 Open Access Article
Open Access ArticleControlling the fate of two triplet states: solid-state annihilator design for photon upconversion
Jussi
Isokuortti
*a and
Lea
Nienhaus
 *abcde
*abcde
aDepartment of Chemistry, Rice University, Houston, Texas 77005, USA. E-mail: ji30@rice.edu; ln27@rice.edu
bDepartment of Physics and Astronomy, Rice University, Houston, Texas 77005, USA
cDepartment of Materials Science and NanoEngineering, Rice University, Houston, Texas 77005, USA
dDepartment of Chemical and Biomolecular Engineering, Rice University, Houston, Texas 77005, USA
eRice Advanced Materials Institute, Rice University, Houston, Texas 77005, USA
First published on 24th February 2026
Abstract
Photon upconversion, the process of converting low-energy light into higher-energy photons, offers transformative opportunities for energy conversion and optoelectronics. In this Perspective, we examine the molecular design principles of solid-state annihilators, emphasizing a unified excitonic framework that links upconversion with related technologies such as organic light-emitting diodes and singlet fission. We discuss key challenges, including controlling triplet exciton diffusion, maximizing singlet formation efficiency, and suppressing loss channels such as excimer and trap-state formation. Strategies that leverage precise intermolecular packing and tailored electronic coupling are highlighted as critical levers for dictating excited-state dynamics and optimizing photophysical outcomes. Looking forward, we propose that future breakthroughs will surpass the “solution-first approach” and hinge on integrating data-driven machine learning approaches with phonon engineering to rationally design materials with enhanced light emission and energy conversion efficiency. This framework provides guiding principles for the next generation of high-performance solid-state upconversion systems.
1. Introduction
Photon upconversion based on triplet–triplet annihilation (TTA-UC) is deeply interconnected with singlet fission (SF), organic light-emitting diodes (OLEDs), room-temperature phosphorescence (RTP), and related areas of organic electronics that exploit spin-dependent excited-state dynamics. Rather than being isolated phenomena, these fields share common physical principles involving triplet generation, triplet pairing, and the control of singlet and triplet energy manifolds through molecular packing and electronic coupling. This Perspective positions solid-state TTA-UC annihilator design squarely within the broader excitonic landscape.TTA and SF represent complementary pathways that interconvert singlet and triplet excitons through correlated triplet-pair states, placing annihilator chromophores at the same conceptual foundation as SF materials and OLED emitters. In OLEDs, control over triplet harvesting, singlet formation, and excimer or exciplex emission is central to device efficiency, while in RTP materials, suppression or modulation of nonradiative triplet decay governs emission lifetimes and brightness. These same design challenges reappear in solid-state TTA-UC, where interchromophore coupling, energetic alignment, and aggregate structure determine whether triplets undergo productive annihilation, form emissive singlets, stabilize excimers, or funnel into undesired loss channels.
By explicitly unifying TTA-UC with singlet fission, OLED photophysics, RTP, and broader organic electronic materials, this Perspective emphasizes that advances in annihilator design are not incremental refinements of upconversion alone but contribute directly to a shared framework for manipulating spin statistics and excited-state fate in organic solids.
The discussion is intentionally focused on molecular and aggregate-level design principles of annihilators and excludes polymeric films, extended frameworks, and device fabrication strategies, for these topics we refer the interested reader to existing literature.1–5 Emphasis is placed on transferable design rules that balance triplet generation, diffusion, and annihilation with selective singlet stabilization and emission, offering guidance relevant across upconversion, light emission, and energy conversion technologies.
2. Overview of the mechanism of TTA-UC and the critical loss channels in solid state
TTA occurs when two (spin-)triplet excited states come into contact and annihilate to generate a higher excited state and a ground state.6,7 UC arises when photoexcitation produces triplet states at an energy lower than that of the higher excited state formed through TTA, as shown in Fig. 1. The desired higher excited state is often a singlet state that can relax radiatively, however the upconverted energy can potentially be directly harvested from other species, such as the triplet pair state.8–11 For TTA to result in a singlet excited state, the annihilator chromophore needs to generally satisfy the energetic requirement of:| E(Sn) ≲ 2 × E(T1), | (1) |
TTA and SF are considered the reverse reactions of each other and can be presented under a unified kinetic model:14,15
| T1 + T1 ⇄ [T1⋯T1] ⇄ 1(TT) ⇄ S1S0, | (2) |
Excited triplet states in TTA-UC are typically produced by separate chromophores called (photo)sensitizers due to the spin-forbidden nature of the direct transition to the spin–triplet state (Fig. 1). Historically, sensitizers have been molecular chromophores that undergo efficient intersystem crossing (ISC) to produce excited triplet states from singlet excited states that can be directly excited.17 However, the use of molecular photosensitizers in solid-state TTA-UC is often hampered by aggregation-induced losses that compete with triplet energy transfer (TET) to the annihilator.18–21 More recently, new classes of photosensitizers have been developed, such as semiconductor quantum dots that possess spin-mixed excited states, alleviating large exchange energy losses.22
Intriguingly for solid-state applications, inspiration has also been drawn from solar cell technologies through the development of layered TTA-UC devices, where the triplet excited states are generated by charge recombination. The triplet generation can occur either in the photosensitizer bulk, for example at an organic donor–acceptor heterojunction, or at the interface between the annihilator and the photosensitizer, such as bulk halide perovskites,23 transition metal dichalcogenides24 or organic semiconductors,25–27 resulting in direct triplet generation in the annihilator layer. The emergence of these layered TTA-UC devices has greatly boosted solid-state upconversion efficiencies.28 However, triplet generation by interfacial charge recombination requires band alignment between the valence band of the sensitizer and the highest occupied molecular orbital (HOMO) of the annihilator,23,29–32 which imposes stricter constraints on the choice of annihilators beyond the rudimentary energetic requirement discussed above (eqn (1)).
As hinted above with eqn (1), TTA-UC can result in a higher-lying excited singlet state. An archetypal example of such an annihilator is Zn-tetraphenylporphyrin, where low energy excitation of the S1 results in efficient ISC to T1 and subsequent TTA to S2.33–35 As the sensitizer and the annihilator are the same chromophore, this process is called homomolecular TTA (HTTA). In addition to being interesting systems from a fundamental photophysical and photochemical perspective, realization of efficient HTTA-UC systems would streamline the design of solid-state TTA-UC. As singlet fission can also occur from higher singlet excited states,36,37 such materials can offer insight into developing more efficient HTTA materials along with other chromophores that break Kasha's rule.38,39 In the solid state, altered relaxation pathways and energy landscapes can make radiative decay from higher excited states competitive; consequently, restricted molecular motion, reduced vibrational energy dissipation, and strong intermolecular interactions slow internal conversion, increasing the likelihood of Kasha's rule breakdown.
The efficiency of TTA-UC can be represented with three figures of merit: (1) the apparent anti-Stokes shift,40 (2) the quantum yield of upconversion (ΦUC)41 and (3) the intensity threshold (Ith)42,43 at which TTA becomes the primary triplet relaxation pathway. The apparent anti-Stokes shift is the energy difference between the excitation wavelength and upconverted emission in a TTA-UC system. ΦUC is often decomposed into the individual efficiencies of the processes that participate in the TTA-UC cascade (Fig. 1):
| ΦUC = ½ΦISCΦTETΦTTAfΦf, | (3) |
As TTA is a bimolecular process that occurs between metastable states, ΦUC is a function of the triplet population and thus the excitation intensity. The maximum ΦUC is reached at excitation intensities where the triplet decay is dominated by TTA over the unimolecular or spontaneous triplet decay. The Ith is the excitation intensity at which the rate of TTA and the rate of spontaneous triplet decay are equal and is typically defined as44
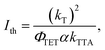 | (4) |
Typically, the performance goals for UC systems are to maximize the apparent anti-Stokes shift and ΦUC, while minimizing the Ith. To illustrate some of the recent advances in the field, we have highlighted TTA-UC systems that are among the state of the art in Table 1.
However, we would like to offer a counterpoint that as the field of TTA-UC matures and becomes more application-oriented, these goals may change.5 For example in photocatalytic applications, maximizing the brightness of the UC system or the reaction rate at the achievable excitation intensity may be more important than as high and low as possible ΦUC and Ith, respectively.8,48 Accordingly, the focus of this Perspective is to offer annihilator design guidelines for tuning the properties of annihilators to best suit the application at hand.
3. Maximizing singlet and fluorescence yields
As eqn (3) shows, achieving high ΦUC requires annihilators with high probability of singlet formation upon TTA (f) and Φf. In this section we will investigate the various annihilator attributes to maximize the production of upconverted photons.3.1 How to (statistically) make two triplets into one singlet
Eqn (1) shows the most rudimentary energetic conditions for TTA-UC to occur, i.e., to yield an upconverted singlet state. 9,10-Bis(phenylethynyl)anthracene (BPEA, Fig. 2) is a common annihilator as well as SF material since it fulfils the energy requirements of both SF and TTA: 2 × E(T1) ≅ E(S1).49,50 Gray et al. have shown that the free rotation of the phenyl groups and consequent change in π-conjugation alters the energies of the excited triplet and singlet state.51 Thus, only certain conformations of BPEA have sufficiently high triplet energy to annihilate into a singlet excited state and results in poor ΦUC with BPEA in solution phase. In a similar vein, the groups of Campos and Congreve have engineered diketopyrrolopyrrole (DPP, Fig. 2), tetracene, anthracene and perylene annihilators by relatively decreasing the singlet energy compared to the triplet energy and therefore increasing the energetic driving force of singlet formation upon TTA and ΦUC.52,53 While these results were obtained in solution-based TTA-UC, this approach was shown to work in solid-state TTA-UC by Radiunas et al. in rubrene-based annihilators.54Alternatively, the relative triplet and singlet can be altered in the solid state without modifying the chemical structure of the chromophore. Ye et al.55 boosted the TTA efficiency of a DPP annihilator by coupling its singlet state to a polaritonic state, thus providing larger energy overhead for accessing an excited radiative state upon TTA. Aggregation may also lower the singlet excited state energy, ultimately facilitating TTA-UC.56 For example, Nienhaus and co-workers have demonstrated that TTA is boosted in naphtho[2,3-a]pyrene (NaPy, Fig. 2) aggregates due to the redshift of the excited singlet state in strongly coupled dimer states.57,58 A similar strategy has been employed to turn on thermally activated delayed fluorescence by reducing the S1–T1 energy gap.59 As such, the selective tuning of the singlet and triplet energies either through synthetic methods, light–matter interactions or intermolecular coupling is a viable strategy to tip the scale towards singlet generation in TTA-UC.
