The inverting mechanism of the metal ion-independent LanGT2: the first step to understand the glycosylation of natural product antibiotic precursors through QM/MM simulations†
Abstract
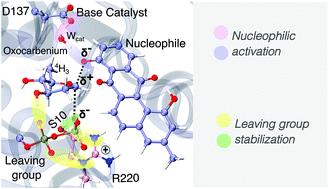
Glycosyltransferases (GTs) from the GT1 family are responsible for the glycosylation of various important organic structures such as terpenes, steroids and peptide antibiotics, making it one of the most intensely studied families of GTs. The target of our study, LanGT2, is a member of the GT1 family that uses an inverting mechanism for transferring olivose from TDP-olivose, the donor substrate, to the natural product tetrangulol (Tet), the precursor of the antibiotic landomycin A. X-ray crystallography in conjunction with mutagenesis experiments has revealed the catalytic significance of 3 amino acids (Ser10, Ser219 and Asp137), suggesting Asp137 as the base catalyst. In the absence of X-ray structures that include the acceptor substrate Tet, in silico experiments and MD simulations that have modeled ternary complexes propose that Asp137 could recruit a water molecule to facilitate the nucleophilic activation of Tet, since the distance between Asp137 and the nucleophile is too long to directly deprotonate the nucleophilic moiety. So far, there is no computational evidence regarding the precise mechanism by which LanGT2 catalyzes the transfer of olivose, which raises questions such as: is a water-assisted mechanism possible? and how does this metal ion-independent GT stabilize the growing negative charge of the diphosphate leaving group? In this work, the QM/MM approach was used to unravel the catalytic mechanism of LanGT2, and to identify the role of crucial catalytic amino acids at a molecular level. Our calculations show that the minimum energy path (MEP) describes an SN2-like mechanism, identifying an oxocarbenium ion-like TS in which the olivosyl moiety adopts a 4H3 conformation. Interactions established between the diphosphate group of TDP and Ser10, Ser219, Arg220 and His283 are key to stabilize the development of charge on the leaving group. Our work also suggests that a water-mediated proton transfer mechanism is feasible, in which the water molecule is key to stabilize the phenolate ion-like nucleophile in the TS. This is the first computational insight into the inverting mechanism of an antibiotic natural product GT, and its implications may serve to guide the design of new biocatalysts for natural product glycodiversification.
- This article is part of the themed collection: Mechanistic, computational & physical organic chemistry in OBC


 Please wait while we load your content...
Please wait while we load your content...