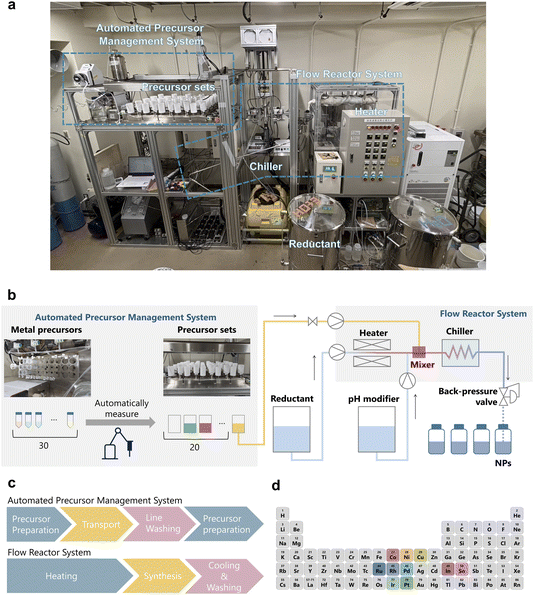
High-throughput synthesis of multi-element alloy nanoparticles using solvothermal continuous-flow reactor
Megumi
Mukoyoshi
 *a,
Kohei
Kusada†
*a,
Kohei
Kusada†
 *ab,
Xin
Zhou
a,
Takaaki
Toriyama
c,
Tomokazu
Yamamoto
c,
Yasukazu
Murakami
cd and
Hiroshi
Kitagawa
*ab,
Xin
Zhou
a,
Takaaki
Toriyama
c,
Tomokazu
Yamamoto
c,
Yasukazu
Murakami
cd and
Hiroshi
Kitagawa
 *a
*a
aDivision of Chemistry, Graduate School of Science, Kyoto University, Kitashirakawa-Oiwakecho, Sakyo-ku, Kyoto 606-8502, Japan. E-mail: mukoyoshi@ssc.kuchem.kyoto-u.ac.jp; kitagawa@kuchem.kyoto-u.ac.jp
bThe HAKUBI Center for Advanced Research, Kyoto University, Kitashirakawa-Oiwakecho, Sakyo-ku, Kyoto 606-8502, Japan
cThe Ultramicroscopy Research Center, Kyushu University, 744 Motooka, Nishi-ku, Fukuoka 819-0395, Japan
dDepartment of Applied Quantum Physics and Nuclear Engineering, Graduate School of Engineering, Kyushu University, 744 Motooka, Nishi-ku, Fukuoka 819-0395, Japan
First published on 5th August 2025
Abstract
High-throughput synthesis of multi-element alloy nanoparticles (MEA NPs) is essential for accelerating the discovery of advanced materials with complex compositions. Herein, we developed an automated continuous-flow reactor system capable of synthesising a wide variety of MEA NPs under controlled solvothermal conditions (up to 400 °C and 35 MPa). The system demonstrates a high screening throughput, capable of preparing up to 20 distinct samples in a single, automated run, with each synthesis requiring only 30 minutes. A key throughput optimising feature is the parallel process execution, whereby precursor preparation and system cleaning are performed concurrently via the reactor heating, synthesis, and cooling cycles. All washing procedures, for both the precursor preparation module and reactor unit, are fully automated, further minimising downtime. We demonstrated its versatility by successfully synthesising a wide range of MEA NPs, including high-entropy alloys, composed of various combinations of d- and p-block metals. The synthesized materials, ranging from bimetallic RuPd to ten-element CoNiCuRuRhPdInSnIrPt alloys, were all crystalline, single-phase face-centred cubic solid solutions. Furthermore, the platform enables the direct one-step synthesis of supported MEA catalysts, such as RuRhPdIrPt/CeO2. For this supported catalyst, we achieved a practical mass throughput with a theoretical production rate of 0.5 g h−1 for the MEA NPs (corresponding to 27 g h−1 for the total catalyst including the support). The final product yield was approximately 56% under the current protocol, which is designed to prevent cross-contamination by automatically discarding the initial and final portions of the product slurry. We anticipate this yield can be readily improved in a system configuration optimized for mass throughput rather than for high-throughput screening. This study presents a scalable and versatile system for high-throughput MEA NPs synthesis and offers a practical solution for bridging the gap between computational predictions and experimental materials development.
