Perylene with peripherally fused pyrrole and pyridine: columnar packing, anti-Kasha dual emission and reactive oxygen species generation†
Jing
Cao
a,
Wenhao
Zhang
a,
Yeda
Ding
a,
Xuejin
Zhang
a,
Yuanyuan
Zhang
a,
Dan
Qiao
a,
Shumin
Biao
a,
Xuefei
Dong
a,
Jiayi
Song
a,
Gaole
Dai
 *b,
Ruiguo
Zhao
*a and
Qing
Wang
*b,
Ruiguo
Zhao
*a and
Qing
Wang
 *a
*a
aSchool of Chemistry and Chemical Engineering, Inner Mongolia University, 235 West University Street, Hohhot 010021, China. E-mail: qingwang@imu.edu.cn
bCollege of Material Chemistry and Chemical Engineering, Key Laboratory of Organosilicon Chemistry and Material Technology, Ministry of Education, Hangzhou Normal University, Hangzhou 311121, Zhejiang, China. E-mail: daigaole@hznu.edu.cn
First published on 15th April 2024
Abstract
Simultaneously fusing pyrrole and pyridine at perylene's opposite edges is facilely achieved in this work, yielding PerNN and its various derivatives. All targets demonstrate global diamagnetic ring current. Benefiting from a spatially separated donor–acceptor group within one planar skeleton, PerNN exhibits efficient narrow blue emission and columnar stacking. More importantly, its alkylated derivatives PerNN-MeCl and PerNN-MeI display anti-Kasha dual emission responsive to solvent polarity and reveal highly efficient ROS generation, paving the way to great potential applications.
The combinations of a donor (D) and acceptor (A) play a significant role in constructing functional electronic materials, among which efficient emitters especially require D–A composition to facilitate ΔEST and spin–orbit coupling (SOC).1 Owing to difficulties in synthesis, in most cases, D and A are connected through a single bond, a significant drawback of which is the low florescence photo-luminescence quantum yield (QY) and broad emission.2 Thus, compact D–A-type fused-ring systems with flat structures, though limited, are essential for obtaining some desired functions.3
Fusing polycyclic aromatic hydrocarbons (PAHs) with N-heterocycles, especially doping N heteroatoms marginally at the perimeters are always related to intriguing properties.4 Firstly, a pyrrole type heterocycle is electron rich with a five-membered ring, which will induce acid response, high strain and supramolecular interaction/assembly with electron acceptors.5 Conversely, a pyridine type heterocycle is a six-membered ring with strong electron affinity, which always afford high n-type mobility, stimulus response attributing to facile derivatization (e.g. hydrogen bonding, protonation, metal coordination, quaternizations, etc.) and strong intermolecular interactions.6 Secondly, N doping on the periphery is appreciably significant as it can involve global (anti-)aromaticity, facilitate heteroatom–heteroatom interactions and simplify post structural modifications.7 However, PAHs simultaneously fused with pyrrole and pyridine heterocycles are inadequate.8 To the best of our knowledge, studies on the synthesis and investigations of molecules with pyrrolic N and pyridinic N atoms doped peripherally at PAH's opposite edges are few. Thus, we aim to insert pyrrole and pyridine into PAHs’ opposing sides, developing a novel push–pull system.
