 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Concluding (closing?) remarks
Félix M.
Goñi
ab
aInstituto Biofisika (UPV/EHU, CSIC), and Department of Biochemistry and Molecular Biology, University of the Basque Country, 48940 Leioa, Spain. E-mail: felix.goni@ehu.es
bDonostia International Physics Center (DIPC), Paseo Manuel de Lardizabal 4, 20018 Donostia-San Sebastián, Spain
First published on 26th June 2025
Abstract
This paper summarizes a series of ideas and/or concepts, most of which were the object of the Faraday Discussion on ‘Structural and functional asymmetry of plasma membranes’. A historical review is provided of the early symmetrical and asymmetrical models for membrane structure. Membrane asymmetry in the framework of evolution is suggested as a possible field of study. Functional membrane asymmetry in experimental models is briefly discussed, and the hypothesis that functional asymmetry preceded structural asymmetry in evolution is proposed. Lipid asymmetry and lipid scrambling in bilayers are presented as two complementary aspects of the same process. The use of sphingomyelinases in experimental studies of asymmetry is criticized, due to the lipid scrambling properties of the sphingomyelinase end-product ceramide. The paper ends with a note on the (apparently?) cyclical nature of scientific research.
1. Concluding/closing remarks
I would like to start these formally called “Concluding Remarks” by slightly objecting to the title, as given by the organizers/editors. According to the OED, “to conclude” means, for sure, “to bring to a close or end; to bring something to an end”. In that sense, it is certainly appropriate to place the concluding remarks at the end of this issue of Faraday Discussions. However, the OED provides another meaning of the same word, namely: “to decide or believe something as a result of what you have heard or seen” [italics mine]. The latter I find alarming in the present context. It could mean that I must make decisions on my colleagues' proposals or discussions, something for which I do not feel qualified. Even worse, would I be asked to “believe” anything? I confess I am a man of little faith, and the modicum of faith I have I consume in church, leaving none of it for the lab. How much easier, then, would it be to write a little paper with some Closing Remarks [OED, “close: to put something into a position so that it covers an opening”. That's it. Any opening is covered. The matter is closed]. Let me follow, then, once again, the facile path, and leave decisions and beliefs to a more audacious author.If I am allowed to follow in this rather personal vein, I would note that these are, if I am correct, my fourth Faraday Discussions, the earliest of which took place in 1986, with a paper entitled “A study of phospholipid phosphate groups in model membranes by Fourier transform infrared spectroscopy”, that I presented together with my sadly missed colleague and friend Professor J. L. R. Arrondo.1 As it happens, the current edition will also be the last one for me, since in three months from now I will be closing down my lab. Thus, these are my Closing Remarks indeed.
2. Historical notes
The first model for cell membrane structure that received wide, if not universal approval, was the one proposed by J. Danielli and H. Davson in 1935. In this model the phospholipids were organized in a bilayer, the proteins (in a β-conformation) disposed in contact with, and outside of, the phospholipid polar headgroups.2 This was a symmetrical model, with a symmetry plane corresponding approximately to the fatty acyl end methyl groups in the bilayer hydrophobic matrix (Fig. 1).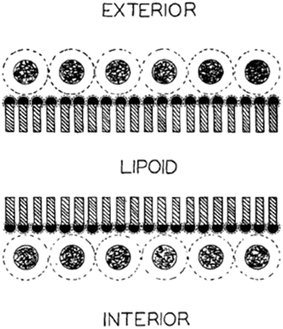 | ||
| Fig. 1 Symmetric bilayer model of Danielli and Davson2 (the original legend was “Schema of molecular conditions at the cell surface”). Reprinted from ref. 2, with permission from John Wiley and Sons. | ||
The Sixties were rather turbulent years, not only for University campuses and miniskirt manufacturing ateliers, but also in the business of membrane model development. A plethora of experimental data, from X-ray scattering, electron microscopy, and other newly available physical and biochemical techniques, gave rise to as many models, apparently in contradiction with Danielli–Davson's, and also contradicting with each other. This was the panorama against which, early in 1972, the Singer–Nicolson model was projected.3 The success of the model was phenomenal, eliciting even some jealous response from experimentalists who had all the data in mind, all except the unifying model, to which they disdainfully referred as “the cartoon”. The fact is that over half a century has elapsed, and the “fluid mosaic model”, as its official name goes, remains as the universally accepted model for cell membrane structure and dynamics (Fig. 2). Importantly for our purpose, it is an asymmetrical model, in which the bilayer-embedded intrinsic proteins emerge differently from each side of the bilayer. Moreover, the text in that paper3 states that lipid distribution is also asymmetric. The sub-title in the journal is explicit: “Cell membranes are viewed as two-dimensional solutions of oriented globular proteins and lipids” [italics mine].