In the early days of the TTA-UC renaissance in the 2000s, there were conflicting conceptions whether two annihilating triplets can produce a singlet state encounter complex with either a 1/9 (∼11%) or ∼40% probability.17,60 Both of these limits were first challenged by Schmidt et al. by observing higher singlet yields in TTA in rubrene.61–63 Followed by their initial discoveries, several pathways of boosting f, and consequently ΦUC, have been identified. Here, we break them into three individual mechanisms: (1) relative energetics of the higher triplet and singlet excited states, (2) inter-chromophore orientation and distance and (3) changing the singlet character of the triplet pair state by tuning the triplet–triplet coupling.
Above, we discussed how altering the singlet and triplet energies can lead to higher singlet yields. However, states of higher multiplicity than singlets, namely higher excited triplet states (Tn, n ≥ 2), can also be generated upon TTA. Accessing these higher triplets can be considered a loss channel for TTA as the Tn undergoes rapid internal conversion to T1.64,65 Similarly, close energetic alignment of Tn and S1 is considered detrimental for SF as a single triplet state may be generated via ISC instead of two triplets via SF in such chromophores.66,67 Considerable efforts, mostly using computational methods, have been made to discover annihilators,68–73 TTA-OLED74 and SF materials75–83 where Tn is not energetically accessible, i.e., 2 × E(T1) < E(Tn). However, as most of these studies are performed using isolated molecules, their predictive capability has been limited in solid-state systems, where the complex chromophore–chromophore interactions complicate the model.84 It will be interesting to see how quickly new data-driven methods, such as machine learning,85 can start producing translatable predictions for solid-state TTA and SF materials.
In contrast to the assumption stated above that energetically accessible Tn states are outright adverse for high f and consequently ΦUC, Xu et al.86 discovered in 2019 that closely matched S1 and T2 energy levels in an anthracene-based emitter increased the efficiency of TTA-OLEDs and demonstrated that T2 → S1 ISC occurred and was the likely cause for the boost. Since then, this high-level ‘reverse’ intersystem crossing (HL-RISC, Fig. 3) has been identified as an active channel in increasing f rubrene87–89 and in a structurally diverse set of TTA-OLED materials.90–96 Since the discovery of HL-RISC as an active channel in TTA is recent, we expect that its mechanism and existence in other annihilators, such as TIPS-naphthalene,97 that surpass the ‘conventional’ limit of f = 0.4 will be investigated. For example, the high f > 0.4 in 9,10-diphenylanthracene (DPA) has been attributed to T2 → S1 HL-RISC by Miyashita et al.,98 whereas Shao et al.99 concluded it results from direct ISC from 3(TT) to S1. This underlines the need for more in-depth mechanistic studies for different annihilators to elucidate the mechanism.
Efficient HL-RISC naturally requires close energetic alignment between Tn and S1 and large spin–orbit coupling (SOC) to compete with the often rapid Tn → T1 internal conversion. Kim et al.100 demonstrated in silico that introducing electron-donating and withdrawing functional groups, heavy atoms and geometric distortions enhances T2–S1 SOC and presumably HL-RISC. They also postulate that since the T2 → T1 internal conversion requires significant geometric reorganization due to the large T2–T1 energy gap, in solid-state T2 → S1 HL-RISC may offer significant competition to T2 → T1 internal conversion and contribute to achieving high f. However, we would like to cautiously note that the requirements for efficient HL-RISC, i.e., a small energy gap between Tn and S1 and significant SOC may be at odds with the other desired attributes for annihilators, such as high Φf and small kT. Thus, systematic experimental studies are needed to balance the outcome to create new efficient annihilators.
We now turn our investigation from energetics to the spin-character triplet pair state and its influence on f. The spin interactions between two triplets, A and B, can be expressed with a Hamiltonian that, without spin–orbit coupling, can be approximated as88,101
 | (5) |
From a purely spin-character perspective, the strongly coupled triplet pair is not expected to have an angular dependence on f. However, the exchange interaction and thus the triplet–triplet coupling depends on the orbital overlap that is a function of the overall chromophore distance and alignment (Fig. 3),110,111 which will influence the branching ratios between singlet formation and other competing processes occurring in the triplet pair, such as internal conversion to an individual triplet (3(TT) → Tn)88 or dissociation from the strongly to the weakly coupled triplet pair (1(TT) → [T1⋯T1]),107,112,113 which can have a significant effect on f.64
Going beyond the exchange interaction in the triplet pair, Naimovičius et al.114 have suggested that silylethynyl (often referred to as ‘TIPS’) functionalization of perylene (Fig. 2) more than doubled its singlet yield from 18% to almost 40% by introducing significant charge transfer character to the annihilator S0S1 dimer. Similarly, charge transfer character in the dimer has been shown to increase the coupling between S0S1 and 1(TT) in singlet fission.115 The involvement of charge transfer states in boosting f could be probed by, for example, changing solvent polarity (in solution-based TTA-UC),116–118 or by applying an external electric field (in solid-state TTA-UC).119,120 Another interesting method for controlling the dielectric properties of solids is co-crystallization,121,122 although disentangling changes in annihilator coupling from purely dielectric stabilization of the charge transfer state may be challenging.
In summary, the seminal work of Bossanyi et al.88 using rubrene indicates that f can be dominated by the spin character of the weakly coupled triplet pair and HL-RISC, however we eagerly wait for equally thorough studies on other solid-state annihilators beyond rubrene to expand and generalize these rules. DPPs are synthetically straightforward to functionalize and are both SF123 and TTA47 active, thus offering a highly tuneable platform for systematic studies. Some covalently linked annihilator dimers have shown improved f over their monomers and thus offer a precisely tuneable platform for studying the exchange interaction and its role in determining f.124,125
3.2 The fate of the singlet excited state
Having outlined, in a somewhat shaky fashion that perhaps reflects the current level of certainty in the field, the design factors that contribute to f, we turn our focus to the newly generated excited singlet state. The two annihilators, one in a singlet excited state and the other in singlet ground state, are inherently coupled, and the nature and extent of this coupling determines the fate of the singlet excited state. As we mentioned in Section 2, the goal for TTA-UC systems is often to yield a Sn that would emit as high-energy photon with as high probability as possible.Above we discussed how aggregation can be used to modify the singlet energy of the annihilator in respect to the triplet. In these aggregates, S1 was red-shifted in respect to the monomer, which is often attributed to J-type aggregation (Fig. 4a) that also tends to enhance the radiative decay rate and thus Φf.56,126 On the other hand, H-type aggregation (Fig. 4a) is often associated with blue-shifted emission but at a cost of quenched emission compared to the monomer.127 Based on this rather simple comparison, it would be most beneficial for TTA-UC to realize aggregates that bear the benefits of both types of aggregates, i.e., enhanced and blue-shifted emission. Considering that the chromophore couplings that lead to the aggregate properties are an ensemble of coulombic and charge transfer interactions that can have opposite signs, systems that exhibit both H- and J-type (called HH, HJ, JH and JJ-aggregates128 or more recently referred to I aggregates58,129,130) properties can be realized. As an example, by carefully balancing the exciton and charge transfer (CT) coupling, aggregates that exhibit both high carrier mobilities (often associated with H-type aggregates due to co-facial stacking motifs) and high emissivity can be designed (Fig. 4).131 Disorder may also relax these rules to some extent.132 These factors naturally carry great importance to the design of annihilators, not only in terms of their emissivity and singlet energies, but also enhancing triplet diffusion, which we will discuss later.
The mention of CT coupling naturally brings about another possible decay pathway for Sn, which is the formation of an excited state dimer (excimer, Fig. 4b) where the exciton is shared between two chromophores through coulombic coupling and stabilized through charge transfer interaction. Historically, excimer and TTA research have advanced together and, in many systems, excimers have been identified as a direct and thus competing product of TTA instead of a monomeric Sn.133–135 Excimers have also been identified as either an intermediate or a product state in SF, underlying the importance of understanding the mechanism and control of excimer formation in TTA-UC. Further, excimers can serve as (defect) trap states to which excitons can migrate rapidly over long distances even if the dominant crystal structure of the annihilator is not conducive for excimer formation.
Excimer formation is often associated with H-type aggregation (Fig. 4) as strong CT coupling typically requires π–π overlap.56 However, in some closely spaced π-stacks, excimer formation is supressed, which can result from, for example, the balanced coulombic and CT coupling136 and intermolecular vibrational modes.137 Thus, transient methods, such as ultrafast Raman,138,139 multidimensional electronic spectroscopy140 and ultrafast electron diffraction,141 that provide time-resolved vibrational or structural dynamics can be valuable tools for untangling the conformational changes that contribute to excimer formation and thus provide insight into molecular/aggregate design. This vibronic coupling highlights the fact that excimers depend on intermolecular reorganization and can provide a prominent handle for tuning the fate of the singlet exciton.142,143
As mentioned, excimer formation often occurs in closely spaced cofacial dimers. Thus, adding steric bulk has often been the method for preserving monomeric emission.144,145 Naturally, this may come at the cost of for example reduced f or ΦTTA but balancing these effects is possible as TTA can occur at larger intermolecular distances than excimer formation.146,147
We have used in the discussion above the ‘classical’ definitions, i.e. H and J-aggregation as they often satisfactorily explain the aggregate properties. However, there are naturally other aggregation or stacking modes, such as magic angle and crossing stacking, that do not follow these models, and we refer the interested reader to these recent reviews.148,149
There is a wealth of physical insight into controlling the fate of the singlet excited states in molecular aggregates and crystals. This is evident for example in covalently linked dimers where the interchromophore couplings can be tuned to a remarkable degree.150 However, translating these results into solid-state systems can be hit-or-miss. The unfortunate reality is that ab initio design of organic crystals with predetermined optical properties is very challenging from first principles. As with energetic prediction, we are hopeful that machine learning methods that utilize large crystal databases as training materials will soon yield tangible results.151,152
Meanwhile, reverse engineering viable annihilators from known efficient solid-state emitters may be a feasible strategy. Chromophores that exhibit aggregation-induced emission (AIE) show weak or no emission in solution, whereas in the aggregate states they become emissive.153 This enhancement of radiative decay is often attributed to the inactivation of vibrational or rotational modes that can lead to large conformational changes and provide access to non-radiative relaxation pathways such as minimum energy crossing points between excited and ground state manifolds.154 As TTA-UC research often follows the “solution first” principle due to the relative ease of solution-based measurements, annihilators that possess for example rotatable bonds have been naturally overlooked. However, there have been a few reports of solid state TTA-UC systems that employ AIE-active annihilators, which serve as encouraging examples of the potential viability of this approach for annihilator design.155–157 To further this point, we would like to point out indigoid dyes (Fig. 5) as an example of potentially overlooked annihilator class. Indigoids are typically associated as photoswitches as they tend to undergo Z-to-E isomerization upon photoexcitation.158 As expected, their Φf tend to be low in solution but increase in crystals or rigid matrix.159–161 Indigo also has a low-lying triplet excited state (E(T1) ≈ 1 eV)162 and thus has been identified as a potential SF material by Michl et al.,66 while isoindigos have been shown to undergo singlet fission.163 Additionally, indigo exhibits high electron and hole mobilities,164 whose importance will be discussed in the next section.