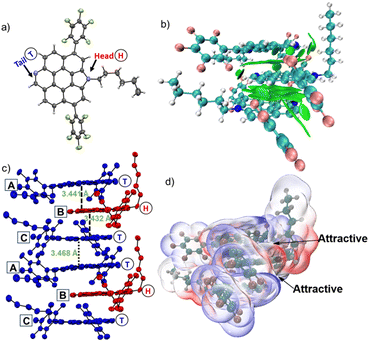
In this work, we focus on fusing pyridine and pyrrole heterocycles into perylene's two bay positions (Fig. 1a). The resultant molecule, PerNN, owns a rigid planar π skeleton with the characteristics of electron donor pyrrolic N atom and electron-deficient pyridinic N atoms being disposed at opposite edges. Primarily, on account of heteroatom doping at the zigzag edges and rigid backbone, these molecules are expected to exhibit high QY, small FWHM and global (anti-)aromaticity.5a,6b Later, dense molecular packing with strong intermolecular interactions may be generated, supporting the fact that molecules tend to approach each other in a complementary manner of electrostatic potential (ESP) to maximize electrostatic interaction energy between the spatially separated electron-donating edge and electron-accepting perimeter.9 As shown in Fig. 1c, the ESP map demonstrated obviously different electrostatic edges with the pyrrole border being relatively positive and pyridine margin being negative as expected. Thus, enhanced intermolecular interactions and simultaneous π stacking may be launched (Fig. 1d). Furthermore, the derivatization of pyrrole and pyridine will be effortless. For example, PerNN's alkylated, acidized, and even oxidized derivatives can be easily prepared via reactions at the pyridine site (Fig. 1b). The oxidized derivative, pyridine N-oxide, can feasibly help to enhance the binding capacity toward biological species and strengthen intermolecular interactions through hydrogen bonding.10 Alkylated compounds can enhance intramolecular D–A interactions.11
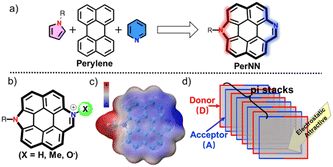 | ||
| Fig. 1 PerNN's (a) molecular design strategy, (b) derivatives, (c) ESP map and (d) possible packing mode. | ||
Synthesis of PerNN is straightforward (Scheme 1): the key precursor bis(perfluorophenyl)-substituted-nitrogen-annulated perylene (NP) was synthesized according to previous study.12 Then, NP undergoes a regioselective formylation to generate NP-CHO, using CH3OCHCl2 and SnCl4. Finally, NP-CHO is nitrogenated using azides as the nitrogen source, yielding the target PerNN. With parent PerNN in hand, we then acidify it with HCl to obtain PerNN-H. A more stable N-methylated compound PerNN-MeI was also synthesized by refluxing PerNN with iodomethane in acetonitrile. Later, ion exchange was performed by stepwise reactions with NH4PF6 and tetrabutylammonium chloride, producing PerNN-MeCl. The oxidized zwitterionic product PerNN-O was also synthesized by reacting it with mCPBA with the yield of 55%.
 | ||
| Scheme 1 Synthetic routes. DCM: dichloromethane; TFA: trifluoroacetic acid; mCPBA: m-chloroperbenzoic acid; TBAC: tetrabutylammonium chloride. | ||
Single crystals of PerNN suitable for X-ray crystallographic analysis were obtained by slow diffusion of hexane into the chloroform solution of PerNN, and XRD was measured at 193 K, which displayed a planar skeleton (Fig. 2a).13 Although the PerNN crystal had two bulky aryl substituents, it exhibited a well-ordered slipped lamellar packing arrangement in which three neighboring conjugated skeletons pack in an ABC mode as the repeating unit in the column along the a axis (Fig. 2c). The π–π distance in AB is 3.441 Å (dA–B), in BC is 3.432 Å (dB–C), and the two adjacent ABC pairs further interact through a secondary π–π stacking (dC–A = 3.468 Å). Notably, ABC unit is arranged in a tail–head–tail alternating mode (Fig. 2c). To understand the physical forces for such a unique alternating mode and for stabilizing the self-assembly, the ABC trimer cutout from the column was investigated using the independent gradient model based on Hirshfeld partition (IGMH)14 and ESP analysis.15 The visual molecular dynamics (VMD) program was used to visualize the isosurfaces of weak interactions.16 As shown in Fig. 2b, obvious green regions between the neighboring skeleton were observed, indicating the formation of strong intermolecular attractions. ESP mutual penetration graph clearly show that there is a ESP-complementary region between the positive pyrrole-positioned molecular head and negative pyridine-sited molecular tail (Fig. 2d). As a result, PerNN tend to arrange tail–head–tail stacking when they approach each other. On the contrary, tail–tail is mutually exclusive, thus neighboring dC–A is slightly larger than dA–B and dB–C owing to the repulsive Coulombic interaction. These simulations well explain the fact that electrostatic attractive peripheral pyrrole- and pyridine-subunits play a key role in such self-assembly mode.