1 Introduction
Since the concept of high-entropy alloys (HEAs) was first introduced,1,2 the scope of alloy materials research has greatly expanded. In particular, multi-element alloy nanoparticles (MEA NPs) containing five or more elements have been increasingly studied in recent years because they exhibit superior properties compared with conventional NPs in areas such as catalysis.3,4 High-throughput technologies are indispensable for comprehensive exploration of such complex systems. While the advancement of computational methods, including machine learning,5 has enabled in silico screening for optimal catalytic performance, experimental realisation—especially through high-throughput synthesis—remains a major bottleneck. Bridging this gap is essential, as experimental synthesis is the foundation upon which practical discovery and validation ultimately depend.Although still in their infancy, several high-throughput strategies for MEA NP synthesis have been developed. When evaluating these “high-throughput” methods, it is helpful to consider two main aspects: (i) mass throughput, which refers to the quantity of material produced per unit of time (e.g., g h−1) and is crucial for applications requiring larger sample amounts like thermal catalysis testing; and (ii) screening throughput, which measures the number of different material compositions that can be synthesised in a given period and is essential for rapid materials discovery. Several methods have been developed, each with distinct advantages across these aspects. For example, Yao et al. developed high-throughput synthesis method based on thermal-shock, which involves precursor solution impregnation followed by rapid thermal treatment.6 This approach successfully yields carbon-supported MEA NPs with narrow size distribution and uniform distribution, making it suitable for electrocatalysis research. The simplicity of the process, involving only printing precursor salts and rapid thermal shock, suggests excellent potential for future scale-up and thus a high mass throughput. Furthermore, its ability to synthesise up to 20 samples at once demonstrates a high screening throughput.7 However, the screening versatility of this method is constrained by the limited range of support materials compatible with the thermal-shock process. For instance, gas-phase catalysis typically requires metal oxide supports, which are incompatible with this method. Löffler et al. employed laser ablation synthesis to prepare CrMnFeCoNi alloy NPs for the oxygen reduction reaction and varied the Mn content to optimise the metal composition.8 This technique is excellent at obtaining samples with varying composition ratios for a specific set of metals and offers the potential for scalable mass throughput.9,10 While these approaches are powerful for exploring specific compositional landscapes, they are often constrained by factors like the flexibility to explore vastly different elemental combinations. Therefore, a method with superior screening throughput—one that can rapidly synthesise a diverse range of elemental combinations—is highly desirable to accelerate materials discovery.
For the synthesis of solid–solution alloy NPs, it is critical to supply all constituent atoms simultaneously during both the nucleation and growth stages.11,12 Advancing this method employed in batch synthesis, we previously developed a flow reactor that successfully synthesized a variety of multi-element alloy NPs, including HEA NPs.13–15 Building on this, herein we developed an automated continuous-flow reactor system specifically designed to excel in screening throughput of MEA NPs. The system demonstrates superior screening capabilities due to its ability to utilise a wide variety of metal salts and to synthesise nanoparticles both with and without supports. Moreover, the entire synthesis process is fully automated. As a demonstration, we synthesised a series of MEA NPs containing a wide range of elements, including platinum-group metals (PGMs), iron-group metals, and p-block metals (pMs). Furthermore, we achieved a one-step synthesis of HEA NPs supported on materials such as CeO2, a widely used and catalytically active support in industrial applications, to demonstrate the system’s practical utility. We expect that this automated continuous-flow reactor system will play a pivotal role in accelerating the discovery and optimisation of MEA NPs for various applications.