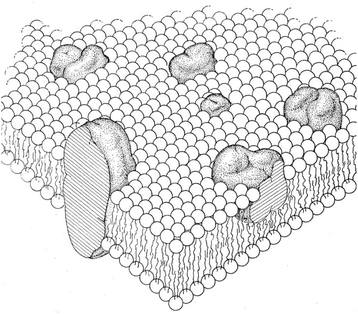 | ||
| Fig. 2 Asymmetric fluid mosaic model of S. J. Singer and G. L. Nicolson.3 Figure reproduced with permission of AAAS, Science, 1972, 175(4023) 720–731. | ||
In fact, by 1972, ideas about cell membrane asymmetry had been developing for over 30 years. To mention but a few important hallmarks in a continuous thread, A. L. Hodgkin and A. F. Huxley recorded, in 1939, action potentials from inside a nerve fibre,4 an observation that obviously implied an essential asymmetry. A decade later, the same authors explained the ionic mechanisms of action potentials.5 Five years later, J. C. Skou6 described “the influence of some cations on an adenosine triphosphate from peripheral nerves” in what appears to have been an understatement for the discovery of the Na+/K+ ATPase pump, the word “pump” deemed too novel or revolutionary at the time. Also in 1957, P. Mitchell proposed the existence of membrane-embedded enzymatic transporters.7 Thus in the late Fifties membrane asymmetry was implicitly accepted, though not until 1972 was this concept made explicit by Singer and Nicolson.
As it often happens in many branches of science, in the field of membrane structure theory helped and was helped by experimentation. The fluid mosaic model was a rationalization of seemingly incoherent experimental results from the Sixties. At the same time, it fueled powerful experimental studies in the Seventies and following decades. Four giant names should be mentioned in this context, while acknowledging an unavoidable personal bias in the selection. They appear in order of their birth dates, and their merits are exposed in a telegraphic way. They are: Vittorio Luzzati (1923–2016), who performed an unsurpassed description of the membrane lipid phases, including the non-lamellar phases, using X-ray scattering;8Dennis Chapman (1927–1999), who studied the phospholipid thermotropic transitions, and explained the role of cholesterol in bilayers;9Laurens L. van Deenen (1928–1994), founder of the Utrecht school of biomembrane studies, which in turn made fashionable this area of biology/biochemistry;10 and Don M. Small (1931–2019), the author of flawless analyses of the physical biochemistry of bile salts, lipoproteins, and atherosclerotic lesions.11
3. Membrane asymmetry and evolution
Membrane asymmetry in the broader framework of evolution has not been dealt with in a specific way during the meeting. However, I consider that it deserves at least a brief consideration. From the times of M. Schleiden12 and T. Schwann,13 cells have been considered as the building blocks of all living beings. Large efforts have been dedicated to reconstruct the long, winding way from the abiotic synthesis of biomolecules to the first cell, a process that took perhaps 300 m years.Many of these studies are based, albeit implicitly, on the work by E. Overton who, from systematic measurements of the entry of apolar compounds in the cells, concluded that the plasma membrane ought to be lipidic in nature.14 One century later, vesicles defined by symmetric bilayers, often consisting of pure oleic acid or other equally simple compositions, have been used as model systems to study the properties of membranes that could be the evolutionary precursors of more complex, stable, and impermeable phospholipid biomembranes.15 These structures would be able to encapsulate macromolecules, e.g. RNA, but they could hardly support ionic gradients, because of their instability, and/or high permeability. Increasing the complexity of bilayer composition may lead to more stable membranes. In particular 10- to 14-C-atom alcohols were shown to contribute to the stability of oleic acid vesicles under a wider range of experimental conditions.16
The current efforts at reconstructing the pre-biotic to biotic evolution are still far from reaching a minimum of detail. There is however good agreement that the last universal common ancestor (LUCA) of present-day cells likely generated ATP through chemiosmotic coupling at a semi-permeable, obviously asymmetric, membrane-like interface. Energy from hydrothermal vents at the seafloor could have played a crucial role in the appearance of LUCA. Even a proto-cell with self-replicating properties is not conceivable with a symmetric membrane. Nevertheless, we are far from understanding, to begin with, how asymmetric membranes first appeared in what would become the Biosphere.