While the physical principles of maximizing the fluorescence yield from an annihilator are clear, optimizing the singlet yield upon TTA still requires more fundamental research. The need for further research becomes even more evident, as the applications of TTA-UC often requires optimizing both. In the light of current evidence, these requirements may be somewhat at odds as, for example, maximal f is achieved in parallel chromophore configurations, while simultaneously this arrangement is conducive of excimer formation. Thus, it may be more desirable for applications of TTA-UC to find molecular systems that offer a suitable balance between high singlet and fluorescence yields.
4. Triplet diffusion
For two triplet states to come into contact in solid-state devices, they will likely need to migrate over some distance from their sensitization sites. Maximizing this diffusion distance carries two-fold significance for TTA-UC efficiency, as both high ΦTTA and low Ith require highly mobile triplet excitons.165–167 The triplet diffusion length can be expressed as168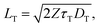 | (6) |
Conventional annihilators based on polyaromatic hydrocarbons such as acenes, perylene and pyrene tend to have long intrinsic τT due to lack of heteroatoms (either those with non-bonding orbitals to invoke El-Sayed type spin–orbit coupling169 or heavy atoms). However, as the library of annihilators is becoming more chemically diverse beyond hydrocarbons, more sophisticated strategies to extend τT are required.
Though the T1 → S0 transition is spin-forbidden, vibrational coupling between different spin manifolds cannot be neglected.170 For example, the isotope effect on triplet lifetimes has been known since the 1960s when it was discovered that deuteration of polyaromatic hydrocarbons, such as naphthalene,171,172 increases their triplet lifetimes by reducing the frequency of the C–H(D) stretching modes. More recently, Danos et al.173 showed that perdeuteration indeed increased τT of perylene and thus improved its TTA-UC efficiency at lower excitation intensities in agreement with eqn (4). Similarly, deuteration has been used to increase the efficiency of thermally activated delayed fluorescence174 and room temperature phosphorescence dyes.175
Despite its proven efficacy, we are not advocating for everyone to start deuterating their annihilators, as other tools to achieve similar results exist at a much lower cost factor. These approaches are sometimes summarized under the term ‘phonon engineering’.176 As vibrational motion cannot be completely suppressed at typical operating conditions of most TTA-UC systems (room temperature or even higher), identifying and isolating modes that contribute to the triplet decay become paramount. Such modes are those that for example enhance the T1–S0 spin–orbit coupling or have large reorganization energies.177,178 Rational design of annihilators where such modes are suppressed will require collaboration between computational and experimental researchers. This requires accurate calculations of the triplet energy surface, which is traditionally challenging and resource intensive, however recent advances in incorporating machine learning into the computational pipeline are encouraging.179–181
Naturally, the triplet exciton decay may be bimolecular, TTA being the desirable pathway. However, as was the case with singlet excitons, triplet states may also succumb to excimer formation.182 Triplet excimers (Fig. 7) have been thoroughly investigated in naphthalenes, however their presence has been shown in other TTA-relevant chromophores such as anthracene, tetracene and pyrene.183–186 Additionally, exciplex (excited state complex) formation between triplet excited DPA and ground state PtOEP has been identified,187 which further highlights the importance of deciphering this decay channel. As triplet states are ‘dark’ excitons, the stabilization in a triplet excimer is presumably of purely CT character. Expectedly, triplet excimer is most stable in naphthalene dimers with face-to-face stacking,188,189 however other stacking motifs can also lead to triplet delocalization.
 | ||
| Fig. 7 Triplet excimer (Tex) formation delocalizes the triplet state and serves as a trap state that may prevent TTA from occurring. | ||
Triplet excimers are challenging to detect due to their inherent dark nature and transient absorption spectra that may be convoluted by monomeric triplets or impurities.183,190–192 Typical method for enhancing phosphorescence, i.e., cooling to cryogenic temperatures, may prevent the intermolecular reorganization that is required for excimer formation and would be accessible at ambient temperature.193 Therefore, we conclude that excimer formation may be a significantly underestimated triplet decay pathway in TTA-UC systems and warrants more extensive investigation in different annihilator classes, especially in the solid state.
Long-lived triplets still need to diffuse to achieve efficient TTA-UC. Rapid diffusion will naturally enhance TET between the sensitizer and the annihilator, at least by maximizing the mixing entropy associated with TET by expanding the number of annihilator sites accessible within the triplet lifetime.194 For interfacial sensitizer–annihilator systems triplet diffusion can act as the rate limiting step for TTA-UC, further highlighting the need for highly mobile triplets.195,196 Factors that influence triplet diffusion are rather well-known, thanks to the abundance of research in the organic electronics and photovoltaics community. As triplet transfer requires orbital overlap,197 or more particularly HOMO–HOMO and LUMO–LUMO overlap, TET coupling is maximized in symmetric face-to-face stacking.198,199 However, as we have discussed, such assembly motifs can induce other undesirable effects such as excimer formation. Therefore, finding annihilator systems where triplet diffusion is rapid without trading off other performance parameters, such as efficient TTA, is paramount.200,201 For example, achieving significant SF/TTA coupling typically requires that the molecules are slipped along both x and y coordinates.202 Naturally, a molecular aggregate or crystal is not static, and thermal effects can induce significant changes in the intermolecular couplings relevant to TTA-UC. Again, using rubrene as a prototypical annihilator, Diaz-Andres et al.203 showed in silico that TET coupling was considerable between stacked rubrene molecules at 0 K but SF/TTA coupling was negligible. Notably, at 298 K TET coupling decreased minimally, whereas SF/TTA coupling increased greatly. Similarly, Volek et al. have demonstrated that SF occurs faster at the edge than in the bulk of rubrene crystal due to disordered packing.204 These results indicate that rational design of solid-state annihilators requires taking such perturbations, whether thermal (vibrational) or packing-related, into account.
Not surprisingly, such holistic phonon engineering efforts have been carried out by the organic electronics and more recently by the singlet fission community to optimize exciton and charge carrier transport. Rather frustratingly, phonons can either make or break transport and dissecting the factors that play a part are still somewhat debated and warrant their own reviews.84,205–211 However, we are happy to provide some examples here that highlight the importance of understanding the dynamic nature of exciton/charge transfer: the seminal work of Schweicher et al.212 coined the term “killer phonon mode” that contributes strongly to the electron–phonon coupling and thus magnifies disorder and effectively hampers charge mobility. On a more positive note, Lynch et al.213 were able to identify ‘spectator’ and ‘driving’ phonon modes of exciton transport following SF in pentacene crystals. These two instances highlight that it is possible, at least in theory, to phonon engineer materials with very mobile triplets.
Finally, we would like to summarize that designing annihilators that exhibit long-lived and mobile triplets requires holistic understanding of both intramolecular and intermolecular vibrational modes that may contribute to triplet decay or transport as well as balancing the packing to facilitate both triplet transport, TTA and emissive singlets as outlined in the previous section. A possible solution, albeit beyond the strict scope of this Perspective, is to include a triplet mediator that facilitates triplet diffusion without interfering with TTA.214 Further, as we will discuss in the next section, rapidly diffusing triplets may rather controversially reduce the TTA-UC yield (in addition to facilitating trapping),215 which is why we especially encourage efforts to find annihilators with long-lived triplets.
5. Decoupling singlet fission
As already highlighted with eqn (2), SF and TTA always represent the two sides of the same coin. In this last section we will investigate different methods to bias that coin to show the obverse heads (TTA) rather than the reverse tails (SF) side.By a first glance, differentiating between these two processes seems rather facile by their different energetic requirements. However, as already discussed above, SF can occur in systems where the triplet pair has significantly higher energy than the singlet. Typical examples of such systems are anthracene and tetracene,216,217 where an energy barrier of more than 200 meV needs to be overcome, yet SF is still fast. These energetic assignments are often based on optical transitions, which reflect more the enthalpy of excitation rather than the total free energy. As such, entropic contributions are often cited as the driving force in such endothermic systems SF, as multiexciton generation, or more precisely the triplet pair dissociation, increases the multiplicity or statistical weight of the excitation.12,13,218 As annihilators are often chosen based on (spectroscopic) energies that indicate exothermic TTA, endothermic SF systems are especially useful models for discerning factors that bias TTA over SF.
The dissociation of 1(TT) to free triplets (Fig. 8) must overcome an energy barrier. As expected, this step has been shown to be thermally activated in endothermic SF.219 Naturally, this energy barrier is sensitive to the interchromophore coupling220–223 and the binding energy of the triplet pair. Besides energetic contributions, intramolecular vibrational modes may promote the dissociation step.224 Thus, tuning these inter and intramolecular handles can hinder SF by, for example, making the binding energy of the triplet pair higher.225,226 More relevantly, SF has been shown to be strongly driven by the triplet separation following dissociation in both exothermic and endothermic SF, i.e., rapid triplet transport increases the yield of free triplets.212,221,227–231
The fact that the dissociation step requires triplet diffusion makes the SF/TTA-UC dynamics both morphology and size-dependent. As an example, BPEA can undergo both SF and TTA shows higher triplet yields in larger particle sizes, and a crystal-structure dependent SF yield.50,232,233 Volek et al. demonstrated that the SF/TTA balance in N,N′-bis(2-phenylethyl)-3,4,9,10-perylenedicarboximide can be tuned by crystal size.234 In terms of morphology, Wieghold et al. have shown that rubrene can be biased towards TTA over SF by rendering it amorphous instead of (poly)crystalline.235 These results highlight the importance of sample and device preparation processes, while we are mainly concerned with molecular design in this Perspective.
