Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Covalent functionalization of MoSe2 nanosheets with hydrogen bond-donating functionalities for the sensing of nitroaromatics
Rabiaa
Hajlaoui
ab,
Sabrine
Baachaoui
 b,
Sami
Ben Aoun
b,
Sami
Ben Aoun
 c,
Said
Ridene
c,
Said
Ridene
 *a and
Noureddine
Raouafi
*a and
Noureddine
Raouafi
 d
d
aAdvanced Materials and Quantum Phenomena Laboratory, Department of Physics, Faculty of Sciences of Tunis, Tunis El Manar University, 2092, Tunis, Tunisia. E-mail: said.ridene@fst.utm.tn
bAnalytical Chemistry and Electrochemistry Lab (LR99ES15), Department of Chemistry, Faculty of Sciences of Tunis, University of Tunis El Manar, 2092, Tunis, Tunisia
cDepartment of Chemistry, Faculty of Science, Taibah University, P. O. Box 30002, Al-Madinah Al-Munawwarah, Saudi Arabia
dBasic and Applied Scientific Research Center (BASRC), Imam Abdulrahman Bin Faisal University, P. O. Box 1982, Dammam, 31441, Saudi Arabia
First published on 15th January 2026
Abstract
Nitroaromatic compounds serve as indicators of environmental pollution and are specifically related to contamination by pesticides and explosive residues. Consequently, the development of sensitive sensing materials for their detection is of significant interest in environmental monitoring. This study investigates the chemical functionalization of molybdenum diselenide (MoSe2) with organic halogenated groups possessing hydrogen-bonding donor functionalities (i.e., –CONH2, –CO2H, and –SO3H), with the objective of employing them as sensing materials for the detection of selected nitroaromatics, including nitrobenzene (NB), m-nitrotoluene (m-NT), p-nitrotoluene (p-NT), p-nitrophenol (p-NP), picric acid (TNP), and p-nitroaniline (p-NA). These findings indicate that the chemical tethering of functional alkyl groups enhances physical adsorption, with chemical reaction energies 100 times greater than the physical adsorption energies. Electronic properties, such as the density of states (DOS), projected DOS, and band structures, demonstrate significant alterations in conduction and bandgap modulation, with a reduction of 0.5 eV. In the presence of target molecules, high adsorption energies were observed, particularly in the range of –1.84 to –2.26 eV, notably for MoSe2/2c bearing the –SO3H moiety. The partial charges, electronic density differences, and recovery times further corroborated the potential application of these materials in assessing pollution by nitroaromatic compounds for environmental monitoring in ecosystems.
1. Introduction
Nitroaromatic compounds (NACs) are a significant class of organic pollutants widely used in the production of explosives, dyes, pesticides, pharmaceuticals, and fine chemicals.1–4 They are characterized by the presence of one or several nitro groups attached to an aromatic ring, which confer high chemical stability and strong electron-withdrawing properties. After use, nitroaromatic residues persist in nature and contaminate food and water supplies. For instance, the breakdown of the widely used organophosphorus pesticide parathion yields 4-nitrophenol, which has a half-life of about 40 days under aerobic conditions and longer under anaerobic conditions and may persist in soil for 3–4 months after use.5,6 Many researchers have made substantial progress in the detection of nitroaromatic compounds.7–10 Nitrobenzene (NB), m-nitrotoluene (m-NT), p-nitrotoluene (p-NT), picric acid (TNP), 4-nitrophenol (p-NP) and p-nitroaniline (p-NA) are particularly noteworthy because of their environmental persistence, toxicity, and role in military and industrial applications.11 For example, p-NA is associated with hematological, splenic, and renal toxicities and is classified as a potential Group 2B carcinogen by the IARC.12 Even at low concentrations, NACs pose significant risks to human health by affecting the liver, blood, and nervous system and can lead to long-lasting contamination of soil and groundwater.13,14 These hazardous effects highlight the urgent need to develop sensitive and selective sensing methods based on nanomaterials with tailored properties.15,16A survey of recent literature shows that particular attention has been devoted to the design of responsive small-molecule systems for the detection of nitroaromatics such as trinitrophenol, trinitrotoluene (TNT), dinitrotoluene (DNT), dinitrobenzene (DNB), nitrobenzene, and nitrotoluene (NT).17–19 For instance, melanin oligomers have been used to detect nitroaromatics, showing a high affinity for TNT and TNP.17 Only a few reports have focused on the study of new materials useful for sensing nitroaromatics.20–22 The Perdew–Burke–Ernzerhof (PBE) functional is widely used in material-based studies, illustrating its robustness in solid-state quantum simulations.23 The interaction energy ranged between −1.1 and −2.8 eV for transition metal-modified materials18,21 and between −0.1 and −0.7 eV for sensing materials relying on hydrogen bonding and other weak physical interactions.18,19,24 For instance, Hashmi et al. used pentacarbon dinitride (C5N2) nanosheets for nitroaromatic detection,25 which showed selectivity in the order TNP > TNT > DNB. Functionalized BC3 nanosurfaces have also been used to detect various nitroaromatics.26 Luminescent organic metal frameworks have been used to selectively detect NB in the presence of benzene and acetone.22 C24-TM materials, where TM is Sc, Cr, or Mn, showed higher interaction energies of −2.0 to −2.8 eV in the presence of nitrobenzene.21
Numerous 2D materials, such as graphene, MoS2, WS2, and other TMDs, have been studied for nitroaromatic sensing applications. Despite these encouraging instances, the detection of NACs in these systems is primarily controlled by π–π stacking and charge–transfer interactions between the conductive surface and aromatic rings, which frequently lead to limited chemical selectivity and moderate sensitivity.27,28 Similarly, virgin, doped, or defect-engineered TMD monolayers have been used in extensive research on MoS2 and WS2-based sensors, where the sensing mechanism is primarily governed by physisorption and weak charge transfer effects.29 These methods can increase the strength of adsorption, but they typically lack chemical recognition sites, which limit their selectivity towards structurally identical nitroaromatic compounds.