4. Functional asymmetry in reconstituted membranes
This is an aspect of asymmetry that is often overlooked in reviews and discussions on this topic. I call functional asymmetry the situation in which a structurally symmetric membrane, containing non-oriented carriers or transporters, becomes functionally asymmetric when the transported molecule is added on one side only, typically from the outside. When the initial transport rate is measured, only the transporters having the appropriate orientation will operate, all others being blind to the assay, and the membrane will be, in practical terms, asymmetric. Non-oriented, or partially non-oriented transporters occur when a protein purified in detergent is reconstituted with phospholipids, and the detergent is removed. Perhaps the first example of this technique was the celebrated experiment by E. Racker and W. Stoeckenius,17 in which H+-ATPase from beef heart mitochondria and Halobacterium bacteriorhodopsin were reconstituted in soybean lecithin vesicles, with the outcome of asymmetric ATP synthesis. Functionally asymmetric transport in cell membranes may be achieved, in either direction, whenever right-side-out and inside-out vesicles can be derived from the membranes; red blood cell membranes are a typical example.18 The hypothesis that functional asymmetry preceded structural asymmetry in evolution might deserve some consideration.5. Lipid asymmetry and scrambling in cell bilayers
While the asymmetric topology of cell membrane proteins, as proposed by Singer and Nicolson,3 was experimentally demonstrated at a rather early stage,19–21 lipid asymmetry has been more difficult to assess. This should be partly attributed to the fact that protein asymmetry is absolute, or qualitative, i.e. each and every molecule of a given protein exhibit a common topology, while lipid asymmetry is relative, or quantitative, i.e. certain lipids predominate in one monolayer, while others do not.Lipid asymmetry should be considered together with its molecular nemesis, scrambling. Scrambling destroys asymmetry, leading to a symmetric distribution of lipids in both monolayers. Entropic considerations predict the spontaneous transbilayer (or flip-flop) motion of lipids, which is effectively prevented by the lack of affinity of the lipid polar headgroups for the hydrophobic membrane matrix. In fact, when some degree of transbilayer lipid motion is required, the cell makes use of scramblases, intrinsic membrane enzymes constituting a group of homologous bidirectional lipid translocators that are conserved in all eukaryotic organisms.22,23 Lipid scrambling may help understand lipid asymmetry in the same way as studying catabolic processes helps in understanding biosynthesis, oxidation reactions help in the study of reduction, etc. The same two-way kind of studies have facilitated the comprehension of esterification/hydrolysis, ATP hydrolysis/ADP phosphorylation, membrane fusion/fission, and many others. Note, however, that a biological process is rarely, if ever, the exact reversal of the reciprocal process. Actually, the Second Law of Thermodynamics prevents any such exact reversion. To follow the biochemical reasoning, many of the reactions in the gluconeogenesis pathway are the same as those in the anaerobic glycolysis, and yet e.g. the key glycolytic reaction of pyruvate kinase cannot be exactly reversed to produce glucose synthesis under the cellular conditions.
