A silicone-based stretchable micropost array membrane for monitoring live-cell subcellular cytoskeletal response†
Abstract
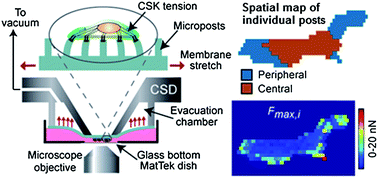
External forces are increasingly recognized as major regulators of cellular structure and function, yet the underlying mechanism by which cells sense forces and transduce them into intracellular biochemical signals and behavioral responses (‘mechanotransduction’) is largely undetermined. To aid in the mechanistic study of mechanotransduction, herein we devised a cell stretching device that allowed for quantitative control and real-time measurement of mechanical stimuli and cellular biomechanical responses. Our strategy involved a microfabricated array of

 Please wait while we load your content...
Please wait while we load your content...