The conjugated aromatic structure and electron-deficient nitro groups favor charge–transfer interactions with electron-rich surfaces. These properties make NACs ideal targets for detection using nanomaterials, especially 2D materials, whose active surfaces can interact through π-stacking, dipole–dipole interactions, or hydrogen bonding.25,30 However, pristine 2D materials, such as transition metal dichalcogenides, have limited chemical reactivity and reduced adsorption efficiency and selectivity towards complex molecules, such as NACs.31 To enhance the sensing performance, surface functionalization strategies, including covalent and non-covalent approaches, have been widely explored.32–34 Functionalization introduces active sites that improve the charge transfer between the surface and the target analyte. Recent studies have shown that chemical modification of TMDs can significantly tune their electronic and adsorption properties.35–37 Nevertheless, most reported functionalization strategies focus on MoS2 or other TMDs, while covalent functionalization of MoSe2 for chemical sensing applications, particularly for nitroaromatic compounds, remains largely unexplored.38 MoSe2, a prominent member of the TMDs family, has recently attracted significant attention owing to its unique two-dimensional layered structure and exceptional electronic properties. As a single layer, MoSe2 exhibits a direct bandgap, which makes it highly versatile for electronic and optoelectronic applications.39–41
Recent advances in flexible chemical sensors have explored the use of 2D materials and nanostructured composites for gas and nitroaromatic detection. Metal-doped MoS2 nanoflowers demonstrated high SO2 sensitivity at room temperature with DFT-confirmed charge transfer.42 MXene/metal–organic framework-derived CuO nanohybrids enabled self-powered ammonia sensing with high sensitivity and flexibility,43 while ZnO nanoflower–cellulose acetate/polyaniline composites improved ammonia detection via QCM devices.44 Despite these advances, most sensors rely on pristine or doped materials, where interactions are dominated by physisorption, limiting selectivity. In this context, this work introduces the covalent functionalization of MoSe2 with hydrogen-bond donor groups to enhance charge transfer and provide specific binding sites for nitroaromatic compounds, improving the sensitivity and selectivity compared to existing designs. However, despite these promising properties, MoSe2 has been significantly less explored for nitroaromatic hydrocarbon sensing compared to other TMDs such as MoS2, with only a few studies addressing its interactions with NAC molecules.45–47 Its high electrical conductivity and strong surface activity make it a promising candidate for gas-sensing applications, particularly for the detection of volatile compounds. Alkyl halide molecules can alter the local electronic density, adjust the Fermi level, and promote specific interactions with electron-deficient analytes such as NACs. These modifications can potentially increase the sensitivity and selectivity for the detection of toxic or explosive compounds.48,49 Importantly, although nitro groups are strong hydrogen bond acceptors, the deliberate introduction of hydrogen bond donor functional groups via covalent functionalization of MoSe2 has not been reported for NAC sensing. Addressing this gap is a central focus of the present work.
In this study, we investigated the covalent functionalization of 1H-MoSe2 using halogenated organic precursors, specifically iodoacetamide, iodoacetic acid, and iodomethanesulfonic acid. These alkyl halides are initially adsorbed physically on the substrate surface, followed by a chemical reaction in which the carbon–iodine bond is cleaved. The iodine atom is substituted by selenium in the MoSe2 lattice, resulting in the formation of a stable carbon–selenium (C–Se) covalent bond. The newly introduced active sites tune the local electronic environment of the substrate, thereby enhancing its interactions with electron-deficient nitroaromatic compounds such as NB, m-NT, p-NT, TNP, p-NA, and p-NP. Using density functional theory (DFT) calculations, we systematically explored the adsorption behavior, charge redistribution, and electronic structural changes induced by functionalization and subsequent analyte binding. These results provide detailed insights into how C–Se covalent bonding modulates the sensing properties of MoSe2, offering promising strategies for the design of high-performance two-dimensional sensors for environmental monitoring and security applications.
2. Methodology
Simulations were performed using the Quantum ESPRESSO package (v7.3).50 The generalized gradient approximation in the PBE form was used to describe the exchange–correlation interactions between electrons.51 GBRV high-throughput pseudopotentials were employed for all the atomic species.52 The valence electron wavefunctions and charge densities were expanded in a plane-wave basis set with kinetic energy cutoffs of 60 and 540 Ry, respectively, to ensure accurate convergence of the total energies and forces. A 5 × 5 MoSe2 supercell was used in the functionalization and adsorption studies using a 4 × 4 × 1 Monkhorst–Pack k-point grid, which was sufficiently dense to ensure converged total energies and accurate geometry.53 A denser 12 × 12 × 1 k-point grid was employed to ensure convergence of the density of states. Non-spin-polarized calculations were performed since the MoSe2 monolayer and NAC compounds were not magnetic and contained no unpaired electrons. Test calculations confirmed that including spin polarization does not significantly affect the geometries, adsorption energies, or electronic properties.For electronic optimization, the self-consistent field energy convergence was set to 10−6 Ry. Geometry relaxation was performed until the total energy variation was less than 10−6 Ry, and the residual atomic forces were less than 10−3 Ry Bohr−1. A vacuum spacing (25 Å) was applied along the z-direction to avoid interactions between the periodic images of the MoSe2 monolayer. For the adsorption studies, van der Waals interactions were accounted for using the DFT-D3 (version 4) method with Grimme's correction.54,55 The functionalization of MoSe2 with halogenated molecules was modeled at various adsorption sites (top, hollow, and bridge) to identify the most stable configurations. These computational parameters guarantee the stability and accuracy of the optimized geometry and electronic structure calculations for pristine and functionalized MoSe2 systems.