The interplay of bilayer lipid asymmetry and lipid scrambling should be underlined in a correct interpretation of old and recent experimental studies of the lipid composition of red blood cell (RBC) membranes. In a classical paper from van Deenen's group,24 when the RBC were treated successively with phospholipase A2 and sphingomyelinase no lysis occurred, and up to 48% of the total phospholipids were degraded. Since the enzymes were too large to diffuse across the membrane, it was concluded that the hydrolyzed phospholipid fraction (containing the majority of the choline-containing phospholipids and some phosphatidylethanolamine) formed the outer monolayer of the membrane. The two-enzyme method has survived to our days, having been applied to a very recent investigation on cholesterol asymmetry.25 However, the use of sphingomyelinase in bilayer asymmetry studies leads to almost inevitable flaws, due to the powerful scrambling activity of the sphingomyelinase reaction end-product, ceramide.
A series of studies from our laboratory23,26,27 have shown that ceramide, either produced in situ from sphingomyelin by sphingomyelinase, or added in solvent to pre-formed bilayers, destroys lipid asymmetry, facilitating the flip-flop motion of lipids, including those with a bulky polar headgroup, e.g. gangliosides, which should be particularly resistant to diffusion through the hydrophobic matrix (Fig. 3). In consequence, lipid asymmetry data obtained with sphingomyelinase are probably flawed, and should be corrected for the ceramide-induced flip-flop. In other words, real asymmetry may be larger than the apparent one.
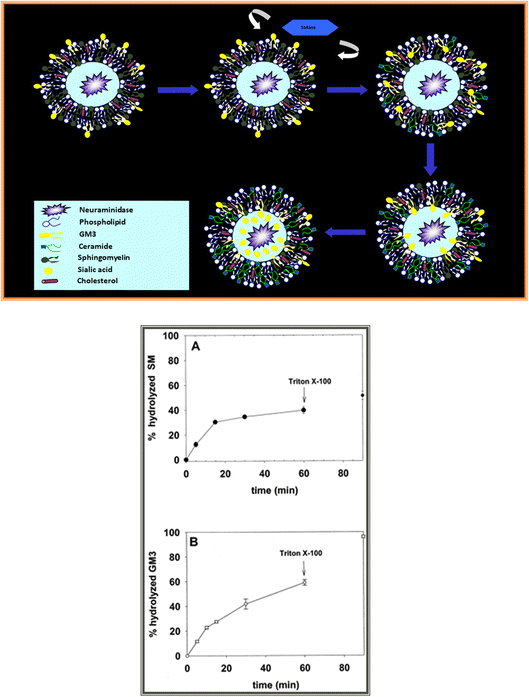 | ||
Fig. 3 Top: outline of an experiment demonstrating the flip-flop motion of gangliosides induced by sphingomyelinase-generated ceramide in large unilamellar vesicles. Bottom: flip-flop of gangliosides induced by sphingomyelinase activity in large unilamellar vesicles.26 (A) Sphingomyelin hydrolysis by sphingomyelinase. (B) GM3 ganglioside hydrolysis by entrapped neuraminidase. Vesicle composition was sphingomyelin![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) phosphatidylethanolamine:cholesterol (2 phosphatidylethanolamine:cholesterol (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) + 10 mol% GM3 on the outer leaflet only. 1) + 10 mol% GM3 on the outer leaflet only. | ||
6. The cyclical nature of scientific research
Perusal of the present and of the opening paper in this volume reveals a number of coincidences that are probably not due to chance. They rather reveal that very few, if any, of the investigations dealt with in these Faraday Discussions have reached completion. Perhaps we will never bring to a close any of our inquisitions. Is it, then, possible, to write any Closing Remarks about this, or any other scientific meeting?When one reads the scientific literature with a certain temporal perspective, the idea of a circular/cyclical return of questions and answers (less so of methods) is almost inevitable. One could even argue that revisiting the classical problems is often accompanied by the generation of new ideas. Science moves in circles (or appears to), just like time moves in cycles (or appears to), just like most celestial bodies are circular/spherical (or appear to be). The spherical shape, and its circular projection on the plane, have fascinated the scientists since the earliest stages of philosophical and scientific endeavor.28 Aristotle's universe was spherical, with the Earth taking the central place. Also, a perfect sphere was the natural shape of heavenly bodies and uniform rotation in circles was the natural state of their motion.
