) and [Ag(tptp)2(napr)] (2) have been synthesized and characterized by m.p., vibrational spectroscopy (mid-FT-IR), Raman, 1H-NMR, UV-Vis, ESI-MS spectroscopic techniques and X-ray crystallography. The complexes show high photo-sensitivity to UVC light. Photolysis of 1–2 was studied and the results showed monotonic degradation of the complexes with simultaneous triarylphosphine oxide formation. The complexes 1–2 were tested for their antiproliferative activity against human breast adenocarcinoma (MCF-7) cells. Complexes 1–2 were more active than cisplatin against cells. UVC light increases the effectiveness of complexes 1–2 on MCF-7 cells by 13% and 38% respectively. Due to the morphology of the MCF-7 cells, which were incubated with the complexes 1–2, the cell death was ascribed to apoptosis. Electrophoresis to genomic DNA of MCF-7 cells confirmed the apoptosis through DNA fragmentation. The binding affinity of 1–2 towards the intracellular molecules CT-DNA and lipoxygenase (LOX) was studied for the evaluation of the mechanism of cell death. Thus, the binding constants (Kb) of 1–2 towards CT-DNA calculated by UV-Vis spectra are 32.8 ± 8.5 × 104 (1) and 4.7 ± 1.8 × 104 (2) M−1, respectively. Changes in fluorescent emission light of ethidium bromide (EB) in the presence of DNA suggest intercalation or electrostatic interactions into DNA of both complexes 1–2 in the minor groove. The corresponding apparent binding constants (Kapp) of 1–2 towards CT-DNA calculated through fluorescence spectra are 2.9 ± 0.3 × 104 (1) and 1.6 ± 0.4 × 104 (2) M−1 respectively. Docking studies on DNA-complexes interactions show the binding of 1 in the major groove and the corresponding one of 2 in the minor one. Moreover, the influence of complexes 1–2 on the catalytic peroxidation of linoleic acid to hydroperoxylinoleic acid by the enzyme lipoxygenase (LOX) was kinetically and theoretically studied. Only 1 inhibits lipoxygenase activity (IC50 = 5.1 (1), >30 (2) μM).

 Please wait while we load your content...
Please wait while we load your content...