3. Results and discussion
3.1. Structural model and adsorption sites
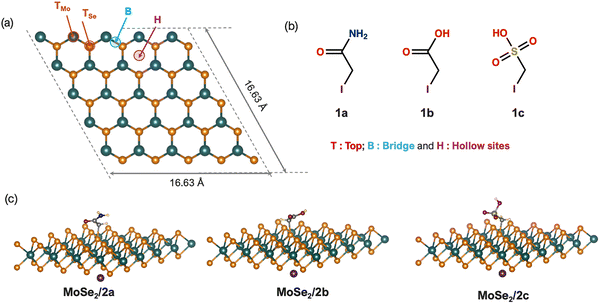
The pristine 1H-MoSe2 monolayer consists of a single layer of molybdenum atoms sandwiched between two layers of selenium atoms, forming trigonal prismatic coordination. In the supercell, each lattice vector was expanded five times along the a- and b-directions, resulting in a periodic slab sufficiently large to accommodate adsorbed halogenated molecules without introducing artificial interactions between periodic images. The monolayer consisted of 25 Mo and 50 Se atoms. A vacuum of 25 Å was introduced in the direction perpendicular to the plane to avoid inter-plane interactions. The lateral dimensions of the simulation box are 16.63 Å × 16.63 Å. For hexagonal TMD monolayers, the optimized lattice preserves the P63/mmc (No. 194) space-group symmetry. This supercell ensured adequate spatial separation between adsorbates and accurate electronic structure estimates by offering a suitable framework for the systematic investigation of covalent functionalization and adsorption using DFT. We investigated the physisorption of three halogenated molecules of interest, iodoacetamide (1a), iodoacetic acid (1b), and iodomethanesulfonic acid (1c). All three compounds contain an iodine atom but differ in their terminal functional groups, which can interact with the MoSe2 surface through van der Waals forces, dipole–surface interactions, or weak coordination bonds. For each molecule, adsorption was explored at four distinct sites on the MoSe2 monolayer, as depicted in Fig. 1. This systematic evaluation allowed the identification of the most energetically favorable adsorption configuration for each functionalized molecule, providing insight into the surface reactivity and potential sensing performance.3.2. Evaluation of the energetic and electronic properties
After performing full geometric relaxation for each adsorption configuration, the energetic stability and local geometric characteristics of the resulting structures were examined. The adsorption energy (ΔEads) of each molecule was calculated using eqn (1).| ΔEads = Eadsorbate+MoSe2 − Eadsorbate − EMoSe2 | (1) |
We investigated the interactions between nitroaromatic compounds and functionalized MoSe2 surfaces to assess their efficacy as chemical detectors. The targeted molecule was placed close to the modified surface of each structure, and the full system was relaxed. The interaction energies (ΔEint) were calculated using eqn (2).
| ΔEint = ENACs+fMoSe2 − ENACs − EfMoSe2 | (2) |
A study of the charge density difference (Δρ) was conducted to gain a better understanding of the interaction mechanism between the functionalized surface and target molecules. The differential charge density was computed using eqn (3):
| Δρ = ρcomplex – ρfMoSe2 – ρtarget | (3) |
The recovery time (τ) represents the time required for an adsorbed molecule to desorb from the surface, indicating the reusability and response reversibility of the sensor.56 This was calculated using the transition-state approximation theory,57 which relates the desorption rate to the adsorption energy and temperature, and can be determined using eqn (4):
| τ = ν−1exp(|ΔEint|/KBT) | (4) |
3.3. Physisorption analysis
The geometric configurations of the systems formed by the physical adsorption of three halogenated molecules, iodoacetamide (1a), iodoacetic acid (1b), and iodomethanesulfonic acid (1c), on the MoSe2 surface were optimized for each considered site (Fig. 1b). The top and side views of the geometries obtained for each case are shown in Fig. S1 (see SI), highlighting the relative orientations of functional groups and iodine atoms with respect to the surface. After the full geometric relaxation of each configuration, the adsorbed structures were analyzed in terms of energy stability and local geometry. The minimum distances between the iodine and selenium atoms from the surface (Se–I) were extracted for each configuration to evaluate the adsorption proximity.The adsorption energies showed that the interaction between adsorbates and the MoSe2 surface was weak, which is characteristic of van der Waals-type interactions. As presented in Table 1, the energy values varied between −0.013 and −0.033 eV, confirming the physical adsorption. Among the different positions studied, the bridge configuration was systematically the most stable for all three molecules, with a minimum energy of −0.033 eV. This preference can be attributed to the better distribution of the interaction between the iodinated group and the surface.
| Systems | ΔEads/eV | d Se–X/Å |
|---|---|---|
| a TSe position. b TMo position. c Bridge position. d Hollow position. | ||
| MoSe2/1a | −0.027a | 4.59 |
| −0.031b | 3.85 | |
| −0.032c | 3.79 | |
| −0.018d | 4.17 | |
| MoSe2/1b | −0.021a | 4.36 |
| −0.032b | 3.86 | |
| −0.033c | 3.94 | |
| −0.013d | 5.12 | |
| MoSe2/1c | −0.022a | 3.94 |
| −0.018b | 4.01 | |
| −0.033c | 4.08 | |
| −0.029d | 4.14 | |
| MoS2/2a | −2.11 | 2.02 |
| MoS2/2b | −1.93 | 2.04 |
| MoS2/2c | −2.00 | 2.03 |
The minimum distances Se–I associated with the bridge configuration range from 3.79 Å for MoSe2/1a to 4.08 Å for MoSe2/1c, which confirms that the molecules remain at a reasonable distance from the surface without direct bond formation.