From the Middle Ages, Islamic culture conceived the structure of the human soul and of the intellect as reflecting the spherical shape of the skies and of the heavenly motions. Also, Muslims believe that God does not have a beginning or an end, like the concept of infinity conveyed by the image of a circle. An important XVIIIth century manuscript,29 “The Emperor's Gift”, recently discovered at the Gulbenkian Museum, Lisbon, is a graphic encyclopedia about Heaven, the World and Humanity, presented in 108 circular diagrams, an example of which is shown in Fig. 4.
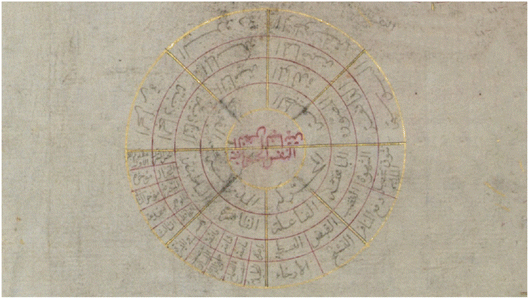 | ||
| Fig. 4 The soul: a diagram from “The Emperor's Gift”.29 Upper half: the ‘vegetative soul’, it concerns the simplest fundamental ‘nature’ and ‘faculties’ of all beings. Lower half: the ‘animal soul’, which relates to ‘movement’ and ‘perception’. | ||
What is, then, the meaning of progress? Is progress compatible with the cyclical return of our basic questions and research endeavors? This conundrum has been, if not solved, at least beautifully exposed by a poet, T. S. Eliot, who wrote:30
“We shall not cease from exploration
And the end of all our exploring
Will be to arrive where we started
And know the place for the first time”.
Data availability
The original data for Fig. 3 (bottom) are available from the author upon reasonable request.Conflicts of interest
The author discloses no conflicts of interest.Acknowledgements
This work was funded in part by the Spanish Ministry of Science, Innovation, and Universities (MCIU), Agencia Estatal de Investigación (AEI), Fondo Europeo de Desarrollo Regional (FEDER) (Grant No. PID2021-124461NB-I00), the Basque Government (Grant No. IT1625-22), Fundación Biofísica Bizkaia, and the Basque Excellence Research Center (BERC) program of the Basque Government.References
- F. M. Goñi and J. L. Arrondo, A study of phospholipid phosphate groups in model membranes by Fourier transform infrared spectroscopy, Faraday Discuss. Chem. Soc., 1986, 81, 117–126 RSC.
- J. F. Danielli and H. Davson, A contribution to the theory of permeability of thin films, J. Cell. Comp. Physiol., 1935, 5, 495–508, DOI:10.1002/jcp.1030050409.
- S. J. Singer and G. L. Nicolson, The fluid mosaic model of the structure of cell membranes, Science, 1972, 175(4023), 720–731 CrossRef CAS PubMed.
- A. L. Hodgkin and A. F. Huxley, Action potentials recorded from inside a nerve fibre, Nature, 1939, 144, 710–711 CrossRef.
- A. L. Hodgkin and A. F. Huxley, A quantitative description of membrane current and its application to conduction and excitation in nerve, J. Physiol., 1952, 117, 500–544 CrossRef CAS PubMed.
- J. C. Skou, The influence of some cations on an adenosine triphosphate from peripheral nerves, Biochim. Biophys. Acta, 1957, 23, 394–401 CrossRef CAS PubMed.
- P. Mitchell, A general theory of membrane transport from studies of bacteria, Nature, 1957, 180(4577), 134–136, DOI:10.1038/180134a0.
- V. Luzzati, Biological significance of lipid polymorphism: the cubic phases, Curr. Opin. Struct. Biol., 1997, 7, 661–668, DOI:10.1016/s0959-440x(97)80075-9.
- D. Chapman, Phase transitions and fluidity characteristics of lipids and cell membranes, Q. Rev. Biophys., 1975, 8, 185–235, DOI:10.1017/s0033583500001797.