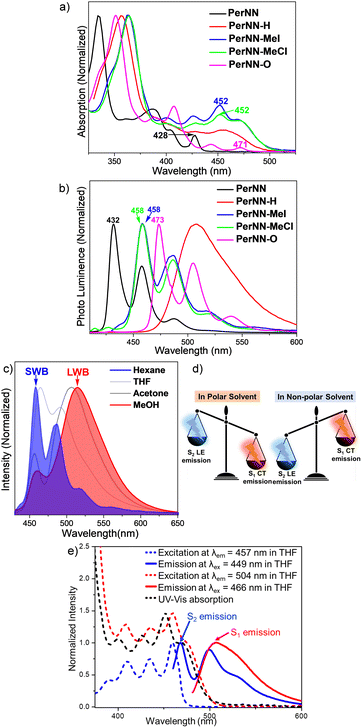
Optoelectronic properties of PerNN and its derivatives were explored using UV-vis-NIR absorption and photoluminescence (PL) spectroscopies in various solvents (Fig. 3a and b). For all compounds, their maximum absorption is within 325–375 nm range, with small to moderate band from 425 nm to 500 nm. Simulations suggested that these long-wavelength tiny bands (425 nm to 500 nm) are attributed to S0–S1/S2 transitions involving HOMO/HOMO−1 to LUMO/LUMO+1 orbitals (Fig. S25–S28 and Tables S8–S11, ESI†). Detailly, the transition intensity of methylated PerNN-MeI's and PerNN-MeCl's S0–S1 (around 470 nm) is solvent dependent (Fig. S8 and S14, ESI†), with negligible S0–S1 in the non-polar solvent, hexane, but obvious in polar solvents. These may indicate the charge transfer nature of the S0–S1 transition, which is in good agreement with DFT calculations (Fig. S24 and Table S9, ESI†). Overall, the parent PerNN owns the most blue-shifted absorption, while all derivations result in a bathochromic,shift. Notably, PerNN-MeI and PerNN-MeCl exhibit almost identical band structures, suggesting that counter anions affect the electronic structure slightly. All compounds are fluorescent with fluorescence quantum yield ranging from 8% to 62% in various solvents (Tables S1, S4–S7, ESI†). Except PerNN-H, all the other compounds demonstrated structured emission spectra and small Stokes shift owing to a rigid backbone, with PerNN being 4 nm, PerNN-O being 2 nm, PerNN-MeCl being 6 nm and PerNN-MeI being 11 nm. More importantly, solvatochromism is very negligible for PerNN, PerNN-O and PerNN-H both in absorption and emission, but a marked solvent polarity-dependent behavior is observed for methylated PerNN-MeCl's and PerNN-MeI's emission (Fig. 3c and Fig. S8, S14, ESI†). Observably, emission spectra for these two compounds in non-polar hexane is a fine-structured short wavelength band (SWB) (≈450–500 nm) tailing to 650 nm, but in several polar solvents (e.g. acetone, DMSO, MeOH, and MeCN), and obvious dual emission can be observed with the SWB gradually decreasing, and a featureless long wavelength band (LWB) (≈510 nm) progressively showed up (Fig. 3c and Fig. S8, S14, ESI†). Normally, photoluminescence of organic molecules originates only from the lowest excited state of a given spin multiplicity and is irrespective of the excitation wavelength, which is also known as the Kasha's rule.17 The significant overlap between the absorption bands and fluorescence of PerNN-MeCl and PerNN-MeI (Fig. S8 and S14, ESI†), their excitation-dependent emission behaviors (Fig. S11 and S14, ESI†) and excimer exclusion (polar solvent, MeOH, is good solvent for them, while hexane is the poor solvent, Fig. S9, ESI†) may indicate the possibility of anti-Kasha emissions. Subsequently, excitation and emission spectra were measured in THF to validate the anti-Kasha mechanism. As can be seen in Fig. 3e, selectively excite S0-1 (λex = 466 nm) led to a single fluorescence emission peaking at 480–600 nm, and the associated broad band can be hereafter denoted as S1-0. Conversely, selective excitation of S0-2 (λex = ≤ 450 nm) yielded a second structured band appearing with maximum around 450 nm, hereafter denoted as S2-0. Each excitation spectrum was mirror-image of their corresponding emission spectrum, and well resembled the UV-vis absorption spectrum, which suggests that the two emission bands originated