3.4. Covalent functionalization
The chemical functionalization of MoSe2 using chemical reagents relies on a substitution mechanism involving breaking of the C–I bond. Under favorable conditions (thermal energy or assistance by surface defects), second-order nucleophilic substitution replaces the iodide ion with a nucleophilic doublet from the selenium atom exposed on the surface of MoSe2, leading to the formation of a C–Se covalent bond and the stable grafting of the functional group. From the perspective of chemical adsorption, the formation of a C–Se covalent bond between the halogenated molecule and MoSe2 constitutes a strong and irreversible interaction.The optimized geometries of the functionalized MoSe2 systems (Fig. 1c) revealed the successful formation of a stable C–Se covalent bond after cleavage of the carbon–iodine bond. This bond enhances the electronic integration of the molecule within the MoSe2 lattice, potentially modifying its electronic structure and sensing properties. During the functionalization process, the released iodide ions were positioned beneath the surface to maintain overall charge neutrality, preventing the system from carrying a positive net charge, which could otherwise result in unphysical and unreliable adsorption energies. By preserving neutrality, the calculations provided a realistic assessment of the structural stability, electronic configuration, and potential detection performance of covalently functionalized MoSe2–molecule complexes.
The results of chemical adsorption are summarized in Table 1, where for each molecule, the chemical adsorption energy ΔEads and the covalent distance C–Se, indicative of the strength of the bond formed, are indicated. The analysis of the results reflected significant stabilization of the system after the formation of the covalent bond Se–C. Among the studied reagents, compound (1a) exhibited the most negative adsorption energy (−2.11 eV), which indicates a more favorable reaction than the other two compounds. This affinity can be attributed to the nature of the amide group, which promotes efficient alignment of the reactive carbon with the surface. Iodomethanesulfonic acid (1c) has an adsorption energy of –2.00 eV, while iodoacetic acid (1b) is slightly less attracted to the surface (–1.93 eV), although all energies remain within a narrow range, illustrating comparable reactivity for the three structures.
For the covalent distance Se–C, values vary very little: they range from 2.02 Å to 2.04 Å, which is consistent with the formation of a typical stable covalent bond between a selenium atom and a sp3 carbon atom. These results suggest that these molecules can form robust chemical bonds with the MoSe2 surface, while maintaining sufficient structural flexibility.
The different functional groups (i.e., –CONH2 in 1a, –CO2H in 1b, and –SO3H in 1c) played a crucial role in the final functionality of the modified surface. Indeed, these groups can influence the polarity of the surface, the possibility of hydrogen interactions, and the molecular recognition ability in applications such as detection or catalysis. Thus, although the grafting mechanism remains identical, the chemical nature of the R group strongly modulates the physicochemical properties of the functionalized material.
3.5. Electronic properties
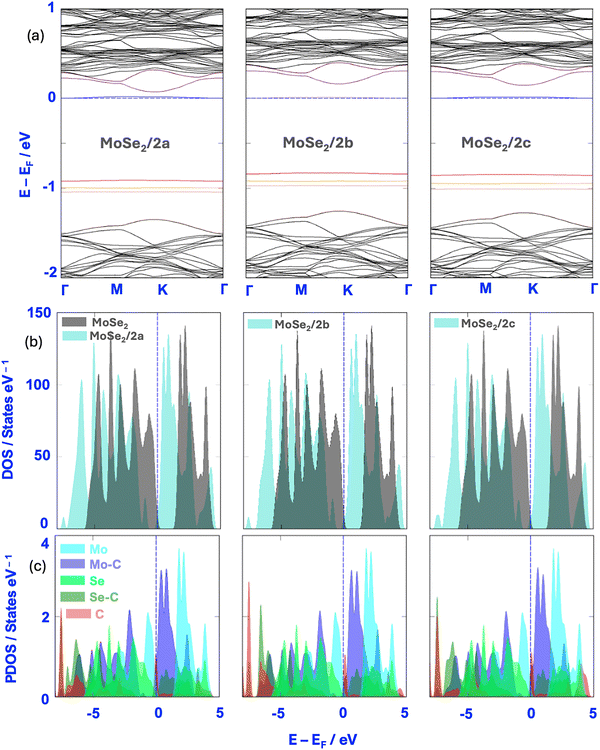
The electronic band structures of functionalized MoSe2 materials show a significant reduction in the bandgap energy, ranging from 0.91 to 0.83 eV compared to pristine MoS2 (Eg = 1.43 eV). This can be attributed to chemical functionalization, which leads to the formation of localized states near the Fermi level. These states manifest as flat bands in the band structure, indicating low dispersion and high localization, typically on the adsorbed molecule. This effect is more pronounced with highly polar groups, such as –CO2H and –SO3H, which induce stronger perturbations in the electronic structure. Furthermore, the adsorbate is positively charged, and its charge is fully distributed over the supercell, which can introduce new charge carriers into the material. It is important to note that the material retains its semiconducting nature, which makes these functionalized systems promising candidates for chemical sensing applications based on band-structure modulation.
In the case of pristine MoSe2, the DOS is distributed mainly within the range of −6 to 5 eV, whereas for functionalized MoSe2, the distribution extends wider, ranging from −8 to 5 eV. This extension towards lower energies reflects the appearance of new deep electronic levels, originating from the orbitals of the functional molecule (amide, carboxyl, or sulfonate groups). The expanded DOS form indicates a broadening of the electronic response window, which is favorable for detection applications where sensitivity depends on subtle changes in the electronic profile. Overall, the three systems studied exhibited similar behavior in this shift tendency, confirming the systematic effect of covalent functionalization on the density of states of the base material.