Chemically modifying rubrene by inserting sterically bulky groups has been shown to effectively decouple SF.236,237 Baronas et al.236 attributed the suppression of SF to hindered triplet diffusion in the substituted rubrene. Naimovicius et al.47 furthered this strategy beyond the well-worn rubrene scaffold by boosting the ΦUC of DPPs and dipyrrolonaphthyridinediones (DPNPs, Fig. 2) through the insertion of bulky sidegroups. As such, ‘bulking up’ annihilators appears to be a straightforward blueprint for improving solid-state annihilators, where SF is an active and undesired loss channel.
Overall, this leads us to postulate that fast triplet transport may hinder solid state TTA-UC by tilting the balance towards SF. This relaxes the design parameters of annihilators compared to singlet fission materials to some extent, since efficient SF needs to strike a balance between triplet pair formation and triplet transport, two processes that inherently require different interactions from orbital overlap perspective.
In this section we have so far discussed controlling the triplet dissociation as a way of biasing TTA over SF. However, SF and TTA share another intermediate, 1(TT). Although still somewhat debated, the coupling between S1 and 1(TT) is CT mediated (Fig. 9).238 This raises the question whether this coupling can be tuned to steer the triplet pair into forming singlets rather than dissociate into triplets. By lowering the energy of the CT state below S1 and 1(TT), SF can be effectively quenched as a relaxation pathway.239 Similarly, excimer formation has been shown to hinder singlet fission.146 As such, careful balance between excitonic and charge transfer character, as already discussed in Section 3, or fully leaning into excimer formation can aid in distinguishing TTA from SF. This is further supported by the fact that the recombination of non-geminate triplets produces relatively more excimers than geminate triplet pairs, i.e., chromophore assemblies that participate in SF are comparatively less likely to generate excimers.240 This finding is also consistent with the results that emissive states formed in solution-based TTA rather than direct excitation are more predisposed to excimer formation.147,241
6. Conclusions and outlook
In summary, the realization of efficient solid-state photon upconversion depends on the sophisticated control of energetics, spin-character, and molecular packing within the annihilator layer which ultimately dictate the fate of the singlet and triplet state. While the basic energy conservation requirement of E(Sn) ≲ 2 × E(T1)remains a good starting point for determination of viable annihilators, achieving a high singlet formation yield requires a deeper understanding of pathways such as HL-RISC and the role of orbital overlap in determining triplet–triplet coupling. Furthermore, managing triplet diffusion is a critical challenge; although high mobility is necessary to lower the intensity threshold Ith, it can also facilitate loss channels like SF or the formation of “dark” triplet excimers. Looking forward, the development of solid-state annihilators must transition from empirical “solution-first” approaches to predictive, data-driven design. The integration of machine learning and computational pipelines to model complex chromophore–chromophore interactions and crystal properties will be essential for identifying next-generation materials. We anticipate that future research will increasingly leverage phonon engineering to suppress non-radiative decay modes and optimize exciton transport, moving toward materials characterized as solids on excitonic timescales, such as liquid crystals. As TTA-UC moves toward specific applications like photocatalysis, the fundamental aspects of annihilator design remain paramount while more practical device development will likely shift focus from achieving, for example, the lowest possible Ith to maximizing absolute brightness or reaction rates. Moreover, as the underlying science becomes better understood and more mature, device engineering considerations, including light outcoupling strategies, optimized device architectures, and the mitigation of challenges such as scattering and inner filter effects, will play an increasingly critical role. Ultimately, the continued synergy between the TTA-UC, OLED, and singlet fission communities will be the primary driver for mastering the fate of triplet states in the solid state.Author contributions
J. I. – conceptualization, writing – original draft, writing – review & editing. L. N. conceptualization, writing – original draft, writing – review & editing, funding acquisition.Conflicts of interest
There are no conflicts to declare.Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this review.Acknowledgements
We acknowledge funding by the National Science Foundation under Grant No. DMR-2517590, the Camille and Henry Dreyfus Foundation (TC-23-050) and the Alfred P. Sloan Foundation (FG-2024-22474).Notes and references
- X. Chen, X. Zhang and Y. Zhao, Chem. Soc. Rev., 2025, 54, 152–177 RSC.
- H.-J. Feng, M.-Y. Zhang, L.-H. Jiang, L. Huang and D.-W. Pang, Acc. Chem. Res., 2025, 58, 3543–3557 Search PubMed.
- P. Venkatesan, P. Pal, S. S. Ng, J.-Y. Lin and R.-A. Doong, Coord. Chem. Rev., 2025, 523, 216266 Search PubMed.
- L. Wei, C. Yang and W. Wu, Mater. Chem. Front., 2023, 7, 3194–3208 Search PubMed.
- T. Schloemer, P. Narayanan, Q. Zhou, E. Belliveau, M. Seitz and D. N. Congreve, ACS Nano, 2023, 17, 3259–3288 Search PubMed.
- C. A. Parker, C. G. Hatchard and E. J. Bowen, Proc. R. Soc. London, Ser. A, 1962, 269, 574–584 Search PubMed.
- C. A. Parker, Proc. R. Soc. London, Ser. A, 1963, 276, 125–135 Search PubMed.
- C. J. O'Dea, J. Isokuortti, E. E. Comer, S. T. Roberts and Z. A. Page, ACS Cent. Sci., 2024, 10, 272–282 Search PubMed.
- H. Kim, B. Keller, R. Ho-Wu, N. Abeyasinghe, R. J. Vázquez, T. I. Goodson and P. M. Zimmerman, J. Am. Chem. Soc., 2018, 140, 7760–7763 Search PubMed.
- W.-L. Chan, M. Ligges, A. Jailaubekov, L. Kaake, L. Miaja-Avila and X.-Y. Zhu, Science, 2011, 334, 1541–1545 CrossRef CAS PubMed.
- D. G. Bossanyi, M. Matthiesen, S. Wang, J. A. Smith, R. C. Kilbride, J. D. Shipp, D. Chekulaev, E. Holland, J. E. Anthony, J. Zaumseil, A. J. Musser and J. Clark, Nat. Chem., 2021, 13, 163–171 Search PubMed.
- W.-L. Chan, M. Ligges and X.-Y. Zhu, Nat. Chem., 2012, 4, 840–845 Search PubMed.
- K. Miyata, F. S. Conrad-Burton, F. L. Geyer and X.-Y. Zhu, Chem. Rev., 2019, 119, 4261–4292 Search PubMed.
- A. J. Carrod, V. Gray and K. Börjesson, Energy Environ. Sci., 2022, 15, 4982–5016 Search PubMed.
- G. D. Scholes, J. Phys. Chem. A, 2015, 119, 12699–12705 Search PubMed.
- L. Nienhaus, Chimia, 2024, 78, 836–844 Search PubMed.
- T. N. Singh-Rachford and F. N. Castellano, Coord. Chem. Rev., 2010, 254, 2560–2573 Search PubMed.
- V. Gray, K. Moth-Poulsen, B. Albinsson and M. Abrahamsson, Coord. Chem. Rev., 2018, 362, 54–71 Search PubMed.
- H. Goudarzi and P. E. Keivanidis, ACS Appl. Mater. Interfaces, 2017, 9, 845–857 Search PubMed.
- R. Karpicz, S. Puzinas, V. Gulbinas, A. Vakhnin, A. Kadashchuk and B. P. Rand, Chem. Phys., 2014, 429, 57–62 Search PubMed.
- H. Goudarzi, L. Koutsokeras, A. H. Balawi, C. Sun, G. K. Manolis, N. Gasparini, Y. Peisen, G. Antoniou, S. Athanasopoulos, C. C. Tselios, P. Falaras, C. Varotsis, F. Laquai, J. Cabanillas-González and P. E. Keivanidis, Chem. Sci., 2023, 14, 2009–2023 Search PubMed.
- K. Chen, Q. Luan, T. Liu, B. Albinsson and L. Hou, Responsive Mater., 2025, 3, e20240030 Search PubMed.
- Z. A. VanOrman, C. M. Sullivan, C. W. Yang, J. Isokuortti and L. Nienhaus, Chem. Rev., 2025, 125, 11426–11460 Search PubMed.
- S. Maiti and L. D. A. Siebbeles, J. Phys. Chem. Lett., 2023, 14, 11168–11176 Search PubMed.
- S. Izawa and M. Hiramoto, Nat. Photonics, 2021, 15, 895–900 CrossRef CAS.
- R. Kani, K. Fujimoto, K. Banno, Y. Majima, M. Takahashi and S. Izawa, Jpn. J. Appl. Phys., 2025, 64, 091004 CrossRef CAS.
- R. Hamid, D. Feng, P. Narayanan, J. S. Edwards, M. Hu, E. Belliveau, M. Kim, S. Yin, S. Deshpande, C. Wan, L. Pucurimay, D. A. Czaplewski, D. N. Congreve and M. A. Kats, Adv. Funct. Mater., 2025, e15334 Search PubMed.
- Y. Sakamoto, S. Izawa, H. Ohkita, M. Hiramoto and Y. Tamai, Commun. Mater., 2022, 3, 76 Search PubMed.
- Z. A. VanOrman and L. Nienhaus, ACS Energy Lett., 2021, 6, 3686–3694 CrossRef CAS.
- J. Duan, Y. Liu, Y. Zhang, Z. Chen, X. Xu, L. Ye, Z. Wang, Y. Yang, D. Zhang and H. Zhu, Sci. Adv., 2022, 8, eabq4935 Search PubMed.
- S. Zhao, C. Sun, G. Xiang, Y. Zhang, S. He, S. Li, Y. Jin, D. Zhang and H. Zhu, Angew. Chem., Int. Ed., 2025, 64, e202420070 Search PubMed.
- M. Xu, C. Sun, G. Xiang, X. Shen, Y. Liu and H. Zhu, ACS Nano, 2025, 19, 36517–36526 Search PubMed.
- G. F. Stelmakh and M. P. Tsvirko, Opt. Spectrosc., 1980, 49, 278–281 Search PubMed.