from two different excited states (Fig. 3e). To supplementarily verify this anti-Kasha dual emission, calculations on PerNN-Me+ (without the I− or Cl− counter anion) using the Multiwfn program were then performed.14 According to calculations, the excited state of S2 is 0.33 eV higher in energy than that of the S1 state, and emission from S2 is centered at 461 nm, while S1 emission is around 527 nm (Fig. S29, ESI†). The simulations replicated experimental results well. Fig. 3c and Fig. S9, S12 (ESI†) reveal that dual emission is responsive to solvent polarity: LWB dominates in polar solvents, while SWB governs in less polar solvents. To deeply understand the nature of these two excited states, fluorescence decay of both the SWB and LWB bands were measured (Fig. S13, ESI†). It turned out that both exhibit monoexponential decays in MeOH with SWB being 3.99 ns while LWB being 6.72 ns. The prolonged lifetime of LWB indicates that LWB may behave more like a CT process, while the SWB may have a highly localized excitation (LE) characteristic. Furthermore, we also confirmed the excited-state dipole moments separately for SWB and LWB bands using the Lippert–Mataga (LM) equation.18 It shows that the transition state dipole moments of LWB (Δμ = 9.6 D) is larger than that of SWB (Δμ = 2.1 D), consistent with above CT, LE analysis (Fig. S19, ESI†). Notably, for push–pull fluorophores with CT character, low QYs is quite common in increasingly polar solvents.19 However, the effect is absent for PerNN-MeCl and PerNN-MeI with both emissions from the two excited states are virtually high in various solvents (see Table S4 and S5 for details in ESI†).
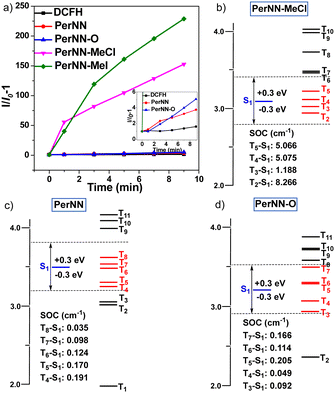
D–A structures, large conjugated systems and heteroatoms usually enhance the ISC rate constant (kISC), speeding up the generation of the reactive oxygen species (ROS).20 Given that molecules in this work can be batch synthesized, we investigated their ROS generation properties. 2′,7′-dichlorodihydrofluorescein (DCFH) was selected as the ROS indicator (DCFH is transformed to 2′,7′-dichlorofluorescin (DCF) with green fluorescence after being activated by ROS). As depicted in Fig. 4a and Fig. S2 (ESI†), the fluorescence emission of DCFH alone varied negligibly under continuous exposure to white light. However, the fluorescence intensity increased swiftly with PerNN-MeI or PerNN-MeCl, indicating that these luminogens can efficiently generate ROS. Remarkably, after white light irradiation for 9 min with PerNN-MeI, the PL intensity of DCFH showed about 228-fold enhancement, exhibiting obviously superior ROS generation efficiency than PerNN and PerNN-O (3-fold and 5-fold, respectively). PerNN-MeCl can also efficiently produce ROS (150-fold within 9 minutes), but slightly lower than PerNN-MeI, suggesting that the heavy iodide element can further enhance kISC. To gain an in-depth insight into why PerNN-MeI or PerNN-MeCl show boosted ROS efficiency, time-dependent density functional theory (TD-DFT) was conducted.21 Previous reports suggest that effective ISC channels between S1 and Tn could be achieved within a maximal single-triplet energy gap with |ΔEST| ≤ 0.3 eV.22 As shown in Fig. 4b–d, PerNN, PerNN-O and PerNN-MeCl, possess comparable number of main ISC transition channels (namely, five, five, and four, respectively). However, the related SOC constants vary a lot. For example, the maximal SOC constant of PerNN and PerNN-O were only 0.191 cm−1 and 0.205 cm−1, respectively. However, this value for PerNN-MeCl was up to 8.266 cm−1 and significantly larger. These results indicated that the larger SOC constant was the reason that PerNN-MeCl had higher ROS generation efficiency. Considering the high water solubility of both PerNN-MeCl and PerNN-MeI (Fig. S9, ESI†), they will be potentially applied in photodynamic therapy.23