3.6. Sensing of nitroaromatics
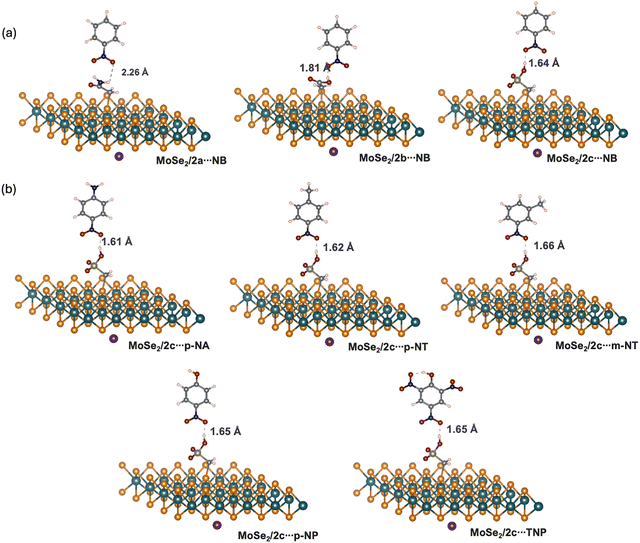 | ||
Fig. 3 Relaxed geometries of (a) NB⋯MoSe2/2a–c and (b) other NACs interacting with MoSe2/2c showing hydrogen bonds between the acidic O–H from the surface and O![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N from the target NACs. N from the target NACs. | ||
The results summarized in Table 2 show that the MoSe2/2c system exhibits the most stable interaction with nitrobenzene, displaying a more favorable adsorption energy of −2.07 eV and a minimum N–O⋯H hydrogen bonding distance of 1.64 Å, indicating a more powerful interaction between the surface and the target molecule.60 This may be due to the strong acidity of the OH from the sulfonic group (pKa = −1.961) compared with that of the carboxylic acid or carbamide groups (pKa of ∼4.762 and ∼15.1,63 respectively).
| Targets | Substrates | ΔEint/eV | HB lengths/Å |
|---|---|---|---|
| NB | MoSe2/2a | –1.84 | N–O⋯H = 2.26 |
| MoSe2/2b | –1.85 | N–O⋯H = 1.81 | |
| MoSe2/2c | –2.07 | N–O⋯H = 1.64 | |
| m-NT | MoSe2/2c | –2.03 | N–O⋯H = 1.66 |
| p-NT | –2.08 | N–O⋯H = 1.62 | |
| TNP | –1.95 | N–O⋯H = 1.65 | |
| p-NA | –2.26 | N–O⋯H = 1.61 | |
| p-NP | –2.09 | N–O⋯H = 1.65 |
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O⋯H–O hydrogen bonds formed between the sulfonic acid moiety and the nitroaromatic adsorbates. The associated bond lengths vary from 1.61 to 1.66 Å, reflecting differences in interaction strength across the tested molecules. Among these, p-nitroaniline exhibited the most pronounced interaction. These results highlight the capability of the functionalized MoSe2/2c material to interact strongly with a wide spectrum of nitroaromatic pollutants, underscoring its potential for comprehensive environmental monitoring of contaminated water and soil systems.
O⋯H–O hydrogen bonds formed between the sulfonic acid moiety and the nitroaromatic adsorbates. The associated bond lengths vary from 1.61 to 1.66 Å, reflecting differences in interaction strength across the tested molecules. Among these, p-nitroaniline exhibited the most pronounced interaction. These results highlight the capability of the functionalized MoSe2/2c material to interact strongly with a wide spectrum of nitroaromatic pollutants, underscoring its potential for comprehensive environmental monitoring of contaminated water and soil systems.
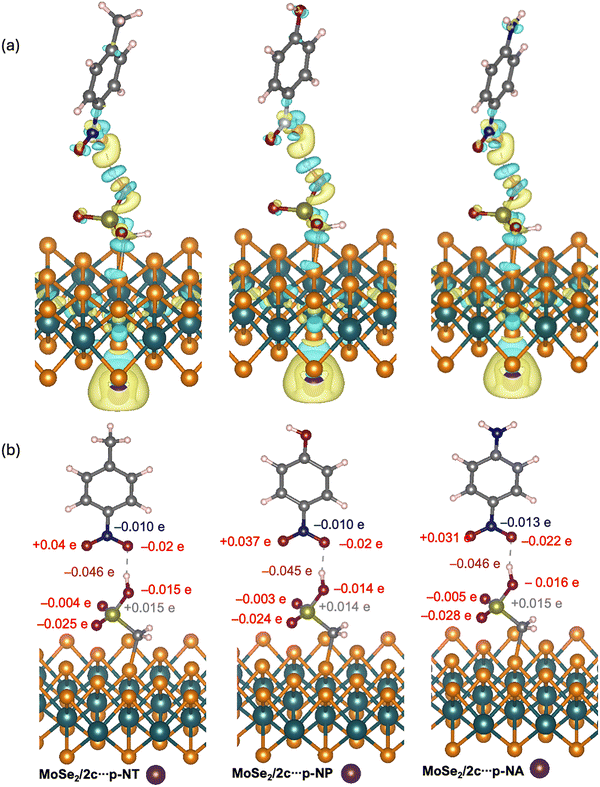
Fig. 4a shows a three-dimensional map of the charge density difference generated following the adsorption of analyte molecules (p-NT, p-NP, and p-NA) on the functionalized MoSe2/2c surface. The blue areas corresponding to the electron-depleting regions (charge density loss) are located mainly around the oxygen atoms of the –NO2 group of the analyte molecules, as well as partly around the sulfonic acid hydrogen. This depletion indicates charge donation from these regions to the functionalized surface. In contrast, the yellow areas indicate charge-accumulation regions (charge density gain), which are visible around the bonds between the functional groups of the two species, between the oxygen or nitrogen atoms of the nitro group, and between the sulfur and oxygen atoms of the sulfonic acid. The complementary charge distribution zones confirm the existence of partial charge transfer between the target molecules and the sensing surface. This phenomenon is a direct indicator of chemisorption interactions, which is consistent with the high adsorption energies obtained previously.
The acidic hydrogen from SO3H exhibits a negative charge variation (∼–0046e), reflecting electron gain due to hydrogen bonding with the electronegative O atom. The sulfur atom displays a positive charge change (∼+0.016e), indicating local electron loss. The hydrogen-bonded oxygen of SO3H is negatively charged because of charge accumulation due to the strengthening of the O–H bond during interaction, while the other two oxygens of the SO3H group have partial charges of (∼–0.026e) and (∼–0.006e). This asymmetry reflects the uneven distribution of the electrostatic field, which is likely related to the spatial orientation of the target molecules. Regarding the –NO2 grouping of molecules, the nitrogen atom (N) gains a small number of electrons (−0.010 and −0.013e), which is typical for its role in stabilizing the charge by resonance in –NO2. Among the two oxygen atoms, the one involved in hydrogen bonding gains a slightly higher charge (∼–0.020e), whereas the other loses electrons (∼+0.031e), suggesting an internal reorganization of the electron cloud within the group –NO2.