- J. A. O'Brien, S. Rallabandi, U. Tripathy, M. F. Paige and R. P. Steer, Chem. Phys. Lett., 2009, 475, 220–222 Search PubMed.
- A. Arshad and F. N. Castellano, J. Phys. Chem. A, 2024, 128, 7648–7656 CrossRef CAS PubMed.
- R. P. Steer, Phys. Chem. Chem. Phys., 2023, 25, 23384–23394 RSC.
- Y. Bai, W. Ni, K. Sun, L. Chen, L. Ma, Y. Zhao, G. G. Gurzadyan and M. F. Gelin, J. Phys. Chem. Lett., 2022, 13, 11086–11094 CrossRef CAS.
- A. P. Demchenko, V. I. Tomin and P.-T. Chou, Chem. Rev., 2017, 117, 13353–13381 CrossRef CAS.
- T. Itoh, Chem. Rev., 2012, 112, 4541–4568 CrossRef CAS PubMed.
- V. Gray, D. Dzebo, M. Abrahamsson, B. Albinsson and K. Moth-Poulsen, Phys. Chem. Chem. Phys., 2014, 16, 10345–10352 RSC.
- Y. Zhou, F. N. Castellano, T. W. Schmidt and K. Hanson, ACS Energy Lett., 2020, 5, 2322–2326 CrossRef CAS.
- F. Edhborg, A. Olesund and B. Albinsson, Photochem. Photobiol. Sci., 2022, 21, 1143–1158 Search PubMed.
- Y. Murakami and K. Kamada, Phys. Chem. Chem. Phys., 2021, 23, 18268–18282 RSC.
- A. Monguzzi, J. Mezyk, F. Scotognella, R. Tubino and F. Meinardi, Phys. Rev. B: Condens. Matter Mater. Phys., 2008, 78, 195112 CrossRef.
- P. Bi, T. Zhang, Y. Guo, J. Wang, X. W. Chua, Z. Chen, W. P. Goh, C. Jiang, E. E. M. Chia, J. Hou and L. Yang, Nat. Commun., 2024, 15, 5719 CrossRef CAS.
- L. Wang, J. J. Yoo, T.-A. Lin, C. F. Perkinson, Y. Lu, M. A. Baldo and M. G. Bawendi, Adv. Mater., 2021, 33, 2100854 CrossRef CAS.
- L. Naimovicius, L. Wolek, S. K. Zhang, J. E. Kim, M. J. Tauber and A. B. Pun, J. Am. Chem. Soc., 2026, 148, 3811–3819 CrossRef.
- P. Baronas, J. Lekavičius, M. Majdecki, J. L. Elholm, K. Kazlauskas, P. Gaweł and K. Moth-Poulsen, ACS Cent. Sci., 2025, 11, 413–421 CrossRef CAS PubMed.
- L. Naimovičius, P. Bharmoria and K. Moth-Poulsen, Mater. Chem. Front., 2023, 7, 2297–2315 RSC.
- Y. J. Bae, G. Kang, C. D. Malliakas, J. N. Nelson, J. Zhou, R. M. Young, Y.-L. Wu, R. P. Van Duyne, G. C. Schatz and M. R. Wasielewski, J. Am. Chem. Soc., 2018, 140, 15140–15144 CrossRef CAS.
- V. Gray, A. Dreos, P. Erhart, B. Albinsson, K. Moth-Poulsen and M. Abrahamsson, Phys. Chem. Chem. Phys., 2017, 19, 10931–10939 RSC.
- K. J. Fallon, E. M. Churchill, S. N. Sanders, J. Shee, J. L. Weber, R. Meir, S. Jockusch, D. R. Reichman, M. Y. Sfeir, D. N. Congreve and L. M. Campos, J. Am. Chem. Soc., 2020, 142, 19917–19925 CrossRef CAS PubMed.
- A. B. Pun, L. M. Campos and D. N. Congreve, J. Am. Chem. Soc., 2019, 141, 3777–3781 CrossRef CAS.
- E. Radiunas, L. Naimovičius, P. Baronas, A. Jozeliūnaitė, E. Orentas and K. Kazlauskas, Adv. Opt. Mater., 2025, 13, 2403032 CrossRef CAS.
- C. Ye, S. Mallick, M. Hertzog, M. Kowalewski and K. Börjesson, J. Am. Chem. Soc., 2021, 143, 7501–7508 CrossRef CAS.
- N. J. Hestand and F. C. Spano, Chem. Rev., 2018, 118, 7069–7163 Search PubMed.
- C. M. Sullivan and L. Nienhaus, Chem. Mater., 2024, 36, 417–424 CrossRef CAS.
- C. M. Sullivan, A. M. Szucs, T. Siegrist and L. Nienhaus, J. Phys. Chem. C, 2024, 128, 19248–19259 CrossRef CAS.
- S. Li, D. Zhou, Z. Yu, L. Ma, X. Du and D. Qiu, J. Mol. Struct., 2026, 1351, 144189 CrossRef CAS.
- A. Monguzzi, R. Tubino, S. Hoseinkhani, M. Campione and F. Meinardi, Phys. Chem. Chem. Phys., 2012, 14, 4322–4332 RSC.
- Y. Y. Cheng, B. Fückel, T. Khoury, R. G. C. R. Clady, M. J. Y. Tayebjee, N. J. Ekins-Daukes, M. J. Crossley and T. W. Schmidt, J. Phys. Chem. Lett., 2010, 1, 1795–1799 CrossRef CAS.
- Y. Yap Cheng, T. Khoury, R. G. C. R. Clady, M. J. Y. Tayebjee, N. J. Ekins-Daukes, M. J. Crossley and T. W. Schmidt, Phys. Chem. Chem. Phys., 2010, 12, 66–71 RSC.
- J. E. Auckett, Y. Y. Chen, T. Khoury, R. G. C. R. Clady, N. J. Ekins-Daukes, M. J. Crossley and T. W. Schmidt, J. Phys.: Conf. Ser., 2009, 185, 012002 CrossRef.
- T. W. Schmidt and F. N. Castellano, J. Phys. Chem. Lett., 2014, 5, 4062–4072 CrossRef CAS PubMed.
- J. A. Moghtader, M. Uji, T. J. B. Zähringer, M. Schmitz, L. M. Carrella, A. Heckel, E. Rentschler, N. Yanai and C. Kerzig, JACS Au, 2025, 5, 5707–5716 CrossRef CAS PubMed.
- M. B. Smith and J. Michl, Chem. Rev., 2010, 110, 6891–6936 CrossRef CAS.
- T. J. Penfold, E. Gindensperger, C. Daniel and C. M. Marian, Chem. Rev., 2018, 118, 6975–7025 CrossRef CAS PubMed.
- A. J. Carrod, A. Cravcenco, C. Ye and K. Börjesson, J. Mater. Chem. C, 2022, 10, 4923–4928 RSC.
- L. Naimovičius, E. Radiunas, B. Chatinovska, A. Jozeliūnaitė, E. Orentas and K. Kazlauskas, J. Mater. Chem. C, 2023, 11, 698–704 RSC.
- S. Liu, T. Gou, X. Song, R. Hu, H. Liu, X. Li and X. Jiang, ChemPhysMater, 2024, 3, 187–193 CrossRef.
- N. Nishimura, V. Gray, J. R. Allardice, Z. Zhang, A. Pershin, D. Beljonne and A. Rao, ACS Mater. Lett., 2019, 1, 660–664 CrossRef CAS.
- X. Wang, R. Tom, X. Liu, D. N. Congreve and N. Marom, J. Mater. Chem. C, 2020, 8, 10816–10824 RSC.
- X. Wang and N. Marom, Mol. Syst. Des. Eng., 2022, 7, 889–898 RSC.
- C.-J. Chiang, A. Kimyonok, M. K. Etherington, G. C. Griffiths, V. Jankus, F. Turksoy and A. P. Monkman, Adv. Funct. Mater., 2013, 23, 739–746 CrossRef CAS.
- S. Gao, Y. Luo, X. Liu and N. Marom, Digital Discovery, 2025, 4, 1306–1322 RSC.
- C. F. Perkinson, D. P. Tabor, M. Einzinger, D. Sheberla, H. Utzat, T.-A. Lin, D. N. Congreve, M. G. Bawendi, A. Aspuru-Guzik and M. A. Baldo, J. Chem. Phys., 2019, 151, 121102 CrossRef PubMed.
- R. Grotjahn, T. M. Maier, J. Michl and M. Kaupp, J. Chem. Theory Comput., 2017, 13, 4984–4996 CrossRef CAS.
- J. Wen, Z. Havlas and J. Michl, J. Am. Chem. Soc., 2015, 137, 165–172 CrossRef CAS.
- L. Shen, Q. Wu, J. Lu, H. Zhao, H. Liu, Q. Meng and X. Li, J. Mater. Chem. C, 2022, 10, 10404–10411 RSC.
- D. Padula, Ö. H. Omar, T. Nematiaram and A. Troisi, Energy Environ. Sci., 2019, 12, 2412–2416 RSC.
- C.-H. Li and D. P. Tabor, Chem. Sci., 2023, 14, 11045–11055 RSC.
- A. Akdag, Z. Havlas and J. Michl, J. Am. Chem. Soc., 2012, 134, 14624–14631 Search PubMed.
- T. Zeng, N. Ananth and R. Hoffmann, J. Am. Chem. Soc., 2014, 136, 12638–12647 CrossRef CAS.
- F. J. Hernández and R. Crespo-Otero, Annu. Rev. Phys. Chem., 2023, 74, 547–571 CrossRef.
- M. Ogbaje, V. Bhat and C. Risko, Annu. Rev. Mater. Res., 2025, 55, 285–306 CrossRef.
- Y. Xu, X. Liang, X. Zhou, P. Yuan, J. Zhou, C. Wang, B. Li, D. Hu, X. Qiao, X. Jiang, L. Liu, S.-J. Su, D. Ma and Y. Ma, Adv. Mater., 2019, 31, 1807388 CrossRef.
- X. Tang, R. Pan, X. Zhao, H. Zhu and Z. Xiong, J. Phys. Chem. Lett., 2020, 11, 2804–2811 CrossRef CAS.
- D. G. Bossanyi, Y. Sasaki, S. Wang, D. Chekulaev, N. Kimizuka, N. Yanai and J. Clark, JACS Au, 2021, 1, 2188–2201 CrossRef CAS.