2 Methods
2.1 Materials
RhCl3·3H2O and H2IrCl6·nH2O were purchased from Tanaka Precious Metal Technologies Co., Ltd. CeO2 (HS) was purchased from Daiichi Kigenso Kagaku Kogyo Co., Ltd. All other chemicals were purchased from FUJIFILM Wako Pure Chemical Corporation. The chemicals were used as received.2.2 Automated continuous-flow reactor system
A schematic diagram and image of the automated continuous-flow reactor system is shown in Fig. 1a and b. The reactor is equipped with three separate pumps supplying the reductant, metal precursors, and pH modifier, and these solutions react in the mixer. Before reaching the mixer, the reductant is heated to the target temperature (up to 400 °C), where the constituent ions are concurrently rapidly reduced. After the reaction, the solution is rapidly cooled down to room temperature to suppress further particle growth. Additionally, a back-pressure valve enables the use of lower-boiling-point solvents, such as ethanol, beyond their normal boiling points (up to 35 MPa). This high-pressure capability allows for the utilisation of supercritical fluids such as supercritical water and ethanol, thereby enabling the synthesis of not only alloy NPs but also complex oxide NPs.16 The system automatically measures out metal precursors and has the capacity to prepare up to 20 distinct precursor sets, with each synthesis procedure requiring only 30 min. The metal salt preparation, cooling, washing, and heating processes of the reactor are performed in parallel. This parallel execution allows for streamlined synthesis, thereby significantly enhancing efficiency.The system significantly enhances throughput, featuring an automated precursor dispensing module that can prepare and process up to 20 distinct precursor sets in a single experimental run from a library of 30 different metal sources, thereby effectively eliminating manual preparation steps. Combined with the short synthesis procedure of 30 min, this enables highly efficient throughput and rapid screening of vast compositional spaces. A crucial innovation supporting this high efficiency is the parallel execution of processes within the system, as depicted in Fig. 1c. While the Flow Reactor System performs the heating, synthesis, and cooling of the product, the Automated Precursor Management System simultaneously handles precursor dispensing and automated system cleaning for subsequent reactions. All washing procedures, for both the precursor preparation module and the reactor unit, are fully automated, further minimising downtime and labour.
2.3 Synthesis of MEA NPs
In this study, we successfully synthesised RuPd, RuPdIr, RhPdPt, RuRhPdIrPt, NiCuRhPdPt, RuRhPdInSnIrPt, and CoNiCuRuRhPdInSnIrPt MEA NPs using our home-designed automated continuous-flow reactor. The metal precursors used for all syntheses were CoCl2, NiCl2, CuCl2·H2O, RuCl3·nH2O, RhCl3·3H2O, K2PdCl4, InCl3·4H2O, SnCl2, H2IrCl6·nH2O, and K2PtCl4 (Fig. 1d). As a representative example, RuRhPdIrPt NPs were synthesised by co-injecting the metal precursor in HCl aqueous solution (0.1 mol L−1), heated reductant (25 vol% aqueous ethanol solution), and 0.5 M aqueous NaOH solution as a pH modifier. The precursor solution was prepared by dissolving