These partial load variations indicate that the hydrogen bond between the H of SO3H and the O of NO2 is the main anchor point of the interaction. Electron transfer is oriented from the acid group to the nitro group, which confirms the electrophilic nature of the latter. The entire transfer was consistent with a chemisorption interaction stabilized by local electron polarization, thus enhancing the sensitivity and selectivity of the sensor.
Charge density difference and partial charge analyses provide a direct link between the adsorption strength and sensing response. The significant charge redistribution induced by NAC adsorption, particularly through hydrogen bonding between the SO3H functional group and the –NO2 moieties, leads to modulation of the local carrier concentration near the Fermi level of the functionalized MoSe2 surface. The magnitude of the calculated charge transfer, as revealed by the Löwdin population analysis, quantitatively reflects the degree of electronic perturbation induced by the adsorbates. Therefore, stronger adsorption energies accompanied by larger charge transfer are expected to result in more pronounced conductivity variations. Although explicit charge transport calculations were not performed, the observed charge redistribution served as a reliable quantitative indicator of the change in conductivity, which is consistent with the operating principle of resistive 2D material-based sensors.
3.7. Recovery times
The recovery times as a function of temperature for MoSe2/2c with three target molecules is studied (Table 3). The value, calculated from the adsorption energies, indicated strong temperature dependence for all target molecules. At 298 K, these values were extremely high (on the order of 1019–1022 h), reflecting the stable chemical adsorption of NACs on the functionalized surface.| Entry | Systems | Recovery times/h | ||
|---|---|---|---|---|
| 298 K | 398 K | 498 K | ||
| 1 | MoSe2/2c⋯p-NT | 4.2 × 1019 | 6.1 × 1010 | 3.1 × 105 |
| 2 | MoSe2/2c⋯p-NA | 4.6 × 1022 | 1.1 × 1013 | 2.1 × 107 |
| 3 | MoSe2/2c⋯p-NP | 6.2 × 1019 | 8.1 × 1010 | 3.9 × 105 |
At higher temperatures, the recovery times decreased significantly, reaching 1010–1013 and 105–107 h, confirming the thermally activated nature of the desorption process at 400 and 500 K, respectively. The p-NA exhibits a distinct behavior: although its adsorption energy is the highest (2.26 eV), the estimated recovery time of approximately 2.1 × 107 h suggests faster desorption at high temperatures, which could result from local reorganization or partial relaxation of the bond between the NH2 group and the surface.
These results clearly show that an increase in temperature promotes the destruction of the adsorbed molecules and significantly reduces the recovery time. However, at relatively low temperatures, analyte retention indicates that detection relies mainly on strong chemisorption, providing the system with excellent sensitivity but limiting its ability to quickly regenerate. Among the three molecules, p-NA exhibited the strongest interaction with the functionalized surface, confirming its ability to selectively and stably detect the modified MoSe2.
4. Conclusions
This study demonstrates that the chemical functionalization of MoSe2 with halogenated organic groups containing hydrogen-bond donor functionalities (–CONH2, –CO2H, and –SO3H) markedly enhances its potential as a sensing material for detecting nitroaromatic pollutants. The introduction of these functional groups significantly strengthens the adsorption interactions, with the chemical binding energies exceeding the physical adsorption energies by nearly two orders of magnitude. Electronic structure analyses revealed pronounced modifications in the conduction band and a notable bandgap reduction (approximately 0.5 eV), confirming the strong electronic coupling between MoSe2 and adsorbed nitroaromatics. The adsorption energies in the range of –1.84 to –2.26 eV and the corresponding charge transfer patterns indicate high sensitivity, particularly for the MoSe2/2c system. These findings underscore the potential of functionalized MoSe2 as an efficient and tunable platform for detecting nitroaromatic contaminants such as NB, m-NT, p-NT, TNP, p-NP, and p-NA. Overall, this work provides valuable insights into the rational design of two-dimensional materials for environmental monitoring and pollutant detection in complex ecosystems.Author contributions
RH: investigation, conceptualization, and data curation, writing – original draft; SB: investigation, data curation, writing – original draft; SBA: writing – review & editing; SR: writing – review & editing; NR: methodology, supervision, validation, writing – review & editing.Conflicts of interest
The authors declare no conflict of interest.Data availability
Data for the paper can be found at the Harvard Dataverse: https://doi.org/10.7910/DVN/U2Q0PA.Supplementary information (SI): equilibrium positions for the physical adsorption of the iodated chemical modifiers on atop, bridge and hollow positions with the respective equilibrium distance to surface (Fig. S1) and band structure of the pristine MoSe2 (Fig. S2) that supports the results presented in the main manuscript. See DOI: https://doi.org/10.1039/d5ma01255d.
Acknowledgements
For computer time, this research used Shaheen III, managed by the Supercomputing Core Laboratory at King Abdullah University of Science & Technology (KAUST) in Thuwal, Saudi Arabia (project ref. k10103).References
- M. Fabin, M. Łapkowski and T. Jarosz, Appl. Sci., 2023, 13, 3991 CrossRef CAS.
- P. Ghosh, S. K. Saha, A. Roychowdhury and P. Banerjee, Eur. J. Inorg. Chem., 2015, 2851–2857 CrossRef CAS.
- G. Awasthi, R. Sharma, S. Sundarrajan, S. Ramakrishna and P. Kumar, Polymers, 2022, 14, 4643 CrossRef CAS PubMed.
- K. Anichina, N. Lumov, V. Bakov, D. Yancheva and N. Georgiev, Molecules, 2024, 29, 3475 Search PubMed.