- Y. Wang, Y. Ning, F. Wu, J. Chen, X. Chen and Z. Xiong, Adv. Funct. Mater., 2022, 32, 2202882 CrossRef CAS.
- Z. Deng, C. Huang, Y. Luo, J. He, L. Li, X. Pang, G. Huang and D. L. Phillips, Nat. Commun., 2025, 16, 258 CrossRef.
- J. Chen, H. Liu, J. Guo, J. Wang, N. Qiu, S. Xiao, J. Chi, D. Yang, D. Ma, Z. Zhao and B. Z. Tang, Angew. Chem., Int. Ed., 2022, 61, e202116810 Search PubMed.
- S. Du, M. Luo, D. Li, L. Lyu, W. Li, M. Zhao, Z. Wang, J. Zhang, D. Liu, Y. Li, S.-J. Su and Z. Ge, Adv. Mater., 2023, 35, 2303304 CrossRef CAS.
- C. Lin, P. Han, S. Xiao, F. Qu, J. Yao, X. Qiao, D. Yang, Y. Dai, Q. Sun, D. Hu, A. Qin, Y. Ma, B. Z. Tang and D. Ma, Adv. Funct. Mater., 2021, 31, 2106912 CrossRef CAS.
- Y. Luo, K. Zhang, Z. Ding, P. Chen, X. Peng, Y. Zhao, K. Chen, C. Li, X. Zheng, Y. Huang, X. Pu, Y. Liu, S.-J. Su, X. Hou and Z. Lu, Nat. Commun., 2022, 13, 6892 Search PubMed.
- Y. Gao, Y. Deng, Y. Lv, X. Tian, H. Liu, S.-T. Zhang, J. Sheng and B. Yang, Chem. Eng. J., 2024, 481, 148725 CrossRef CAS.
- L. Xu, Y. Yu, M. Li, Y. Li, W. Tan, B. Wang, D. Yang, D. Hu, L. Ying and Y. Ma, Adv. Opt. Mater., 2024, 12, 2401275 CrossRef CAS.
- A. Olesund, J. Johnsson, F. Edhborg, S. Ghasemi, K. Moth-Poulsen and B. Albinsson, J. Am. Chem. Soc., 2022, 144, 3706–3716 CrossRef CAS.
- T. Miyashita, P. Jaimes, A. Mardini, M. Fumanal and M. L. Tang, J. Phys. Chem. Lett., 2023, 14, 6119–6126 CrossRef CAS.
- J. Shao, Y. Zhang, H. Liu, D. Duan, Z. Sun, Z. Lu, Y. Wei and X. Xu, J. Chem. Phys., 2025, 163, 064503 CrossRef CAS PubMed.
- C. A. Kim, S. Hu and T. Van Voorhis, J. Phys. Chem. A, 2023, 127, 7175–7185 CrossRef CAS.
- A. J. Musser and J. Clark, Annu. Rev. Phys. Chem., 2019, 70, 323–351 CrossRef CAS PubMed.
- L. R. Weiss, S. L. Bayliss, F. Kraffert, K. J. Thorley, J. E. Anthony, R. Bittl, R. H. Friend, A. Rao, N. C. Greenham and J. Behrends, Nat. Phys., 2017, 13, 176–181 Search PubMed.
- S. L. Bayliss, L. R. Weiss, A. Rao, R. H. Friend, A. D. Chepelianskii and N. C. Greenham, Phys. Rev. B, 2016, 94, 045204 Search PubMed.
- R. C. Johnson, R. E. Merrifield, P. Avakian and R. B. Flippen, Phys. Rev. Lett., 1967, 19, 285–287 CrossRef CAS.
- J. Mezyk, R. Tubino, A. Monguzzi, A. Mech and F. Meinardi, Phys. Rev. Lett., 2009, 102, 087404 Search PubMed.
- P. C. Tapping and D. M. Huang, J. Phys. Chem. C, 2016, 120, 25151–25157 CrossRef CAS.
- K. Miwa, T. Okamoto, Y. Kobori and A. Ishizaki, J. Phys. Chem. C, 2025, 129, 12904–12915 CrossRef.
- T. L. Keevers and D. R. McCamey, Phys. Rev. B, 2016, 93, 045210 CrossRef.
- J. Feng, J. Alves, D. M. De Clercq and T. W. Schmidt, Annu. Rev. Phys. Chem., 2023, 74, 145–168 CrossRef CAS PubMed.
- R. Sato, H. Kitoh-Nishioka, T. Yanai and Y. Shigeta, Chem. Lett., 2017, 46, 873–875 CrossRef CAS.
- R. Sato, H. Kitoh-Nishioka, K. Kamada, T. Mizokuro, K. Kobayashi and Y. Shigeta, J. Phys. Chem. C, 2018, 122, 5334–5340 CrossRef CAS.
- T. Okamoto, S. Izawa, M. Hiramoto and Y. Kobori, J. Phys. Chem. Lett., 2024, 15, 2966–2975 CrossRef CAS.
- E. J. Taffet, D. Beljonne and G. D. Scholes, J. Am. Chem. Soc., 2020, 142, 20040–20047 CrossRef CAS.
- L. Naimovičius, M. Dapkevičius, E. Radiunas, M. Miroshnichenko, G. Kreiza, C. Alcaide, P. Baronas, Y. Sasaki, N. Yanai, N. Kimizuka, A. B. Pun, M. Solà, P. Bharmoria, K. Kazlauskas and K. Moth-Poulsen, Chem Sci, 2025, 16(43), 20255–20264, 10.1039/d5sc05248c41098493.
- N. Monahan and X.-Y. Zhu, Annu. Rev. Phys. Chem., 2015, 66, 601–618 Search PubMed.
- I. Papadopoulos, M. J. Álvaro-Martins, D. Molina, P. M. McCosker, P. A. Keller, T. Clark, Á. Sastre-Santos and D. M. Guldi, Adv. Energy Mater., 2020, 10, 2001496 CrossRef CAS.
- J. C. Johnson, A. Akdag, M. Zamadar, X. Chen, A. F. Schwerin, I. Paci, M. B. Smith, Z. Havlas, J. R. Miller, M. A. Ratner, A. J. Nozik and J. Michl, J. Phys. Chem. B, 2013, 117, 4680–4695 CrossRef CAS.
- A. M. Alvertis, S. Lukman, T. J. H. Hele, E. G. Fuemmeler, J. Feng, J. Wu, N. C. Greenham, A. W. Chin and A. J. Musser, J. Am. Chem. Soc., 2019, 141, 17558–17570 CrossRef CAS.
- T. Tonami, R. Sugimori, R. Sakai, K. Tokuyama, H. Miyamoto and M. Nakano, Phys. Chem. Chem. Phys., 2021, 23, 11624–11634 RSC.
- A. R. Srimath Kandada, A. Petrozza and G. Lanzani, Phys. Rev. B: Condens. Matter Mater. Phys., 2014, 90, 075310 CrossRef.
- Z. Sun, T. Chen, J. Luo and M. Hong, Angew. Chem., Int. Ed., 2012, 51, 3871–3876 CrossRef CAS PubMed.
- G. Fan, X. Yang, R. Liang, J. Zhao, S. Li and D. Yan, CrystEngComm, 2016, 18, 240–249 Search PubMed.
- P. E. Hartnett, E. A. Margulies, C. M. Mauck, S. A. Miller, Y. Wu, Y.-L. Wu, T. J. Marks and M. R. Wasielewski, J. Phys. Chem. B, 2016, 120, 1357–1366 CrossRef CAS.
- Y. Bo, Y. Hou, D. Thiel, R. Weiß, T. Clark, M. J. Ferguson, R. R. Tykwinski and D. M. Guldi, J. Am. Chem. Soc., 2023, 145, 18260–18275 CrossRef CAS.
- A. T. Gilligan, R. Owens, E. G. Miller, N. F. Pompetti and N. H. Damrauer, Chem. Sci., 2024, 15, 1283–1296 Search PubMed.
- F. C. Spano and C. Silva, Annu. Rev. Phys. Chem., 2014, 65, 477–500 CrossRef CAS PubMed.
- H. Zhang, Z. Zhao, A. T. Turley, L. Wang, P. R. McGonigal, Y. Tu, Y. Li, Z. Wang, R. T. K. Kwok, J. W. Y. Lam and B. Z. Tang, Adv. Mater., 2020, 32, 2001457 Search PubMed.
- Z. Shuai, Q. Sun, J. Ren, T. Jiang and W. Li, Aggregate, 2025, 6, e70013 CrossRef CAS.
- A. P. Deshmukh, N. Geue, N. C. Bradbury, T. L. Atallah, C. Chuang, M. Pengshung, J. Cao, E. M. Sletten, D. Neuhauser and J. R. Caram, Chem. Phys. Rev., 2022, 3, 021401 CrossRef CAS.
- A. P. Deshmukh, D. Koppel, C. Chuang, D. M. Cadena, J. Cao and J. R. Caram, J. Phys. Chem. C, 2019, 123, 18702–18710 CrossRef CAS.
- Z. Xie, D. Liu, C. Gao, X. Zhang, H. Dong and W. Hu, J. Am. Chem. Soc., 2025, 147, 2239–2256 Search PubMed.
- O. Ostroverkhova, Chem. Rev., 2016, 116, 13279–13412 CrossRef CAS.
- R. P. Butler and M. J. Pilling, J. Chem. Soc., Faraday Trans. 2, 1977, 73, 886–894 RSC.
- J. Saltiel, G. R. March, W. K. Smothers, S. A. Stout and J. L. Charlton, J. Am. Chem. Soc., 1981, 103, 7159–7164 Search PubMed.
- K. R. Naqvi, H. Van Willigen and D. A. Capitanio, Chem. Phys. Lett., 1978, 57, 197–201 CrossRef.
- H. Yamagata, D. S. Maxwell, J. Fan, K. R. Kittilstved, A. L. Briseno, M. D. Barnes and F. C. Spano, J. Phys. Chem. C, 2014, 118, 28842–28854 Search PubMed.
- A. L. Bialas and F. C. Spano, J. Phys. Chem. C, 2022, 126, 4067–4081 Search PubMed.
- Y. Hong, W. Kim, T. Kim, C. Kaufmann, H. Kim, F. Würthner and D. Kim, Angew. Chem., Int. Ed., 2022, 61, e202114474 CrossRef CAS PubMed.
