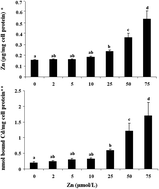
Caco-2 cell metallothionein (MT) formation was studied to determine if MT could be used as a proxy for zinc (Zn) absorption in a cell culture model. The MT intracellular concentration was determined using a cadmium/hemoglobin affinity assay. The cellular Zn uptake was determined by acid digests (5% HNO3) using inductively-coupled argon-plasma emission spectroscopy. The effect of phytic acid (PA) on cellular Zn and MT concentrations was also studied. Cells were treated with a media containing 0, 2, 5, 10, 25, 50, 75 μmol L−1 Zn (ZnCl2). The effect of varying the Zn : PA molar ratios (1 : 0, 1 : 1, 1 : 5, 1 : 10, 1 : 20) on the Zn uptake and MT formation was determined. The results showed a positive linear correlation between Zn-media concentrations and cellular Zn uptake, and MT formation was observed. Zn and MT concentrations in the cells treated with increasing levels of Zn (>25 μmol L−1 Zn) were elevated. The Zn and MT concentrations in the cells incubated with Zn (when <10 μmol L−1) were similar to the untreated cells. PA significantly lowered the cellular Zn and MT concentrations. When the Zn : PA molar ratios were >1 : 5, cellular MT concentrations were no different to untreated cells. When a combined in vitro digestion/cell model was used, the cellular MT concentrations in white or red beans and fish samples were no different to the cell baseline. This study suggests that measurements of cellular Zn and MT concentrations have some limitations (<10 μmol L−1 Zn). PA was observed to be a potent inhibitor of Zn uptake. Under the conditions of this in vitro model, Caco-2 cell monolayers are not useful for evaluating the Zn availability from foods.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...