- N. Pino and G. Peñuela, Int. Biodeterior. Biodegrad., 2011, 65, 827–831 CrossRef CAS.
- K. Tao, H. Tian, Z. Wang, X. Shang, J. Fan, M. Megharaj, J. Ma, H. Jia and W. He, Chemosphere, 2023, 311, 137116 CrossRef CAS PubMed.
- A. Chowdhury and P. S. Mukherjee, J. Org. Chem., 2015, 80, 4064–4075 CrossRef CAS PubMed.
- Y. Wang, A. La, C. Brückner and Y. Lei, Chem. Commun., 2012, 48, 9903–9905 Search PubMed.
- X. Sun, Y. Wang and Y. Lei.
- A. Deshmukh, S. Bandyopadhyay, A. James and A. Patra, J. Mater. Chem. C, 2016, 4, 4427–4433 RSC.
- T. Jadoon, T. Mahmood and K. Ayub, J. Mol. Liq., 2020, 306, 112878 CrossRef CAS.
- J. Yang, L. Xia, Z. Lin, Z. Tang, G. Li and Y. Hu, Chin. Chem. Lett., 2019, 30, 638–642 CrossRef CAS.
- M. Kaur, S. K. Mehta and S. K. Kansal, Spectrochim. Acta, Part A, 2017, 180, 37–43 CrossRef CAS PubMed.
- K.-S. Ju and R. E. Parales, Microbiol. Mol. Biol. Rev., 2010, 74, 250–272 CrossRef CAS PubMed.
- T. Kokulnathan, A. I. Jothi, S.-M. Chen, G. Almutairi, F. Ahmed, N. Arshi and B. AlOtaibi, J. Environ. Chem. Eng., 2021, 9, 106310 CrossRef CAS.
- Y. Ren, Z. Ma, T. Gao and Y. Liang, Molecules, 2023, 28, 4481 CrossRef CAS PubMed.
- J. P. Cachaneski-Lopes, F. Hawthorne, C. F. Woellner, T. L. Nelson, R. C. Hiorns, C. F. O. Graeff, D. Bégué and A. Batagin-Neto, ACS Omega, 2025, 10, 31908–31920 CrossRef CAS PubMed.
- R. Sainda, D. Chodvadiya, I. Zgłobicka, K. J. Kurzydłowski and P. K. Jha, J. Mol. Liq., 2024, 409, 125389 Search PubMed.
- A. L. Desai, N. P. Patel, J. H. Parikh, K. M. Modi and K. D. Bhatt, J. Fluoresc., 2022, 32, 483–504 CrossRef CAS PubMed.
- S. Sarfaraz, M. Yar, A. Ali Khan, R. Ahmad and K. Ayub, J. Mol. Liq., 2022, 352, 118652 CrossRef CAS.
- D. Paul, A. Vaidyanathan, U. Sarkar and B. Chakraborty, Struct. Chem., 2021, 32, 2259–2270 CrossRef CAS.
- L. Liu, X. Chen, J. Qiu and C. Hao, Dalton Trans., 2015, 44, 2897–2906 RSC.
- J. P. Perdew, K. Burke and M. Ernzerhof, Phys. Rev. Lett., 1996, 77, 3865–3868 CrossRef CAS PubMed.
- H. Sajid, M. Asif, K. Ayub, M. A. Gilani, M. S. Akhter and T. Mahmood, Surf. Interfaces, 2021, 27, 101587 CrossRef CAS.
- N. Nabeela, M. A. Hashmi, A. N. S. Saqib, A. Kamran and A. Lakhani, RSC Adv., 2024, 14, 30116–30126 RSC.
- S. Baachaoui, R. Hajlaoui, S. B. Aoun, A. Fortunelli, L. Sementa and N. Raouafi, Nanotechnology, 2024, 35, 425501 CrossRef CAS PubMed.
- X. Chen and B. Chen, Environ. Sci. Technol., 2015, 49, 6181–6189 CrossRef CAS PubMed.
- X. Lu, H. Qi, X. Zhang, Z. Xue, J. Jin, X. Zhou and X. Liu, Chem. Commun., 2011, 47, 12494–12496 Search PubMed.
- S. Ganesan, T. Kokulnathan and A. Palaniappan, ACS Appl. Nano Mater., 2025, 8, 7004–7014 CrossRef CAS.
- S. Baachaoui, S. Aldulaijan, F. Raouafi, R. Besbes, L. Sementa, A. Fortunelli, N. Raouafi and A. Dhouib, RSC Adv., 2021, 11, 7070–7077 RSC.
- J. Gupta, D. S. S. Kandkuri and S. Rattan, J. Mater. Sci., 2024, 59, 3689–3710 CrossRef CAS.
- G. Gu, Z. Cui, X. Du, P. He, C. Rong, H. Tao, G. Wei and Y. Xi, Adv. Funct. Mater., 2024, 34, 2408367 CrossRef CAS.
- T. Zahra, U. Javeria, H. Jamal, M. M. Baig, F. Akhtar and U. Kamran, Anal. Chim. Acta, 2024, 1316, 342880 CrossRef CAS PubMed.
- S. Ambreen, D. K. Gupta, H. Kumar, A. Sharma, S. Arun, S. Kumar, A. Saraswat and A. K. Mishra, Luminescence, 2025, 40, e70273 CrossRef CAS PubMed.
- R. Hajlaoui, S. Baachaoui, S. Ben Aoun, S. Ridene and N. Raouafi, ACS Omega, 2024, 9, 37953–37964 CrossRef CAS PubMed.
- D. Voiry, A. Goswami, R. Kappera, C. de C. C. e Silva, D. Kaplan, T. Fujita, M. Chen, T. Asefa and M. Chhowalla, Nat. Chem., 2015, 7, 45–49 CrossRef CAS PubMed.
- D. Voiry, A. Mohite and M. Chhowalla, Chem. Soc. Rev., 2015, 44, 2702–2712 Search PubMed.