- Y. Hong, M. Rudolf, M. Kim, J. Kim, T. Schembri, A.-M. Krause, K. Shoyama, D. Bialas, M. I. S. Röhr, T. Joo, H. Kim, D. Kim and F. Würthner, Nat. Commun., 2022, 13, 4488 CrossRef CAS.
- J. P. O'Connor, J. D. Schultz, N. A. Tcyrulnikov, T. Kim, R. M. Young and M. R. Wasielewski, J. Chem. Phys., 2024, 161, 074306 Search PubMed.
- S. Hammer, T. L. Britt, L. Kremeyer, M. Rödel, D. Cai, J. Pflaum and B. J. Siwick, Proc. Natl. Acad. Sci. U. S. A., 2024, 121, e2411975121 Search PubMed.
- H. Haghshenas, A. Bialas and F. C. Spano, J. Phys. Chem. C, 2025, 129, 6397–6413 Search PubMed.
- M. Deutsch, S. Wirsing, D. Kaiser, R. F. Fink, P. Tegeder and B. Engels, J. Chem. Phys., 2020, 153, 224104 Search PubMed.
- L. Naimovičius and A. B. Pun, Trends Chem., 2025, 7, 171–174 Search PubMed.
- S. Wang, H. Liu, S. Zhao, Q. Wu, Z. Yang, D. Yang, Y. Lv, Q. Su, S.-T. Zhang and B. Yang, Chem. Sci., 2025, 16, 3275–3284 Search PubMed.
- C. B. Dover, J. K. Gallaher, L. Frazer, P. C. Tapping, A. J. Petty, M. J. Crossley, J. E. Anthony, T. W. Kee and T. W. Schmidt, Nat. Chem., 2018, 10, 305–310 CrossRef CAS.
- J. Isokuortti, C. J. O'Dea, S. R. Allen, S. Vasylevskyi, Z. A. Page and S. T. Roberts, Adv. Opt. Mater., 2025, 13, 2500388 Search PubMed.
- X. Chen, J. Zhou, Z. Xie and Y. Ma, Inf. Funct. Mater., 2024, 1, 68–86 Search PubMed.
- W. Li and Z. Xie, Chem.–Eur. J., 2025, 31, e202403908 Search PubMed.
- R. M. Young and M. R. Wasielewski, Acc. Chem. Res., 2020, 53, 1957–1968 CrossRef CAS.
- Z. Ye, N. Wang, J. Zhou and D. Ouyang, Innovation, 2024, 5(2), 100562 Search PubMed.
- F. Ren, X. Chen and F. Liu, J. Phys. Chem. Lett., 2025, 16, 2541–2552 Search PubMed.
- Z. Zhao, H. Zhang, J. W. Y. Lam and B. Z. Tang, Angew. Chem., Int. Ed., 2020, 59, 9888–9907 CrossRef CAS PubMed.
- Q. Peng and Z. Shuai, Aggregate, 2021, 2, e91 Search PubMed.
- L. Li, J. Wang, J. Zou, J. Hu, S. Liu, S. Wan, Y. Shi, Z. Liang, X. Wang and C. Ye, Chem.–Eur. J., 2025, 31, e202500553 CrossRef CAS PubMed.
- L. Li, Y. Zeng, T. Yu, J. Chen, G. Yang and Y. Li, ChemSusChem, 2017, 10, 4610–4615 CrossRef CAS PubMed.
- P. Duan, N. Yanai, Y. Kurashige and N. Kimizuka, Angew. Chem., Int. Ed., 2015, 54, 7544–7549 Search PubMed.
- C. Petermayer and H. Dube, Acc. Chem. Res., 2018, 51, 1153–1163 Search PubMed.
- M. Ikegami and T. Arai, Chem. Lett., 2005, 34, 492–493 CrossRef CAS.
- Š. Budzák, J. Jovaišaitė, C.-Y. Huang, P. Baronas, K. Tulaitė, S. Juršėnas, D. Jacquemin and S. Hecht, Chem.–Eur. J., 2022, 28, e202200496 Search PubMed.
- C. Yang, Q. T. Trinh, X. Wang, Y. Tang, K. Wang, S. Huang, X. Chen, S. H. Mushrif and M. Wang, Chem. Commun., 2015, 51, 3375–3378 Search PubMed.
- J. S. Seixas de Melo, H. D. Burrows, C. Serpa and L. G. Arnaut, Angew. Chem., Int. Ed., 2007, 46, 2094–2096 CrossRef CAS PubMed.
- L. Wang, S. Bai, Y. Wu, Y. Liu, J. Yao and H. Fu, Angew. Chem., Int. Ed., 2020, 59, 2003–2007 CrossRef CAS.
- M. Irimia-Vladu, E. D. Głowacki, P. A. Troshin, G. Schwabegger, L. Leonat, D. K. Susarova, O. Krystal, M. Ullah, Y. Kanbur, M. A. Bodea, V. F. Razumov, H. Sitter, S. Bauer and N. S. Sariciftci, Adv. Mater., 2012, 24, 375–380 Search PubMed.
- J. Park, M. Xu, F. Li and H.-C. Zhou, J. Am. Chem. Soc., 2018, 140, 5493–5499 CrossRef CAS PubMed.
- M.-Y. Zhang, H.-J. Feng, J.-Y. Li, L.-H. Jiang, A.-X. Ma, L. Zeng, L. Huang and D.-W. Pang, Adv. Mater., 2025, 37, 2502150 CrossRef CAS.
- A. Ronchi and A. Monguzzi, J. Appl. Phys., 2021, 129, 050901 CrossRef CAS.
- B. T. Luppi, D. Majak, M. Gupta, E. Rivard and K. Shankar, J. Mater. Chem. A, 2019, 7, 2445–2463 RSC.
- M. A. El-Sayed, J. Chem. Phys., 1963, 38, 2834–2838 Search PubMed.
- C. M. Marian, Annu. Rev. Phys. Chem., 2021, 72, 617–640 Search PubMed.
- S. H. Lin and R. Bersohn, J. Chem. Phys., 1968, 48, 2732–2736 Search PubMed.
- R. J. Watts and S. J. Strickler, J. Chem. Phys., 1968, 49, 3867–3871 CrossRef CAS.
- A. Danos, R. W. MacQueen, Y. Y. Cheng, M. Dvořák, T. A. Darwish, D. R. McCamey and T. W. Schmidt, J. Phys. Chem. Lett., 2015, 6, 3061–3066 CrossRef CAS.
- J.-F. Cheng, F.-C. Kong, K. Zhang, J.-H. Cai, Y. Zhao, C.-K. Wang, J. Fan and L.-S. Liao, Chem. Eng. J., 2022, 430, 132822 Search PubMed.
- S. Hirata, K. Totani, T. Watanabe, H. Kaji and M. Vacha, Chem. Phys. Lett., 2014, 591, 119–125 CrossRef CAS.
- B. M. T. C. Peluzo, R. Meena, L. Catalano, G. Schweicher and M. T. Ruggiero, Angew. Chem., Int. Ed., 2025, 64, e202507566 CrossRef CAS PubMed.
- S. Hirata, Appl. Phys. Rev., 2022, 9, 011304 CAS.
- Q. Peng, Y. Niu, C. Deng and Z. Shuai, Chem. Phys., 2010, 370, 215–222 CrossRef CAS.
- C. Müller, Š. Sršeň, B. Bachmair, R. Crespo-Otero, J. Li, S. Mausenberger, M. Pinheiro, G. Worth, S. A. Lopez and J. Westermayr, Chem. Sci., 2025, 16, 17542–17567 RSC.
- Z. Li, F. J. Hernández, C. Salguero, S. A. Lopez, R. Crespo-Otero and J. Li, Nat. Commun., 2025, 16, 1194 CrossRef CAS.
- J. Li and S. A. Lopez, Chem. Phys. Rev., 2023, 4, 031309 CrossRef CAS.
- E. C. Lim, Acc. Chem. Res., 1987, 20, 8–17 CrossRef CAS.
- A. Olesund, S. Ghasemi, K. Moth-Poulsen and B. Albinsson, J. Am. Chem. Soc., 2023, 145, 22168–22175 CrossRef CAS PubMed.
- J. Saltiel, G. R. Marchand, R. Dabestani and J. M. Pecha, Chem. Phys. Lett., 1983, 100, 219–222 CrossRef CAS.
- O. L. J. Gijzeman, J. Langelaar and J. D. W. Van Voorst, Chem. Phys. Lett., 1970, 5, 269–272 CrossRef CAS.
- B. Feingold, N. F. Pompetti, M. Martinez, T. J. Aubry, J. L. Blackburn, O. G. Reid, M. C. Beard and J. C. Johnson, ACS Nano, 2025, 19, 40245–40257 CrossRef CAS PubMed.
- A. Kalpattu, D. E. Falvey and J. T. Fourkas, Phys. Chem. Chem. Phys., 2025, 27, 11000–11016 RSC.
- L. Martinez-Fernandez, P. Wu, L.-T. Bao, X. Wang, R.-H. Zhang, W. Wang, H.-B. Yang, J. Chen and R. Improta, Chem. Sci., 2025, 16, 4469–4479 RSC.
- M. Pabst, B. Lunkenheimer and A. Köhn, J. Phys. Chem. C, 2011, 115, 8335–8344 CrossRef CAS.
- H. T. I. Etheridge and R. B. Weisman, J. Phys. Chem., 1995, 99, 2782–2787 CrossRef CAS.
- J. Langelaar, G. Jansen, R. P. H. Rettschnick and G. J. Hoytink, Chem. Phys. Lett., 1971, 12, 86–91 CrossRef CAS.
- J. A. Tanaka, H. Masuhara and N. Mataga, Polym. J., 1986, 18, 181–184 Search PubMed.
- M. Yamaji, H. Tsukada, J. Nishimura, H. Shizuka and S. Tobita, Chem. Phys. Lett., 2002, 357, 137–142 Search PubMed.
- Y. Y. Cheng, B. Fückel, T. Khoury, R. G. C. R. Clady, N. J. Ekins-Daukes, M. J. Crossley and T. W. Schmidt, J. Phys. Chem. A, 2011, 115, 1047–1053 Search PubMed.
- C. M. Sullivan, J. E. Kuszynski, A. Kovalev, T. Siegrist, R. D. Schaller, G. F. Strouse and L. Nienhaus, Nanoscale, 2023, 15, 18832–18841 Search PubMed.
