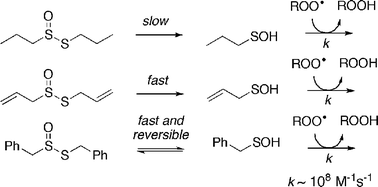
It has long been recognized that garlic and petiveria, two plants of the Allium genus—which also includes onions, leeks and shallots—possess great medicinal value. In recent times, the biological activities of extracts of these plants have been ascribed to the antioxidant properties of the thiosulfinate secondary metabolites allicin and S-benzyl phenylmethanethiosulfinate (BPT), respectively. Herein we describe our efforts to probe the mechanism of the radical-trapping antioxidant activity of these compounds, as well as S-propyl propanethiosulfinate (PPT), a saturated analog representative of the thiosulfinates that predominate in non-medicinal alliums. Our experimental results, which include thiosulfinate-inhibited autoxidations of the polyunsaturated fatty acid (ester) methyl linoleate, investigations of their decomposition kinetics, and radical clock experiments aimed at obtaining some quantitative insights into their reactions with peroxyl radicals, indicate that the radical-trapping activity of thiosulfinates is paralleled by their propensity to undergo Cope elimination to yield a sulfenic acid. Since sulfenic acids are transient species, we complement our experimental studies with the results of theoretical calculations aimed at understanding the radical-trapping behaviour of the sulfenic acids derived from allicin, BPT and PPT, and contrasting the predicted thermodynamics and kinetics of their reactions with those of the parent thiosulfinates. The calculations reveal that sulfenic acids have among the weakest O–H bonds known (ca. 70 kcal mol−1), and that their reactions with peroxyl radicals take place by a near diffusion-controlled proton-coupled electron transfer mechanism. As such, it is proposed that the abundance of a thiosulfinate in a given plant species, and the ease with which it undergoes Cope elimination to form a sulfenic acid, accounts for the differences in antioxidant activity, and perhaps medicinal value, of extracts of these plants. Interestingly, while the Cope elimination of 2-propenesulfenic acid from allicin is essentially irreversible, the analogous reaction of BPT is readily reversible. Thus, in the absence of chain-carrying peroxyl radicals (or other appropriately reactive trapping agent), BPT is reformed.

 Please wait while we load your content...
Please wait while we load your content...