RuCl3·nH2O, RhCl3·3H2O, K2PdCl4, H2IrCl6·nH2O, and K2PtCl4 in deionised water to obtain a total metal concentration of 4.5 mmol L−1. Poly(N-vinyl-2-pyrrolidone) (PVP) was also added to the precursor solution at a concentration of 0.1 mol L−1 as a protective agent. The precursor, reductant, and NaOH solutions were fed at flow rates of 15.0, 80.0, and 1.0 mL min−1, respectively, under a back-pressure of 25 MPa. The reaction was performed at a heater temperature of 350 °C. The resulting black solution was collected, and the NPs were separated by centrifugation and washed with a mixture of ether and ethanol. The detailed synthetic conditions for the remaining samples are summarised in Table 1.| Sample | Metal precursor solution | Reductant | NaOH solution | Pressure [MPa] | Temp [°C] | ||||
|---|---|---|---|---|---|---|---|---|---|
| Metal [mmol L−1] | PVP [mol L−1] | Flow rate [ml min−1] | Ethanol [vol%] | Flow rate [ml min−1] | NaOH [mol L−1] | Flow rate [ml min−1] | |||
| RuPd | 12.0 | 0.1 | 15.0 | 25 | 80.0 | 0.25 | 2.0 | 25 | 350 |
| RuPdIr | 9.4 | 0.1 | 15.0 | 25 | 80.0 | 0.25 | 2.0 | 25 | 350 |
| RhPdPt | 4.6 | 0.1 | 15.0 | 25 | 80.0 | 0.25 | 2.0 | 25 | 350 |
| RuRhPdIrPt | 4.5 | 0.1 | 15.0 | 25 | 80.0 | 0.50 | 1.0 | 25 | 350 |
| NiCuRhPdPt | 9.5 | 0.1 | 15.0 | 25 | 80.0 | 0.50 | 2.0 | 25 | 380 |
| RuRhPdInSnIrPt | 4.7 | 0.1 | 15.0 | 25 | 80.0 | 0.50 | 2.0 | 25 | 380 |
| CoNiCuRuRhPdInSnIrPt | 10.1 | 0.1 | 15.0 | 25 | 80.0 | 0.50 | 6.0 | 25 | 370 |
| RuRhPdIrPt/CeO2 | 4.0 | 0 | 15.0 | 25 | 90.0 | 0.50 | 1.0 | 25 | 350 |
2.4 Characterisation
Scanning transmission electron microscopy (STEM) and energy dispersive X-ray spectroscopy (EDX) measurements were recorded on a JEM-ARM 300F2 STEM instrument (JEOL, Japan) operated at 200 kV. Transmission electron microscopy (TEM) images were acquired using an HT7700 (Hitachi, Japan) at an acceleration voltage of 100 kV. The mean diameters of NPs were estimated by counting 200 particles. Powder X-ray diffraction (PXRD) profiles were obtained by MiniFlex600-C (Rigaku, Japan). The radiation wavelength was 1.542 Å (Cu Kα), and the scan step was 0.05°. Le Bail fitting was performed using GSAS-II software.17 X-ray photoelectron spectroscopy (XPS) analyses were conducted using an ESCA-3400 (SHIMADZU, Japan) with a Mg Kα radiation source (1253.6 eV). The measurements were performed before and after Ar-ion sputtering (20 mA, 2.0 kV, 60 s) to investigate the chemical states of the surface and core parts of the NPs. The binding energy was corrected with reference to the C (1s) peak at 284.6 eV. The peak fitting was performed using the XPST program package developed for IGOR Pro by Dr Martin Schmid, Philipps University, Marburg. The atomic ratios of the synthesised NPs were confirmed using a ZSX Primus IV X-ray fluorescence (XRF) instrument (Rigaku, Japan).3 Results and discussion
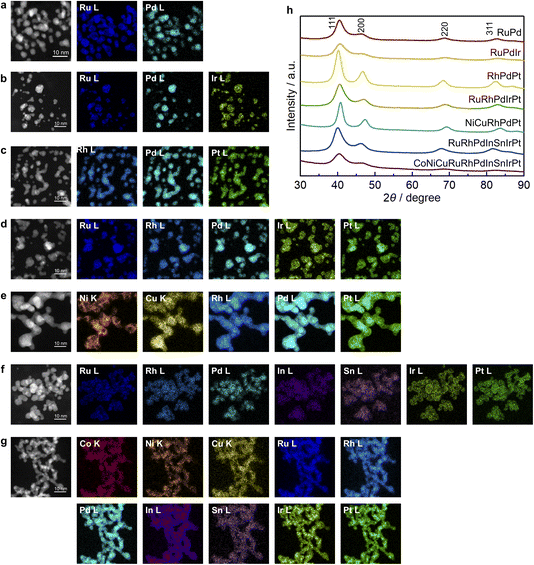
The obtained NPs were characterised using STEM-EDX and PXRD. From STEM-EDX mapping (Fig. 2a–g), we confirmed that all the constituent elements were present within each particle, confirming the successful formation of MEA NPs. The EDX maps of the samples containing pMs (RuRhPdInSnIrPt and CoNiCuRuRhPdInSnIrPt) suggested that the pM elements were partially segregated toward the particle surface, forming a core/shell-like structure. STEM-EDX line analysis of RuRhPdInSnIrPt NPs (Fig. S1) indicated that the alloy NPs were covered by In and Sn oxide species. XPS analysis was performed to determine the oxidation state of the pM species in RuRhPdInSnIrPt NPs. The In 3d XPS spectrum (Fig. S2a and Table S1) showed two peaks at binding energies (BEs) of 445.9 and 453.6 eV, which were attributed to 3d5/2 and 3d3/2 of In2O3, respectively.18 After Ar-ion sputtering, in addition to the In2O3 peak, two peaks at BEs of 443.9 and 451.7 eV were observed, attributed to 3d5/2 and 3d3/2 of metallic In (Fig. S2a and Table S1), respectively.18 Although analysis of the Sn 3d XPS spectrum was complicated due to the peak overlap with Ru 3p and Rh 3p peaks, a similar tendency was observed (Fig. S2b and Table S1). The XPS results indicated that while pMs formed oxidised clusters on the surface, metallic pM species were also present within the particle cores. Similar surface segregation behaviour of pM elements has been reported in other pM–PGM alloy systems.12,19 For CoNiCuRuRhPdInSnIrPt NPs, the XPS peaks were too weak to perform reliable fitting (Fig. S3).
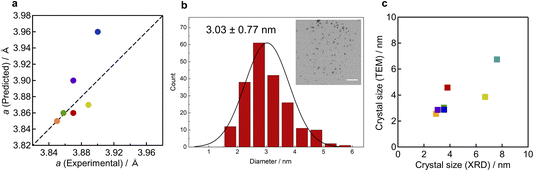
Fig. 2h shows the PXRD patterns of the synthesised materials. The patterns of all obtained NPs indicated the formation of a single face centred cubic (fcc) structure. To discuss the compositional dependence of the structures, the lattice constants estimated from the Le Bail method were compared with the composition-weighted average of elemental lattice constants, as reported previously.20 The calculation was based on the lattice constants of bulk monometals20–22 and the metal compositions confirmed by the XRF measurements (Table S2). For example, the composition-weighted average of the elemental lattice constant for RuRhPdIrPt NPs can be calculated using the following equation:
| aalloy = aRuxRu + aRhxRh + aPdxPd + aIrxIr + aPtxPt |
Furthermore, to demonstrate the applicability of our continuous-flow reactor to industrial catalyst development, we used it to synthesise HEA NPs directly supported on CeO2 (RuRhPdIrPt/CeO2) without a protecting agent. The CeO2 support was added to the metal precursor solution (RuCl3·nH2O, RhCl3·3H2O, K2PdCl4, H2IrCl6·nH2O, and K2PtCl4, each at a concentration of 0.8 mmol L−1) as 3 wt% slurry in HCl aqueous solution (0.1 mol L−1). Aqueous ethanol (25 vol%) was used as the reductant solution. Fig. 4a shows the PXRD profiles of the synthesised RuRhPdIrPt/CeO2. In addition to the diffraction peaks corresponding to CeO2,24 a broad peak assumed to represent the fcc structure of RuRhPdIrPt NPs was also observed, indicating the successful synthesis of the supported NPs. Fig. 4b and c show the HAADF-STEM image of RuRhPdIrPt/CeO2 and the corresponding STEM-EDX maps, respectively. The results suggest that the five elements exist in each particle, and the particles are supported on CeO2. The STEM-EDX line analysis of RuRhPdIrPt/CeO2 (Fig. S7) also confirmed the successful direct synthesis of HEA NPs supported on CeO2. This demonstrates the effectiveness of our developed device for synthesising HEA NPs, not only in laboratory environments but also in practical catalyst development.
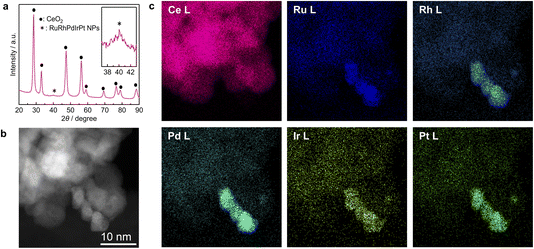 | ||
| Fig. 4 RuRhPdIrPt/CeO2 characterisation: (a) PXRD pattern, (b) HAADF-STEM image, and (c) corresponding elemental maps for (b). | ||
We then evaluated the production throughput and yield for this practical synthesis. For RuRhPdIrPt/CeO2, 100 mL of a precursor solution with a total metal concentration of 4 mmol L−1 (0.8 mmol L−1 each of Ru, Rh, Pd, Ir, and Pt; average molecular weight: 139.5 g mol−1) was used. This corresponds to a theoretical yield of 56.3 mg of the metal alloy. The actual recovered mass of the supported catalyst was 650.81 mg, and XRF analysis determined the metal content to be 2.51 wt%, which means a practical metal component yield of 56%. The main reason for the relatively low yield is attributed to the automated washing process designed to prevent cross-contamination, where the system is programmed to automatically discard the initial and final portions of the sample slurry solution. Based on the flow rate of 15 mL min−1, the theoretical production rate for this catalyst is 0.5 g h−1. We believe this can be easily scaled up for producing a large quantity of a single catalyst type, as demonstrated in our previous report.13 This successful synthesis, combined with a practical production rate, demonstrates the effectiveness of our developed device not only in laboratory environments but also in practical catalyst development.
4 Conclusions
In this study, we developed an automated continuous-flow reactor system that enables the high-throughput synthesis of MEA NPs, including HEAs. This synthetic platform allows for efficient and scalable production of compositionally diverse NPs under precisely controlled reaction conditions. As a demonstration, we successfully synthesized a series of MEA NPs composed of various d- and p-block elements. Furthermore, the reactor system is capable of producing supported HEA catalysts via a one-step process, as demonstrated using CeO2 as the support, highlighting its potential utility in practical catalytic applications. Overall, a versatile, highly efficient, and scalable synthetic platform capable of dramatically accelerating the experimental exploration of complex alloy compositions is proposed. It provides a robust solution, bridging a critical gap between computational predictions and the experimental realisation of advanced multi-element materials. Building on this foundation, collaborative research within a national project is currently ongoing.25Author contributions
M. M., K. K. and H. K. conceived the idea and designed the research. M. M., K. K. and X. Z. performed the synthesis and data analysis. T. T., T. Y. and Y. M. contributed to the STEM measurements. M. M., K. K. and H. K. discussed the results and wrote the manuscript. All the authors discussed and commented on the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information.Supplementary information is available: TEM, STEM-EDX, XPS, XRF, and PXRD analysis. See DOI: https://doi.org/10.1039/d5fd00103j.