- L. L. van Deenen, Structural organization and dynamics of phospholipids in red cell membranes, Prog. Clin. Biol. Res., 1979, 30, 451–456 CAS.
- D. M. Small, The Physical Chemistry of Lipids: from Alkanes to Phospholipids, Plenum, Melbourne, 1986 Search PubMed.
- M. J. Schleiden, Beiträge zur Phytogenesis, Arch. Anat. Physiol. wiss. Med., 1838, 1, 137–176 Search PubMed.
- T. Schwann, Mikroskopische Untersuchungen Über Die Uebereinstimmung in Der Struktur Un Dem Waschstum Der Thiere Und Pflanzen, Reimer, Berlin, 1839 Search PubMed.
- E. Overton, Ueber die allgemeinen osmotischen Eigenschaften der Zelle, Vierteljahresschr. Naturforsch. Ges. Zürich., 1899, 44, 88–135 Search PubMed.
- P. Walde, et al., Autopoietic self-reproduction of fatty acid vesicles, J. Am. Chem. Soc., 1994, 116, 11649–11654 CrossRef CAS.
- A. Rendón, et al., Model systems of precursor cellular membranes: long-chain alcohols stabilize spontaneously formed oleic acid vesicles, Biophys. J., 2012, 102, 278–286, DOI:10.1016/j.bpj.2011.12.026.
- E. Racker and W. Stoeckenius, Reconstitution of purple membrane vesicles catalyzing light-driven proton uptake and adenosine triphosphate formation, J. Biol. Chem., 1974, 249, 662–663 CrossRef CAS PubMed.
- T. L. Steck, et al., Inside-out red cell membrane vesicles: preparation and purification, Science, 1970, 168(3928), 255–257, DOI:10.1126/science.168.3928.255.
- M. S. Bretscher, Human erythrocyte membranes: specific labelling of surface proteins, J. Mol. Biol., 1971, 58, 775–781, DOI:10.1016/0022-2836(71)90039-8.
- M. Bogdanov, et al., A polytopic membrane protein displays a reversible topology dependent on membrane lipid composition, EMBO J., 2002, 21, 2107–2116 CrossRef CAS PubMed.
- G. Pabst and S. Keller, Exploring membrane asymmetry and its effects on membrane proteins, Trends Biochem. Sci., 2024, 49, 333–345 CrossRef CAS PubMed.
- S. K. Sahu, et al., Phospholipid scramblases: an overview, Arch. Biochem. Biophys., 2007, 462, 103–114 CrossRef CAS PubMed.
- F. X. Contreras, et al., Transbilayer (flip-flop) lipid motion and lipid scrambling in membranes, FEBS Lett., 2010, 584, 1779–1786 CrossRef CAS PubMed.
- A. J. Verkleij, et al., The asymmetric distribution of phospholipids in the human red cell membrane. A combined study using phospholipases and freeze-etch electron microscopy, Biochim. Biophys. Acta, Biomembr., 1973, 323, 178–193 CrossRef CAS PubMed.
- M. Doktorova, et al., Cell membranes sustain phospholipid imbalance via cholesterol asymmetry, Cell, 2025, 188, 2586–2602.e24, DOI:10.1016/j.cell.2025.02.034.
- F. X. Contreras, et al., Sphingomyelinase activity causes transbilayer lipid translocation in model and cell membranes, J. Biol. Chem., 2003, 278, 37169–37174, DOI:10.1074/jbc.M303206200.
- F. X. Contreras, et al., Asymmetric addition of ceramides but not dihydroceramides promotes transbilayer (flip-flop) lipid motion in membranes, Biophys. J., 2005, 88, 348–359, DOI:10.1529/biophysj.104.050690.
- A. Alonso, et al., The circle game, Rev. Int. Estud. Vascos, 2024, 69, 2, DOI:10.61879/riev692zkia202402.
- Power of the Word V, https://gulbenkian.pt/museu/en/power-of-the-word-v/.
- T. S. Eliot, Little Gidding, from “Four Quartets”, Faber and Faber, London, 1944 Search PubMed.
| This journal is © The Royal Society of Chemistry 2025 |