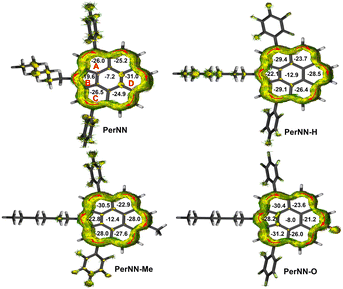
Zigzag-edged PAHs tend to display unique global aromaticity.7 Thus all targets’ aromaticity and induced current were investigated. As suggested in Fig. 5, all the four compounds demonstrate obvious global clockwise (diatropic) ring current delocalized along the anisotropy of the induced current density (AICD) plots, indicating that zigzag edges can indeed induce global ring current. NICS (−1)zz values of all rings are negative, in accordance with ACID results. Overall, ring D is the most aromatic for PerNN, but upon pyridinium formation by methylation or oxidation, its aromaticity is weakened, along with ring A, B and C's aromaticity being enhanced.
Conclusions
To conclude, we successfully doped pyrrolic N and pyridinic N atoms at perylene's opposite edges. The resultant molecule PerNN possesses distinct electrostatic perimeters. As a result, PerNN demonstrates blue emission with CIE of (0.15, 0.05), FWHM of 30 nm (Fig. S5 and Table S2, ESI†), small Stokes shift of 4 nm, and most importantly, column stacking mode attributing to the favored electrostatic interactions. Later, PerNN was facilely derived with moderate to quantitative yield, generating the acidified compound PerNN-H, methylated molecules PerNN-MeCl and PerNN-MeI, and even the oxidized zwitterion PerNN-O. The photophysical, electronic and aromaticity properties of the derivatives were detailly characterized. Taking advantage of the zigzag edges and peripheral heteroatom doping, all the compounds revealed global diamagnetic ring current along the rim. Moreover, both methylated PerNN-MeCl and PerNN-MeI demonstrate anti-Kasha solvent-responsive dual emission ascribed to fluorescence from the S1 CT state and upper excited state of LE S2. Thorough experiments and DFT calculations were performed to clarify this unique phenomenon, which will find wide applications in the sensing filed. Last but not least, PerNN-MeCl and PerNN-MeI also showed highly efficient ROS generation. Overall, the strategy of oppositely disposing donor and acceptor within a plane sheds new light on designing push–pull systems with various interesting properties.Conflicts of interest
There are no conflicts to declare.Acknowledgements
The authors gratefully acknowledge the support of this work by the National Natural Science Foundation of China (Grant 22109111, 22261041), Education Department of Inner Mongolia Autonomous Region (Grant NJYT22098), the Natural Science Foundation of Zhejiang Province (Grant LQ22B030006), Key Laboratory of Organosilicon Material Technology, Zhejiang Province.References
- (a) X. Wan, C. Li, M. Zhang and Y. Chen, Acceptor-donor-acceptor type molecules for high performance organic photovoltaics-chemistry and mechanism, Chem. Soc. Rev., 2020, 49, 2828 RSC; (b) Y. Wang, Y. Huang, T. Huang, J. Zhang, T. Luo, Y. Ni, B. Li, S. Xie and Z. Zeng, Perylene-Based Linear Nonalternant Nanoribbons with Bright Emission and Ambipolar Redox Behavior, Angew. Chem., Int. Ed., 2022, 61, e202200855 CrossRef CAS PubMed.
- Y. Liu, C. Li, Z. Ren, S. Yan and M. R. Bryce, All-organic thermally activated delayed fluorescence materials for organic light-emitting diodes, Nat. Rev. Mater., 2018, 3, 1 CrossRef.
- K. Matsumoto, Y. Tachi and M. Kozaki, A fused polycyclic compound containing phenothiazine and diazapyrene skeletons with weak D–A interactions, Chem. Commun., 2022, 58, 8572 RSC.
- (a) M. Stepien, E. Gonka, M. Żyła and N. Sprutta, Heterocyclic nanographenes and other polycyclic heteroaromatic compounds: synthetic routes, properties, and applications, Chem. Rev., 2017, 117, 3479 CrossRef CAS PubMed; (b) Z. Li, Y. Tang, J. Guo, J. Zhang, M. Deng, W. Xiao, F. Li, Y. Yao, S. Xie, K. Yang and Z. Zeng, Stair-like narrow N-doped nanographene with unusual diradical character at the topological interface, J. Am. Chem. Soc., 2023, 9, 1281 CAS; (c) X. Wang, X. Yao, A. Narita and K. Müllen, Heteroatom-Doped Nanographenes with Structural Precision, Acc. Chem. Res., 2019, 52, 2491 CrossRef CAS PubMed; (d) Z. Li, Y. Bu, S. Xie, Y. Ni, K. Yang and Z. Zeng, Pyridinic Nitrogen Doping: A Versatile Approach for Precise Structure−Property Manipulations of Nanographenes, Chem. Mater., 2024, 36, 3058 CrossRef CAS.
- (a) G. Liu, L. Gao, Y. Han, Y. Xiao, B. Du, J. Gong, J. Hu, F. Zhang, H. Meng, X. Li, X. Shi, Z. Sun, J. Wang, G. Dai, C. Chi and Q. Wang, Symmetry-Broken Intermolecular Charge Separation of Cationic Radicals, Angew. Chem., Int. Ed., 2023, 62, e202301348 CrossRef CAS PubMed; (b) H. Yokoi, S. Hiroto and H. Shinokubo, Reversible σ-bond formation in bowl-shaped π-radical cations: the effects of curved and planar structures, J. Am. Chem. Soc., 2018, 140, 4649 CrossRef CAS PubMed; (c) X. Zhang, M. R. Mackinnon, G. J. Bodwell and S. Ito, Synthesis of a π-Extended Azacorannulenophane Enabled by Strain-Induced 1, 3-Dipolar Cycloaddition, Angew. Chem., Int. Ed., 2022, 61, e202116585 CrossRef CAS PubMed; (d) Y. Morimoto, Y. H. Koo, K. Otsubo, H. Kitakado, S. Seki, A. Osuka and T. Tanaka, Dibenzodiazapyracylenes: Doubly N-Doped Cyclopenta-Fused Polycyclic Molecules That Exhibit High Carrier Mobility, Angew. Chem., Int. Ed., 2022, 134, e202200341 CrossRef.