Acknowledgements
The authors acknowledge the support of JSPS KAKENHI Grant-in-Aid for Specially Promoted Research (20H05623) and Grant-in-Aid for Early-Career Scientists (25K18094). This work was partially supported by the Demonstration Project of Innovative Catalyst Technology for Decarbonisation through Regional Resource Recycling, the Ministry of the Environment, Government of Japan. This work was also supported by FY 2022 Kusunoki 125 of Kyoto University 125th Anniversary Fund, Maekawa Houonkai Foundation Academic Research Grant, and Toyota Riken Scholar Program. STEM analyses were supported by ‘Advanced Research Infrastructure for Materials and Nanotechnology in Japan (ARIM)’ of the Ministry of Education, Culture, Sports, Science and Technology (MEXT) (Proposal Numbers JPMXP12225KU0003 and JPMXP12225KU0004). The authors thank S. Fujiwara, H. Tasaki, O. Matsutake, S. Mori, T. Urasaki, M. Shiokawa, N. Oikawa and K. Aritoku (Kyoto University) for assistance with the experimental processes.References
- M.-H. Tsai and J.-W. Yeh, Mater. Res. Lett., 2014, 2, 107–123 CrossRef.
- J.-W. Yeh, S.-K. Chen, S.-J. Lin, J.-Y. Gan, T.-S. Chin, T.-T. Shun, C.-H. Tsau and S.-Y. Chang, Adv. Eng. Mater., 2004, 6, 299–303 CrossRef CAS.
- Y. Yao, Z. Huang, P. Xie, S. D. Lacey, R. J. Jacob, H. Xie, F. Chen, A. Nie, T. Pu, M. Rehwoldt, D. Yu, M. R. Zachariah, C. Wang, R. Shahbazian-Yassar, J. Li and L. Hu, Science, 2018, 359, 1489–1494 CrossRef CAS.
- D. Wu, K. Kusada, T. Yamamoto, T. Toriyama, S. Matsumura, I. Gueye, O. Seo, J. Kim, S. Hiroi, O. Sakata, S. Kawaguchi, Y. Kubota and H. Kitagawa, Chem. Sci., 2020, 11, 12731–12736 RSC.
- Z. Rao, P.-Y. Tung, R. Xie, Y. Wei, H. Zhang, A. Ferrari, T. P. C. Klaver, F. Körmann, P. T. Sukumar, A. Kwiatkowski da Silva, Y. Chen, Z. Li, D. Ponge, J. Neugebauer, O. Gutfleisch, S. Bauer and D. Raabe, Science, 2022, 378, 78–85 CrossRef CAS.
- Y. Yao, Z. Huang, T. Li, H. Wang, Y. Liu, H. S. Stein, Y. Mao, J. Gao, M. Jiao, Q. Dong, J. Dai, P. Xie, H. Xie, S. D. Lacey, I. Takeuchi, J. M. Gregoire, R. Jiang, C. Wang, A. D. Taylor, R. Shahbazian-Yassar and L. Hu, Proc. Natl. Acad. Sci. U. S. A., 2020, 117, 6316–6322 CrossRef CAS.
- Y. Pan, X. Shan, F. Cai, H. Gao, J. Xu and M. Zhou, Angew. Chem., Int. Ed., 2024, 63, e202407116 CrossRef CAS.
- T. Löffler, F. Waag, B. Gökce, A. Ludwig, S. Barcikowski and W. Schuhmann, ACS Catal., 2021, 11, 1014–1023 CrossRef.
- F. Waag, R. Streubel, B. Gökce and S. Barcikowski, Appl. Nanosci., 2021, 11, 1303–1312 CrossRef CAS.
- F. Waag, Y. Li, A. R. Ziefuß, E. Bertin, M. Kamp, V. Duppel, G. Marzun, L. Kienle, S. Barcikowski and B. Gökce, RSC Adv., 2019, 9, 18547–18558 RSC.
- K. Kusada, M. Yamauchi, H. Kobayashi, H. Kitagawa and Y. Kubota, J. Am. Chem. Soc., 2010, 132, 15896–15898 CrossRef CAS PubMed.