- C. M. Sullivan and L. Nienhaus, Nanoscale, 2023, 15, 998–1013 Search PubMed.
- G. D. Scholes, Annu. Rev. Phys. Chem., 2003, 54, 57–87 CrossRef CAS PubMed.
- S. Bai, P. Zhang and D. N. Beratan, J. Phys. Chem. C, 2020, 124, 18956–18960 CrossRef CAS.
- S. Saha, M. J. Mackintosh, L. M. Thompson and P. M. Kozlowski, J. Phys. Chem. A, 2025, 129, 967–977 CrossRef CAS PubMed.
- R. Sato, H. Kitoh-Nishioka, K. Kamada, T. Mizokuro, K. Kobayashi and Y. Shigeta, J. Phys. Chem. Lett., 2018, 9, 6638–6643 Search PubMed.
- H. Tamura, J. Phys. Chem. A, 2020, 124, 7943–7949 CrossRef CAS PubMed.
- E. A. Buchanan and J. Michl, J. Am. Chem. Soc., 2017, 139, 15572–15575 CrossRef CAS PubMed.
- A. Diaz-Andres, C. Tonnelé and D. Casanova, J. Chem. Theory Comput., 2024, 20, 4288–4297 CrossRef CAS PubMed.
- T. S. Volek, Z. T. Armstrong, J. K. Sowa, K. S. Wilson, M. Bohlmann Kunz, K. Bera, M. Koble, R. R. Frontiera, P. J. Rossky, M. T. Zanni and S. T. Roberts, J. Phys. Chem. Lett., 2023, 14, 11497–11505 CrossRef CAS PubMed.
- L. Xue, X. Song and Y. Bu, J. Phys. Chem. Lett., 2025, 16, 1956–1972 CrossRef CAS PubMed.
- W. Popp, D. Brey, R. Binder and I. Burghardt, Annu. Rev. Phys. Chem., 2021, 72, 591–616 CrossRef CAS PubMed.
- Y. Deng, L. Jiang, L. Huang and T. Zhu, ACS Energy Lett., 2022, 7, 847–861 Search PubMed.
- M. S. Dörfler, H. Bässler, A. Kadashchuk, H. Oberhofer and A. Köhler, J. Mater. Chem. C, 2025, 13, 14962–14971 RSC.
- R. J. Hudson, A. N. Stuart, D. M. Huang and T. W. Kee, J. Phys. Chem. C, 2022, 126, 5369–5377 CrossRef CAS.
- S. Fratini, S. Ciuchi, D. Mayou, G. T. de Laissardière and A. Troisi, Nat. Mater., 2017, 16, 998–1002 Search PubMed.
- T. Zhu and L. Huang, J. Phys. Chem. Lett., 2018, 9, 6502–6510 CrossRef CAS PubMed.
- G. Schweicher, G. D'Avino, M. T. Ruggiero, D. J. Harkin, K. Broch, D. Venkateshvaran, G. Liu, A. Richard, C. Ruzié, J. Armstrong, A. R. Kennedy, K. Shankland, K. Takimiya, Y. H. Geerts, J. A. Zeitler, S. Fratini and H. Sirringhaus, Adv. Mater., 2019, 31, 1902407 CrossRef CAS PubMed.
- P. G. Lynch, S. Baughman, T. N. Mihm, S. Sharifzadeh and R. R. Frontiera, J. Am. Chem. Soc., 2025, 147, 23705–23714 Search PubMed.
- A. J. Carrod, A. M. Berghuis, V. N. Gopalakrishnan, A. Monkman, A. Danos and K. Börjesson, Chem. Sci., 2025, 16, 1293–1301 Search PubMed.
- S. Raišys, O. Adomėnienė, P. Adomėnas, A. Rudnick, A. Köhler and K. Kazlauskas, J. Phys. Chem. C, 2021, 125, 3764–3775 CrossRef.
- S. Singh, W. J. Jones, W. Siebrand, B. P. Stoicheff and W. G. Schneider, J. Chem. Phys., 1965, 42, 330–342 CrossRef CAS.
- C. E. Swenberg and W. T. Stacy, Chem. Phys. Lett., 1968, 2, 327–328 CrossRef CAS.
- A. B. Kolomeisky, X. Feng and A. I. Krylov, J. Phys. Chem. C, 2014, 118, 5188–5195 CrossRef CAS.
- H. L. Stern, A. Cheminal, S. R. Yost, K. Broch, S. L. Bayliss, K. Chen, M. Tabachnyk, K. Thorley, N. Greenham, J. M. Hodgkiss, J. Anthony, M. Head-Gordon, A. J. Musser, A. Rao and R. H. Friend, Nat. Chem., 2017, 9, 1205–1212 Search PubMed.
- N. A. Pace, W. Zhang, D. H. Arias, I. McCulloch, G. Rumbles and J. C. Johnson, J. Phys. Chem. Lett., 2017, 8, 6086–6091 Search PubMed.
- N. V. Korovina, C. H. Chang and J. C. Johnson, Nat. Chem., 2020, 12, 391–398 Search PubMed.
- V. Abraham and N. J. Mayhall, J. Phys. Chem. Lett., 2017, 8, 5472–5478 CrossRef CAS PubMed.
- A. Singh and M. I. S. Röhr, J. Chem. Theory Comput., 2024, 20, 8624–8633 CrossRef CAS.
- N. Elfers, I. Lyskov, J. D. Spiegel and C. M. Marian, J. Phys. Chem. C, 2016, 120, 13901–13910 Search PubMed.
- H. Kim and P. M. Zimmerman, Phys. Chem. Chem. Phys., 2018, 20, 30083–30094 RSC.
- S. Lukman, J. M. Richter, L. Yang, P. Hu, J. Wu, N. C. Greenham and A. J. Musser, J. Am. Chem. Soc., 2017, 139, 18376–18385 CrossRef CAS PubMed.
- L. Mencaroni, F. Elisei, A. Marrocchi, A. Spalletti and B. Carlotti, J. Phys. Chem. B, 2024, 128, 3442–3453 Search PubMed.
- T. S. Lee, Y. L. Lin, H. Kim, R. D. Pensack, B. P. Rand and G. D. Scholes, J. Phys. Chem. Lett., 2018, 9, 4087–4095 CrossRef CAS PubMed.
- N. V. Korovina, S. Das, Z. Nett, X. Feng, J. Joy, R. Haiges, A. I. Krylov, S. E. Bradforth and M. E. Thompson, J. Am. Chem. Soc., 2016, 138, 617–627 CrossRef CAS PubMed.
- E. M. Bu Ali, A. Bertran, G. Moise, S. Wang, R. C. Kilbride, J. E. Anthony, C. E. Tait and J. Clark, J. Am. Chem. Soc., 2025, 147, 28638–28650 CrossRef CAS PubMed.
- I. Breen, R. Tempelaar, L. A. Bizimana, B. Kloss, D. R. Reichman and D. B. Turner, J. Am. Chem. Soc., 2017, 139, 11745–11751 CrossRef CAS PubMed.
- C. M. Sullivan, A. M. Szucs, A. P. Cantrell, K. E. Shulenberger, T. Siegrist and L. Nienhaus, Adv. Energy Mater., 2025, 15, 2404130 CrossRef CAS.
- A. Nandi, B. Manna and R. Ghosh, J. Photochem. Photobiol., A, 2026, 474, 116942 Search PubMed.
- T. S. Volek, M. A. Verkamp, G. N. Ruiz, A. J. Staat, B. C. Li, M. J. Rose, J. D. Eaves and S. T. Roberts, J. Am. Chem. Soc., 2024, 146, 29575–29587 CrossRef CAS.
- S. Wieghold, A. S. Bieber, Z. A. VanOrman, A. Rodriguez and L. Nienhaus, J. Phys. Chem. C, 2020, 124, 18132–18140 CrossRef CAS.
- P. Baronas, G. Kreiza, L. Naimovičius, E. Radiunas, K. Kazlauskas, E. Orentas and S. Juršėnas, J. Phys. Chem. C, 2022, 126, 15327–15335 CrossRef CAS PubMed.
- A. Sawa, S. Shimada, N. Tripathi, C. Heck, H. Tachibana, E. Koyama, T. Mizokuro, Y. Hirao, T. Kubo, N. Tamai, D. Kuzuhara, H. Yamada and K. Kamada, J. Mater. Chem. C, 2023, 11, 8502–8513 Search PubMed.
- J. Kim, D. C. Bain, V. Ding, K. Majumder, D. Windemuller, J. Feng, J. Wu, S. Patil, J. Anthony, W. Kim and A. J. Musser, Nat. Chem., 2024, 16, 1680–1686 Search PubMed.
- E. A. Margulies, C. E. Miller, Y. Wu, L. Ma, G. C. Schatz, R. M. Young and M. R. Wasielewski, Nat. Chem., 2016, 8, 1120–1125 CrossRef CAS.
- J. Feng, P. Hosseinabadi, D. M. de Clercq, B. P. Carwithen, M. P. Nielsen, M. W. Brett, S. K. K. Prasad, A. A. D. Farahani, H. L. Li, S. N. Sanders, J. E. Beves, N. J. Ekins-Daukes, J. H. Cole, P. Thordarson, D. M. Huang, M. J. Y. Tayebjee and T. W. Schmidt, Nat. Chem., 2024, 16, 1861–1867 Search PubMed.
- C. Ye, V. Gray, J. Mårtensson and K. Börjesson, J. Am. Chem. Soc., 2019, 141, 9578–9584 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |




![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200
200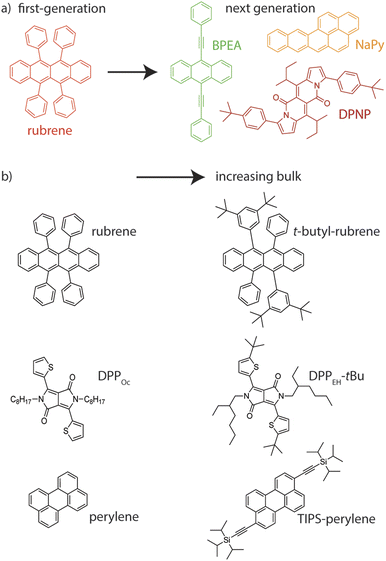



![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond is hindered, leading to potential aggregation-induced emission properties.
C bond is hindered, leading to potential aggregation-induced emission properties.