- (a) E. Jin, Q. Yang, C. W. Ju, Q. Chen, K. Landfester, M. Bonn, K. Müllen, X. Liu and A. Narita, A highly luminescent nitrogen-doped nanographene as an acid-and metal-sensitive fluorophore for optical imaging, J. Am. Chem. Soc., 2021, 143, 10403 CrossRef CAS PubMed; (b) L. Zhou, B. Wu, Y. Chen, J. Gong, J. Wang, G. Dai, C. Chi and Q. Wang, Synthesis and Properties of Aza-Ovalene with Six Zigzag Edges, Org. Lett., 2021, 23, 8640z CrossRef PubMed; (c) Y. Zhang, Z. Zhang and L. Ji, Diagonal and vertical B ← N Lewis pair functionalized perylenes, Org. Lett., 2023, 25, 5273 CrossRef CAS PubMed.
- (a) S. Song, J. Su, M. Telychko, J. Li, G. Li, Y. Li, C. Su, J. Wu and J. Lu, On-surface synthesis of graphene nanostructures with π-magnetism, Chem. Soc. Rev., 2021, 50, 3238 RSC; (b) Q. Wang, T. Y. Gopalakrishna, H. Phan, T. S. Herng, S. Dong, J. Ding and C. Chi, Cyclopenta Ring Fused Bisanthene and Its Charged Species with Open-Shell Singlet Diradical Character and Global Aromaticity/Anti-Aromaticity, Angew. Chem., Int. Ed., 2017, 56, 11415 CrossRef CAS PubMed.
- (a) T. Otani, A. Tsuyuki, T. Iwachi, S. Someya, K. Tateno, H. Kawai, T. Saito, K. Kanyiva and T. Shibata, Facile two-step synthesis of 1, 10-phenanthroline-derived polyaza [7] helicenes with high fluorescence and CPL efficiency, Angew. Chem., Int. Ed., 2017, 56, 3906 CrossRef CAS PubMed; (b) K. Nakamura, K. Ochiai, A. Yubuta, D. He, D. Miyajima and S. Ito, Pyridine-Fused Azacorannulene: Fine-Tuning of the Structure and Properties of Nitrogen-Embedded Buckybowls, Precis. Chem., 2023, 1, 29 CrossRef CAS; (c) J. Y. Lee, J. Y. Shim, H. K. Kim, D. Ko, M. H. Baik and E. J. Yoo, 3, 5-Diarylimidazo [1, 2-a] pyridines as color-tunable fluorophores, J. Org. Chem., 2017, 82, 4352 CrossRef CAS PubMed; (d) G. Hazra, S. Kundu, R. Dandela and B. Thirupathi, Cascade Nucleophilic Addition–Cyclic Michael Addition of Arynes and Indoles Bearing Michael Acceptor: A Strategy to Construct Pyrroloacridines and Study of Their Physicochemical Properties, J. Org. Chem., 2023, 88, 8493 CrossRef CAS PubMed.
- Z. Liu, X. Wang, T. Lu, J. Wang, X. Yan, Y. Wu and J. Xu, Molecular assembly with figure-of-eight nanohoop as a strategy for collection and stabilization of cyclo [18] carbon, Phys. Chem. Chem. Phys., 2023, 25, 16707 RSC.
- R. Puttreddy, J. M. Rautiainen, T. Mäkelä and K. Rissanen, Strong N-X…O-N Halogen Bonds: A Comprehensive Study on N-Halosaccharin Pyridine N-Oxide Complexes, Angew. Chem., Int. Ed., 2019, 58, 18610 CrossRef CAS PubMed.
- C. R. Hoopes, F. J. Garcia, A. M. Sarkar, N. J. Kuehl, D. T. Barkan, N. L. Collins, G. E. Meister, T. R. Bramhall, C. H. Hsu, M. D. Jones, M. Schirle and M. T. Taylor, Donor–Acceptor Pyridinium Salts for Photo-Induced Electron-Transfer-Driven Modification of Tryptophan in Peptides, Proteins, and Proteomes Using Visible Light, J. Am. Chem. Soc., 2022, 144, 6227 CrossRef CAS PubMed.
- W. Zhang, G. Liu, J. Cao, Y. Chen, L. Gao, G. Liu, G. Dai and Q. Wang, Synthesis and Properties of BN-embedded N-Perylene, Chem. – Asian J., 2022, 17, e202200340 CrossRef CAS PubMed.
- CCDC 2300873 containss the supplementary crystallographic data for this paper. This data can be obtained free of charge from The Cambridge Crystallographic Data Centre.
- T. Lu and F. Chen, Multiwfn: A multifunctional wavefunction analyzer, J. Comput. Chem., 2012, 33, 580 CrossRef CAS PubMed.
- F. Li, Z. Zheng, S. Xia and L. Yu, Synthesis, co-crystal structure, and DFT calculations of a multicomponent co-crystal constructed from 1H-benzotriazole and tetrafluoroterephthalic acid, J. Mol. Struct., 2020, 1219, 128480 CrossRef CAS.
- W. Humphrey, A. Dalke and K. Schulten, VMD: visual molecular dynamics, J. Mol. Graphics, 1996, 14, 33 CrossRef CAS PubMed.
- M. Kasha, Characterization of electronic transitions in complex molecules, Discuss. Faraday Soc., 1950, 9, 14 RSC.
- A. R. Brotherton, A. Shibu, J. C. Meadows, N. A. Sayresmith, C. E. Brown, A. M. Ledezma, T. A. Schmedake and M. G. Walter, Leveraging Coupled Solvatofluorochromism and Fluorescence Quenching in Nitrophenyl-Containing Thiazolothiazoles for Efficient Organic Vapor Sensing, Adv. Sci., 2023, 10, 2205729 CrossRef CAS PubMed.
- X. Tian, Y. Xiao, S. Wang, G. Liu, W. Zhang, L. Zhou, J. Gong, X. Zhang, X. Li, H. Meng, J. Wang, G. Dai and Q. Wang, Bowl-Shaped Bispyrrole-Fused Perylene-diimide and Its Anions, Org. Lett., 2023, 25, 1605 CrossRef CAS PubMed.
- B. Lu, Y. Huang, Z. Zhang, H. Quan and Y. Yao, Organic conjugated small molecules with donor–acceptor structures: design and application in the phototherapy of tumors, Mater. Chem. Front., 2022, 6, 2968 RSC.
- (a) X. Gao, S. Bai, D. Fazzi, T. Niehaus, M. Barbatti and W. Thiel, Evaluation of Spin Orbit Couplings with Linear-Response Time-Dependent Density Functional Methods, J. Chem. Theory Comput., 2017, 13, 515 CrossRef CAS PubMed; (b) S. G. Chiodo and M. Leopoldini, MolSOC: A Spin–Orbit Coupling Code, Comput. Phys. Commun., 2014, 185, 676 CrossRef CAS.
- J. Yang, Y. Zhang, X. Wu, W. Dai, D. Chen, J. Shi, B. Tong, Q. Peng, H. Xie, Z. Cai, Y. Dong and X. Zhang, Rational design of pyrrole derivatives with aggregation-induced phosphorescence characteristics for time-resolved and two-photon luminescence imaging, Nat. Commun., 2021, 12, 4883 CrossRef CAS PubMed.
- L. Liu, C. Li, J. Gong, Y. Zhang, W. Ji, L. Feng, G. Jiang, J. Wang and B. Z. Tang, A Highly Water-Soluble Aggregation-Induced Emission Luminogen with Anion-π+ Interactions for Targeted NIR Imaging of Cancer Cells and Type I Photodynamic Therapy, Angew. Chem., Int. Ed., 2023, 26, e202307776 Search PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. CCDC 2300873. For ESI and crystallographic data in CIF or other electronic format see DOI: https://doi.org/10.1039/d4nj01177e |
| This journal is © The Royal Society of Chemistry and the Centre National de la Recherche Scientifique 2024 |