- X. Zhou, M. Mukoyoshi, K. Kusada, T. Yamamoto, T. Toriyama, Y. Murakami, T. Ina, S. Kawaguchi, Y. Kubota and H. Kitagawa, ACS Mater. Lett., 2024, 6, 353–359 CrossRef CAS.
- K. Kusada, T. Yamamoto, T. Toriyama, S. Matsumura, K. Sato, K. Nagaoka, K. Terada, Y. Ikeda, Y. Hirai and H. Kitagawa, J. Phys. Chem. C, 2021, 125, 458–463 CrossRef CAS.
- H. Minamihara, K. Kusada, D. Wu, T. Yamamoto, T. Toriyama, S. Matsumura, L. S. R. Kumara, K. Ohara, O. Sakata, S. Kawaguchi, Y. Kubota and H. Kitagawa, J. Am. Chem. Soc., 2022, 144, 11525–11529 CrossRef CAS PubMed.
- H. Minamihara, K. Kusada, T. Yamamoto, T. Toriyama, Y. Murakami, S. Matsumura, L. S. R. Kumara, O. Sakata, S. Kawaguchi, Y. Kubota, O. Seo, S. Yasuno and H. Kitagawa, J. Am. Chem. Soc., 2023, 145, 17136–17142 CrossRef CAS.
- S. Hanabata, K. Kusada, T. Yamamoto, T. Toriyama, S. Matsumura, S. Kawaguchi, Y. Kubota, Y. Nishida, M. Haneda and H. Kitagawa, J. Am. Chem. Soc., 2024, 146, 181–186 CrossRef CAS.
- B. H. Toby and R. B. Von Dreele, J. Appl. Crystallogr., 2013, 46, 544–549 CrossRef CAS.
- Z. M. Detweiler, S. M. Wulfsberg, M. G. Frith, A. B. Bocarsly and S. L. Bernasek, Surf. Sci., 2016, 648, 188–195 CrossRef CAS.
- X. Zhou, M. Mukoyoshi, K. Kusada, T. Yamamoto, T. Toriyama, Y. Murakami, S. Kawaguchi, Y. Kubota, O. Seo, O. Sakata, T. Ina and H. Kitagawa, Chem. Sci., 2024, 15, 7560–7567 RSC.
- Y. Maruta, K. Kusada, D. Wu, T. Yamamoto, T. Toriyama, S. Matsumura, O. Seo, S. Yasuno, S. Kawaguchi, O. Sakata, Y. Kubota and H. Kitagawa, Chem. Commun., 2022, 58, 6421–6424 RSC.
- M. Mukoyoshi, H. Kobayashi, K. Kusada, K. Otsubo, M. Maesato, Y. Kubota, T. Yamamoto, S. Matsumura and H. Kitagawa, Chem. Commun., 2021, 57, 5897–5900 RSC.
- R. K. Linde, J. Appl. Phys., 1966, 37, 934 CrossRef CAS.
- M. Nakamura, D. Wu, M. Mukoyoshi, K. Kusada, H. Hayashi, T. Toriyama, T. Yamamoto, Y. Murakami, H. Ashitani, S. Kawaguchi, T. Ina, O. Sakata, Y. Kubota, I. Tanaka and H. Kitagawa, ACS Nanosci. Au, 2025, 5, 196–207 CrossRef CAS.
- R. Suresh, V. Ponnuswamy and R. Mariappan, Appl. Surf. Sci., 2013, 273, 457–464 CrossRef CAS.
- Technical Report on Demonstration Project of Innovative Catalyst Technology for Decarbonization through Regional Resource Recycling, FY2024, https://ndlsearch.ndl.go.jp/books/R100000002-I034118902, accessed 16 June 2025.
Footnote |
| † Current address: Institute for Advanced Study, Kyushu University, 6-1, Kasugakoen, Kasuga-shi, Fukuoka, 816-8580, Japan, kusada.kohei.236@m.kyushu-u.ac.jp |
| This journal is © The Royal Society of Chemistry 2026 |