- D. O. Li, M. S. Gilliam, X. S. Chu, A. Yousaf, Y. Guo, A. A. Green and Q. H. Wang, Mol. Syst. Des. Eng., 2019, 4, 962–973 Search PubMed.
- D. Zhang, H. Guan, W. Zhu, J. Yu, H. Lu, W. Qiu, J. Dong, J. Zhang, Y. Luo and Z. Chen, Opt. Express, 2017, 25, 28536–28546 Search PubMed.
- A. S. Murali, S. Lekshmi and B. Saraswathyamma, Tungsten, 2025, 7, 215–242 CrossRef.
- S. Kandar, K. Bhatt, N. Kumar, A. K. Kapoor and R. Singh, ACS Appl. Nano Mater., 2024, 7, 8212–8220 CrossRef CAS.
- D. Zhang, J. Wu, P. Li and Y. Cao, J. Mater. Chem. A, 2017, 5, 20666–20677 RSC.
- D. Wang, D. Zhang, Y. Yang, Q. Mi, J. Zhang and L. Yu, ACS Nano, 2021, 15, 2911–2919 CrossRef CAS PubMed.
- D. Zhou, Z. Kang, X. Liu, W. Yan, H. Cai, J. Xu and D. Zhang, Sens. Actuators, B, 2023, 392, 134072 CrossRef CAS.
- A. Mirzaei, M. Alizadeh, H. R. Ansari, M. Moayedi, Z. Kordrostami, H. Safaeian, M. H. Lee, T. Kim, J. Kim, H. W. Kim and S. S. Kim, Nanotechnology, 2024, 35, 332002 CrossRef CAS PubMed.
- S. Paria and P. Maity, ACS Appl. Nano Mater., 2024, 7, 16516–16524 CrossRef CAS.
- Aneesha and M. S. Mehata, Mater. Res. Bull., 2025, 185, 113287 CrossRef CAS.
- E. X. Yan, M. Cabán-Acevedo, K. M. Papadantonakis, B. S. Brunschwig and N. S. Lewis, ACS Mater. Lett., 2020, 2, 133–139 Search PubMed.
- A. Vaidyanathan, S. Lakshmy, G. Sanyal, S. Joseph, N. Kalarikkal and B. Chakraborty, Appl. Surf. Sci., 2021, 550, 149395 CrossRef CAS.
- P. Giannozzi, S. Baroni, N. Bonini, M. Calandra, R. Car, C. Cavazzoni, D. Ceresoli, G. L. Chiarotti, M. Cococcioni, I. Dabo, A. D. Corso, S. Fabris, G. Fratesi, S. de Gironcoli, R. Gebauer, U. Gerstmann, C. Gougoussis, A. Kokalj, M. Lazzeri, L. Martin-Samos, N. Marzari, F. Mauri, R. Mazzarello, S. Paolini, A. Pasquarello, L. Paulatto, C. Sbraccia, S. Scandolo, G. Sclauzero, A. P. Seitsonen, A. Smogunov, P. Umari and R. M. Wentzcovitch, J. Phys.: Condens. Matter, 2009, 21, 395502 Search PubMed.
- J. P. Perdew, K. Burke and M. Ernzerhof, Phys. Rev. Lett., 1996, 77, 3865–3868 CrossRef CAS PubMed.
- K. F. Garrity, J. W. Bennett, K. M. Rabe and D. Vanderbilt, Comput. Mater. Sci., 2014, 81, 446–452 CrossRef CAS.
- P. Wisesa, K. A. McGill and T. Mueller, Phys. Rev. B, 2016, 93, 155109 CrossRef.
- S. Grimme, A. Hansen, J. G. Brandenburg and C. Bannwarth, Chem. Rev., 2016, 116, 5105–5154 CrossRef CAS PubMed.
- S. Grimme, J. Antony, S. Ehrlich and H. Krieg, J. Chem. Phys., 2010, 132, 154104 CrossRef PubMed.
- S. Baachaoui, S. Aldulaijan, L. Sementa, A. Fortunelli, A. Dhouib and N. Raouafi, J. Phys. Chem. C, 2021, 125, 26418–26428 Search PubMed.
- J.-H. Li, J. Wu and Y.-X. Yu, Appl. Surf. Sci., 2021, 546, 149104 CrossRef CAS.
- K.-A. N. Duerloo, M. T. Ong and E. J. Reed, J. Phys. Chem. Lett., 2012, 3, 2871–2876 CrossRef CAS.
- P. P. Hankare, P. A. Chate, S. D. Delekar, V. M. Bhuse, M. R. Asabe, B. V. Jadhav and K. M. Garadkar, J. Cryst. Growth, 2006, 291, 40–44 CrossRef CAS.
- T. Steiner, Angew. Chem., Int. Ed., 2002, 41, 48–76 Search PubMed.
- D. B. Roitman, J. McAlister and F. L. Oaks, J. Chem. Eng. Data, 1994, 39, 56–60 CrossRef CAS.
- H. S. Harned and R. W. Ehlers, J. Am. Chem. Soc., 1933, 55, 652–656 CrossRef CAS.
- G. E. K. Branch and J. O. Clayton, J. Am. Chem. Soc., 1928, 50, 1680–1686 CrossRef CAS.
- M. Weingarth, N. Raouafi, B. Jouvelet, L. Duma, G. Bodenhausen, K. Boujlel, B. Schoellhorn and P. Tekely, Chem. Commun., 2008, 5981–5983 RSC.
- G. Cavallo, P. Metrangolo, T. Pilati, G. Resnati and G. Terraneo, in Halogen Bonding I: Impact on Materials Chemistry and Life Sciences, ed. P. Metrangolo and G. Resnati, Springer International Publishing, Cham, 2015, pp. 1–17 Search PubMed.
- E. Rahali, M. Zouaghi, J. Sanz, N. Raouafi and Y. Arfaoui, J. Comput. Chem., 2023, 44, 1426–1436 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |